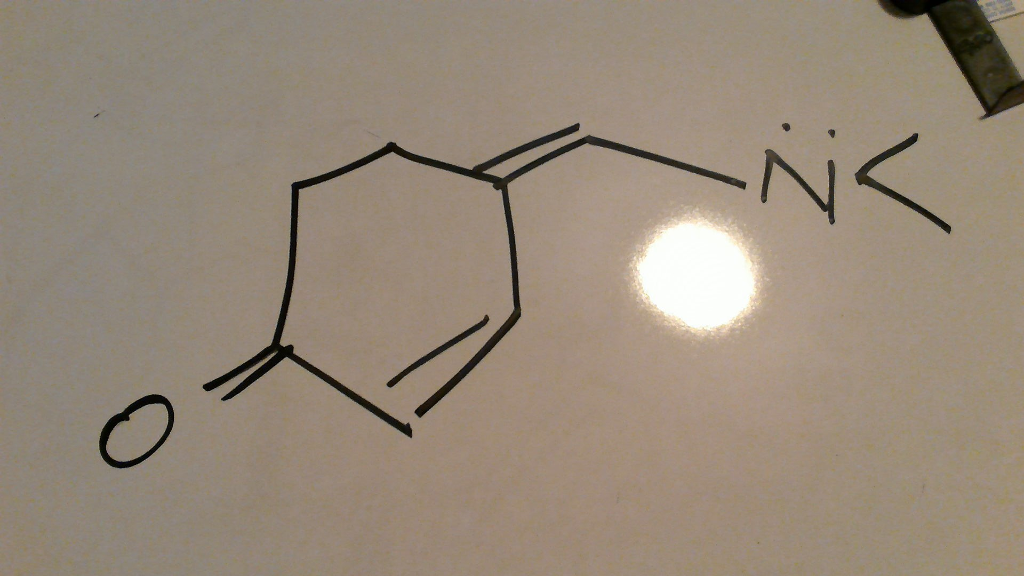
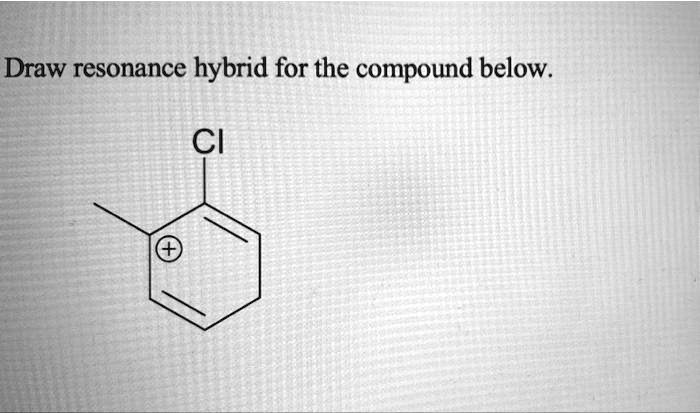
Draw A Resonance Structure For The Compound Below.
Ever feel like your brain needs a little fun workout? Well, get ready for some seriously cool mental gymnastics. We're about to dive into the wacky and wonderful world of drawing resonance structures. It's like giving molecules a little makeover, and trust me, it's way more exciting than it sounds!
Think of it this way: sometimes, a single picture just doesn't tell the whole story. Molecules are a bit like that. They have these hidden talents, these ways of shifting their electrons around that make them more stable and interesting. And that's where resonance structures come in!
Imagine a molecule is like a really popular celebrity. This celebrity has a few different "looks" they can pull off, right? One day they're rocking a casual vibe, the next they're all glam. Our molecules are kind of the same, but instead of clothes, they're playing with their electron clouds.
So, what's the big deal about drawing these structures? It's all about understanding how molecules behave. When we draw resonance structures, we're showing all the possible ways those electrons can be distributed. It's like seeing all the different angles of our celebrity friend, and each angle tells us something unique about them.
This isn't just a boring chemistry lesson, oh no. This is like a puzzle, a visual game where you get to be the artist. You're not just memorizing facts; you're actually seeing the chemistry come alive on the page.
Let's talk about the compound we're going to play with. It's not some super complicated beast, but it's got enough personality to show off its resonance tricks. We're going to focus on how the electrons can move around in it. It's like finding hidden pathways for those tiny, energetic particles.
The key here is to understand that these resonance structures aren't separate molecules. They're like different snapshots of the same molecule. The real molecule is a blend, a sort of average of all these possibilities. It's like saying our celebrity doesn't really look like just one photo, but a combination of all of them.
