Draw A Second Resonance Structure For The Following Radical
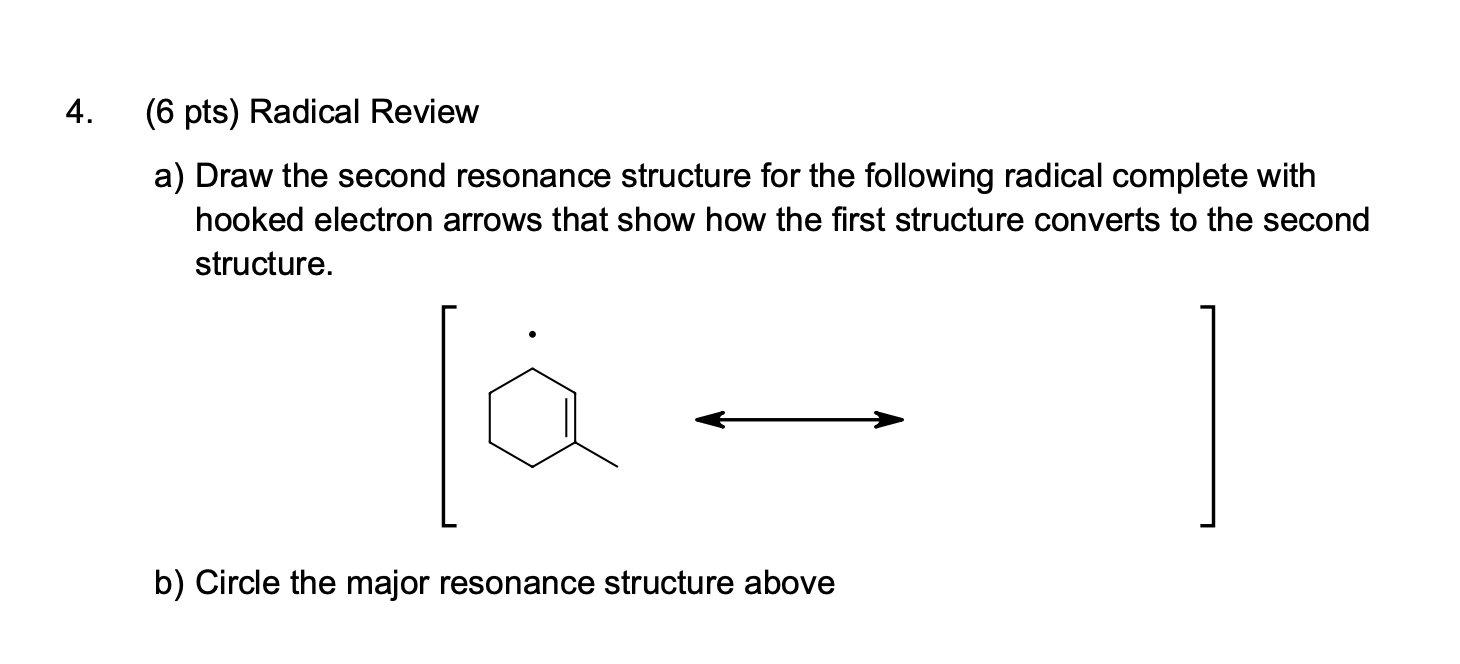
Ever looked at a molecule and thought, "Hmm, that looks a bit…unsettled"? Well, welcome to the wonderfully wiggly world of resonance structures! It's like molecules having a bit of a personality crisis, but in a totally cool, scientifically significant way. Today, we're going to peek at a particularly fun character: a radical. Now, a radical might sound a bit wild, and in a way, it is! It's like a molecule that's missing a dance partner for one of its electrons. That missing electron makes it a bit jumpy, ready to mingle and find a new friend. But instead of just being jumpy, sometimes these radicals have multiple ways they could be jumpy. And that, my friends, is where the magic of drawing a second resonance structure comes in!
Imagine you've got this one specific radical. It's got a certain arrangement of atoms and a lonely electron hanging out. Now, the rules of chemistry are like a playful game of musical chairs, and electrons love to move around. When we draw a second resonance structure, we're basically showing another possible "snapshot" of where those electrons could be. It's not that the radical is flipping back and forth between these two drawings like a switch. Nope! It's more like the real radical is a super-cool blend, a hybrid of all its possible resonance structures. Think of it like a unicorn: you can draw a unicorn, and you can draw another unicorn with slightly different horn placement, but the actual "idea" of a unicorn is something more magical than either single drawing.
So, for our specific radical, we're going to be drawing another version. It's like giving our little electron-deficient friend a makeover! We're not changing the atoms, oh no. The atoms are the main players, the cast of characters. What we are changing is how the electrons are distributed. This is where things get really interesting and, dare I say, a little bit sneaky. We’re going to be nudging those electrons, encouraging them to find new spots. It’s like rearranging furniture in a room to see how the light hits it differently.
