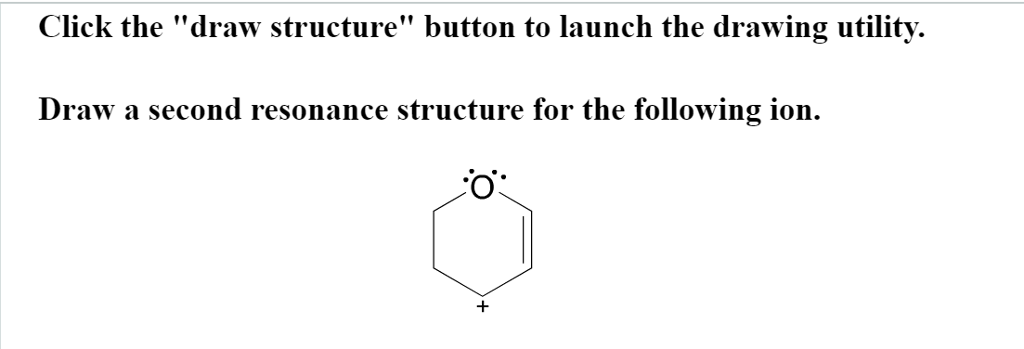
Draw A Second Resonance Structure For The Following Ion
Hey there, fellow wanderers of the digital age! Ever feel like you’re juggling a hundred tabs in your brain, trying to keep everything in focus? We get it. Life’s a vibrant tapestry, and sometimes, even the simplest threads can get a little tangled. Today, we’re diving into a topic that might sound a smidge academic at first glance, but trust us, it’s got a surprisingly chill vibe. We’re talking about resonance structures – specifically, how to draw a second one for a particular ion. Think of it like finding a different angle to appreciate a beautiful piece of art, or perhaps discovering a hidden gem on your favorite streaming service. It’s all about perspective, and how a molecule, much like a good song, can have different, equally valid interpretations.
So, what exactly are these "resonance structures"? Imagine a molecule is like a super-stylish outfit. Sometimes, the way you wear your scarf or the way your jacket drapes can change the whole look, right? Resonance structures are a bit like that. They're different ways of drawing a molecule or ion that show how electrons are spread out. It's not that the molecule is actually switching between these forms; rather, these drawings are just our best attempts to capture the reality of electron delocalization – that's a fancy way of saying electrons are shared more broadly than a single simple drawing can convey. Think of it like a mood ring for electrons – they’re not fixed, they’re a bit fluid, and they like to move around!
Now, you might be wondering, "Why is this important?" Well, understanding resonance helps us predict how molecules will behave. It's like knowing your friend's different moods – sometimes they're energetic and ready to go, other times they need a quiet afternoon with a good book. Knowing these variations helps you interact with them better. In chemistry, understanding resonance helps us predict reactivity, stability, and even color! Yes, some of the vibrant colors in dyes and pigments come from molecules with extensive resonance. So, next time you admire a bold crimson scarf or a brilliant sapphire ring, you can wink at the underlying electron dance.
We’re going to tackle a specific ion today, and it’s a great example for illustrating this concept. Let’s imagine we have an ion that’s like a chameleon, capable of adapting its electron distribution. The trick to drawing a second resonance structure is all about moving electrons. Specifically, we're moving lone pairs (those pairs of electrons hanging out on atoms that aren't involved in bonding) and pi bonds (the second and third bonds between two atoms, which are a bit more flexible than the first, or sigma, bond). Think of it like rearranging furniture in a room. You’re not getting rid of any furniture, you’re just shifting things around to create a different, but still functional, layout. The overall charge of the ion, and the connectivity of the atoms (who is bonded to whom), must remain the same. That’s the golden rule of resonance!
The Art of Electron Shuffle
Alright, let’s get down to the nitty-gritty, but in a way that’s as breezy as a Sunday morning. To draw a second resonance structure, we’re going to focus on three main tools in our electron-moving toolbox: lone pairs, pi bonds, and empty orbitals (though we won't need that last one for every case, it's good to keep in mind for more advanced scenarios). For our ion today, we'll be concentrating on shifting those lone pairs and pi bonds. It’s like learning a few basic dance steps. Once you’ve got them down, you can create a whole routine.
The fundamental principle is this: electrons always move from areas of higher electron density to areas of lower electron density, or to fill an empty space. This movement helps to distribute the charge and stabilize the molecule. Think of it like balancing a budget – you’re spreading your money around to make sure everything’s covered and nothing’s in deficit for too long. In our case, we’re spreading electrons to make sure the charge is distributed and the molecule is happy and stable. A happy molecule is a stable molecule, and who doesn't want to be happy and stable?
Imagine you have a picture of your ion. The first drawing is like the initial snapshot. Now, to get the second drawing, you’re going to take your virtual marker and draw curved arrows. These arrows are crucial. They don't just show movement; they show the direction of electron movement. A curved arrow always starts at the source of the electrons (either a lone pair or a bond) and points to where they are going (either to form a new bond or to become a lone pair).
Let’s consider our ion. We’ll assume it has a certain arrangement of atoms and an initial distribution of electrons. Your task is to identify potential sites where electrons can move. Look for atoms with lone pairs adjacent to double or triple bonds. Also, look for atoms that are double or triple bonded that are adjacent to atoms with lone pairs or negative charges. These are your prime locations for electron movement. It’s like spotting a secret handshake in a crowd – once you know what to look for, it becomes obvious.
Step-by-Step to Your Second Masterpiece
So, let's say our ion has a structure that looks something like this (imagine a simple diagram here – if you’re a visual learner, grab some paper and sketch along!). We have a central atom, let’s call it ‘A’, bonded to a few other atoms, and there’s a charge somewhere. Your first step is to identify all the lone pairs of electrons and all the pi bonds.