It’s a bit like when you see a picture of your pet and think, "Yep, that's them!" but then you see another picture from a different angle and it’s also them, but in a slightly different pose. Resonance structures are those different poses for molecules, showing how their electrons can spread out.

The coolest part? When you draw these structures, you start to see patterns. You'll notice how electrons love to move from areas of high concentration to areas where they're needed. It's like a constant dance, a graceful ballet happening at the molecular level.
So, what do we actually do when we "draw a resonance structure"? We use arrows to show the movement of electrons. Think of these arrows as little messengers, telling us where the electrons are going. They’re like magical breadcrumbs leading you through the molecule’s electron landscape.
And here’s a little secret: there are rules! But don't worry, they're not scary rules. They're more like guidelines that help you draw correctly. It’s like following a recipe to bake a delicious cake. You want to make sure you’re adding the right ingredients in the right order.
The most important rule is that the atoms themselves don't move. Only the electrons get to pack their bags and go on a little adventure. The atomic skeleton stays put, like the stage of a theater while the actors move around.
We're not just adding or removing electrons, either. We're shuffling them around within the molecule. It's like rearranging furniture in a room to make it look and feel different, but it's still the same room.
When you draw a resonance structure, you're often looking for things like double bonds or lone pairs of electrons. These are the places where the magic can happen. They're like the starting points for our electron adventure.
![[SOLVED] Drawing resonance structure Draw resonance structures for the](https://dsd5zvtm8ll6.cloudfront.net/si.experts.images/questions/2024/09/66f8f450186a2_50366f8f44f948ad.jpg)
And that special compound we're looking at? It has just the right features to make drawing its resonance structures a real treat. You’ll see how a little shift here and a little shift there can create a whole new way to look at the molecule.
It’s really about understanding the concept of delocalization. That’s a fancy word that basically means the electrons aren’t stuck in just one place. They can spread out, and that spreading makes the molecule more stable. Think of it like a group of friends sharing a big pizza versus each friend having their own tiny slice. Sharing is often better!
The more resonance structures a molecule has, the more stable it generally is. This is why certain molecules are so common and important in nature and in chemistry. They’ve found a way to be super comfy and happy by spreading their electron love.
When you’re drawing, you’ll also be dealing with formal charges. These are like little labels that tell you which atom has a little extra positive or negative feeling. It's like assigning personalities to each atom based on their electron situation.
It’s important to make sure that your resonance structures still have the same overall charge as the original molecule. You can’t just create or destroy charges out of thin air. That would be like trying to get more money by just wishing for it!
The process of drawing resonance structures is a fantastic way to train your brain to think about molecules in a more dynamic way. You move beyond static images and start to appreciate the constant motion and flexibility within chemical bonds.
It’s like learning a new language, but instead of words, you’re learning the grammar of electrons. Once you get the hang of it, you can start to read and understand the hidden messages in chemical structures.
And for this particular compound, you’ll see how its structure lends itself to some really neat electron shuffling. It's not just a random jumble; there's a logic, a flow to it that becomes clear as you draw.
Don’t be discouraged if it feels a little tricky at first. Like any new skill, it takes practice. But the more you draw, the more intuitive it becomes.
Imagine you're a detective, and the molecule is a crime scene. The electrons are the clues, and resonance structures are your way of reconstructing the events, showing all the possible ways the electrons could have moved to get to where they are.
The beauty of it is that it’s a visual representation of something invisible. You’re taking the abstract concept of electron distribution and making it tangible on paper.
This is what makes chemistry so fascinating. It’s about understanding the tiny, unseen world that governs everything around us. And drawing resonance structures is a key skill to unlocking those secrets.
So, the next time you're looking for a brain teaser, a fun challenge that actually teaches you something, give drawing resonance structures a try. Especially for this compound, it’s a great place to start and a truly rewarding experience.
You might just find yourself hooked on the elegance and the logic of it all. It’s a little bit of art, a little bit of puzzle, and a whole lot of cool science all rolled into one.
Why is it so entertaining? Because it’s like unlocking a secret code. You're revealing the hidden dynamics of a molecule. It’s the feeling of “aha!” that makes it so satisfying.
What makes it special? It’s the way it bridges the gap between what we see and what’s actually happening at the atomic level. It's a glimpse into the true nature of chemical bonding.
So go ahead, grab a pen, paper, and maybe your favorite molecule. Let the electron dance begin! You might be surprised at how much fun you have.