When we draw this second resonance structure, we’re essentially saying, "Hey, what if the electrons wiggled this way instead?" And guess what? The molecule often agrees! It's a testament to the fluidity of electrons. They're not stuck in one place; they can flow and rearrange themselves. This ability to have multiple resonance structures is what makes molecules stable and, in the case of radicals, determines how they'll react. A radical that has more resonance structures is like a social butterfly with lots of friends – it can spread its positive vibes (or in this case, its unpaired electron) around more effectively, making it less reactive on its own.
The fun part about drawing these structures is the detective work. You look at the original radical, you see where that unpaired electron is, and then you start thinking, "Where else could this electron go if it felt like taking a little stroll?" You're looking for opportunities for electrons to move, to form new bonds, or to shift existing ones. It's like a puzzle where the pieces are atoms and the glue is electron movement. And when you find a new arrangement that follows the rules, you've unlocked another piece of the molecule's personality!
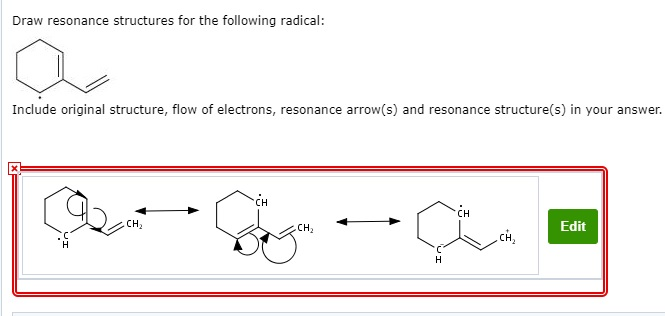
This isn't just about scribbling pretty pictures. These resonance structures tell us a lot about how a molecule behaves. For our radical, the more resonance structures it has, the more it can delocalize that unpaired electron. Think of it like spreading a rumor – if one person knows it, it’s contained. If ten people know it, it’s everywhere! The unpaired electron in a radical is like that rumor. When it can spread out across multiple atoms through resonance, the radical becomes more stable.
"It's like looking at the same thing from different angles, and suddenly, you see a whole new dimension!"
So, as we draw our second resonance structure for this radical, we’re on a little adventure. We’re exploring the hidden possibilities of electron distribution. We’re not changing the underlying substance of the radical, mind you. It’s still the same molecule, with the same atoms. We’re just changing how the electrons are arranged, like dressing up a mannequin in different outfits. Each outfit tells a slightly different story about how the mannequin can be perceived.

And why is this so entertaining? Because it’s a peek behind the curtain of molecular reality! It shows us that things aren't always as simple as they first appear. Molecules are dynamic, fluid, and full of surprises. Drawing resonance structures is like being a molecular choreographer, directing the dance of electrons. For this particular radical, we’re going to see if we can find another elegant step for our unpaired electron. It’s a delightful challenge that requires a bit of visual thinking and a good understanding of electron flow. It’s like a brain teaser with chemical consequences!
What makes this special is that it's not just a static image. It's a representation of a dynamic process. It’s about understanding that the structure we draw is often a simplification. The true nature of the molecule is a blend, a composite of all its possible resonance forms. This is especially true for radicals, where the unpaired electron is itching to find a stable home. By drawing a second resonance structure, we’re giving it more options, more places to potentially settle down, even if just temporarily. It's like offering a friend multiple comfortable chairs to sit in instead of just one.
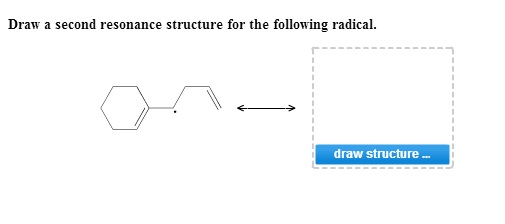
So, get your pencils (or your digital drawing tools) ready! We’re about to embark on a small but mighty quest to uncover another facet of our radical's electron dance. It’s a chance to see the versatility of molecular structures and appreciate the subtle, yet significant, ways electrons can shift and rearrange. It’s a little bit of molecular art, a little bit of detective work, and a whole lot of chemical fun!
When you draw that second structure, you’re not just adding another drawing to your notebook. You’re expanding your understanding of the radical’s electronic landscape. You’re gaining insight into its potential reactivity and stability. It's like having a map with multiple routes to your destination – each route offering a slightly different experience of the journey. This radical, with its potential for multiple resonance structures, is a prime example of this beautiful molecular complexity.
It’s a simple concept, but the implications are huge. Understanding resonance is key to unlocking the behavior of countless molecules. And drawing these structures, especially for a radical, is a fantastic way to start wrapping your head around it. So, as you sketch out that second resonance structure, remember you’re not just following a rule; you’re revealing a deeper truth about the molecule you’re studying. It's a little bit of chemistry magic unfolding right before your eyes!