Now, let’s play the electron shuffle game. Pick a lone pair that is next to a pi bond. You can draw a curved arrow starting from that lone pair, pointing to the space between the two atoms of the pi bond. This signifies that these two electrons from the lone pair are forming a new pi bond.
What happens next? Well, if you’ve just formed a new pi bond, you’ve increased the electron density between those two atoms. If one of those atoms was already involved in a pi bond (making it a double or triple bond), it might not be able to accommodate any more bonds without violating the octet rule (that tendency for atoms to have eight electrons in their outer shell, like filling a perfect six-pack of soda). So, the existing pi bond needs to break. The electrons from that original pi bond then move to become a lone pair on one of the adjacent atoms. You draw another curved arrow starting from the middle of the original pi bond, pointing to the electron-rich atom that will now hold this new lone pair.

And voilà! You've just drawn a second resonance structure. Double-check your work. Does the new structure have the same atoms connected in the same order? Yes. Does it have the same total charge? Yes. Have you only moved electrons (lone pairs and pi bonds), not atoms? Absolutely. You've successfully navigated the electron currents! It’s like learning a new card trick – once you see how it’s done, it’s surprisingly easy to perform.
Let’s think about it this way. Imagine our ion is a popular influencer. The first structure is one of their iconic poses, perfectly curated. The second structure is them in a more candid, relaxed moment, maybe showing a different facet of their personality. Both are authentically them, just captured from a different angle. Resonance structures do the same for molecules – they show us the different, contributing ways electrons can be distributed, leading to a more complete understanding of the molecule’s true nature.
It’s worth noting that not all resonance structures are created equal. Some contribute more to the overall picture than others. Think of it like casting a movie. You have your lead actors (major contributors) and your supporting cast (minor contributors). The lead actors carry the bulk of the story, but the supporting cast adds depth and richness. In chemistry, we assess resonance structures based on factors like the number of bonds, the placement of formal charges, and the octet rule. Structures that minimize charge separation and maximize octets are generally more stable and contribute more significantly.
This concept of electron delocalization is fascinating because it’s not just an abstract chemical idea. It has real-world implications. Think about the colors of the sunset. The way light interacts with molecules in the atmosphere, often involving extensive resonance structures, is what creates those breathtaking hues. Or consider the resilience of certain materials. Molecules with delocalized electrons are often more stable and less prone to breaking down, making them ideal for everything from advanced plastics to pharmaceutical drugs.
And if you're a foodie, you might find a parallel in the way flavors blend in a complex dish. A great sauce isn't just one ingredient; it's a harmonious interplay of many, each contributing its own notes to create a symphony of taste. Resonance structures are like that – a symphony of electron distribution that creates a stable and reactive molecule.

Beyond the Drawing Board: Why It Matters
So, why should you, a modern individual navigating a world of endless possibilities, care about drawing a second resonance structure? Because it's a fundamental skill that unlocks a deeper understanding of the molecular world around us. It’s like learning to read sheet music. Once you can interpret the notes, you can appreciate the complexity and beauty of a symphony. Without it, you’re just hearing sounds.
This understanding of electron movement is the bedrock for so many advanced topics in chemistry and related fields. Whether you’re interested in medicine, materials science, environmental chemistry, or even just understanding how your favorite shampoo works, resonance plays a role. It helps us explain why some reactions happen easily and others require a lot of energy. It helps us design new molecules with specific properties, like designing a new material that's lighter and stronger than steel, or a new drug that targets a specific disease with minimal side effects.
Think about the evolution of technology. From the first clunky computers to the sleek smartphones in our pockets, it’s all built on a foundation of understanding how matter behaves at its most fundamental level. Resonance is a part of that foundational knowledge. It’s the secret ingredient in many of life’s chemical marvels.
And let’s not forget the sheer satisfaction of solving a puzzle! There’s a certain intellectual joy in taking a complex problem and breaking it down into manageable steps, arriving at a correct and elegant solution. Drawing resonance structures is precisely that. It’s a mental workout that sharpens your analytical skills and your attention to detail. It’s like a mini-game of molecular Tetris, where you’re fitting electrons into the right places.
Furthermore, in our hyper-connected world, being able to communicate complex ideas clearly is a superpower. Understanding and being able to represent resonance structures is a form of visual communication that’s universally understood by scientists. It’s like speaking a common language that transcends borders and disciplines.
So, the next time you encounter an ion and are asked to draw a second resonance structure, don't groan. Embrace it! See it as an opportunity to peek behind the curtain of molecular interactions. It’s a chance to explore the dynamic, fluid nature of electrons and to appreciate the elegance of chemical stability.
It’s also a great reminder that there’s often more to something than meets the eye. Just like a person can have multiple facets to their personality, or a song can have different interpretations, molecules and ions often exist in a state of electron delocalization. Our drawings are just our best efforts to represent this intricate reality. This understanding can foster a more nuanced perspective in our own lives. We can learn to appreciate that situations aren't always black and white, that people have multiple layers, and that there's beauty in complexity.
In our daily lives, we are constantly making choices that affect the balance and stability of our own well-being. Just like chemists strive for stable, delocalized electron structures, we too can aim for a balanced lifestyle, spreading our energy and focus across different aspects of our lives – work, relationships, health, and personal growth. Sometimes, we might need to shift our focus, just like moving those electrons, to achieve a better overall harmony.
So, go forth, draw those second resonance structures, and remember that understanding the subtle dance of electrons can lead to a more profound appreciation of the incredible world around us, and perhaps, even a little more balance in our own. Happy drawing!
