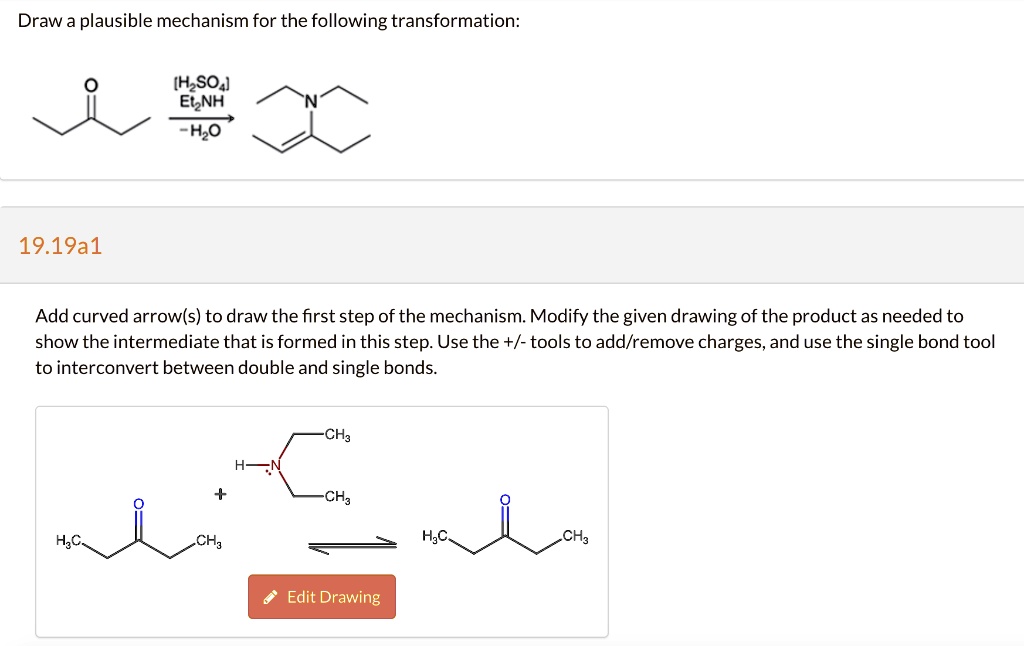
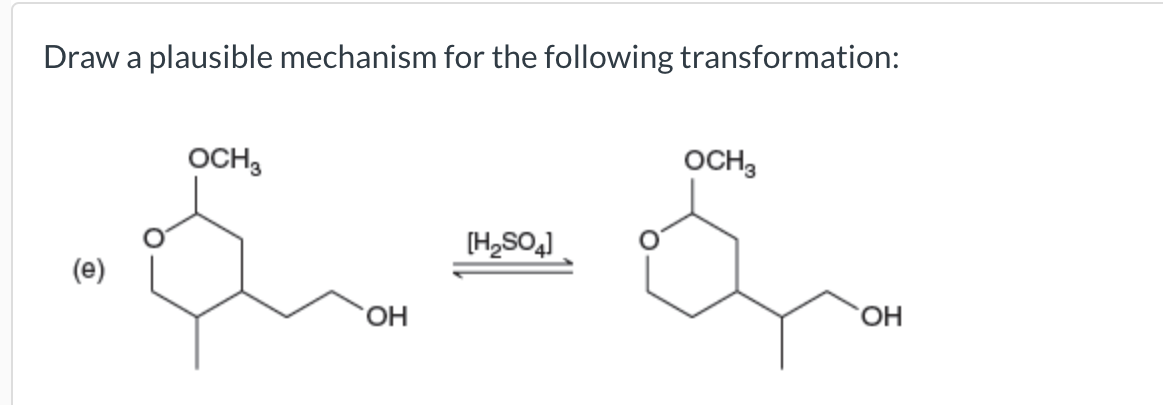
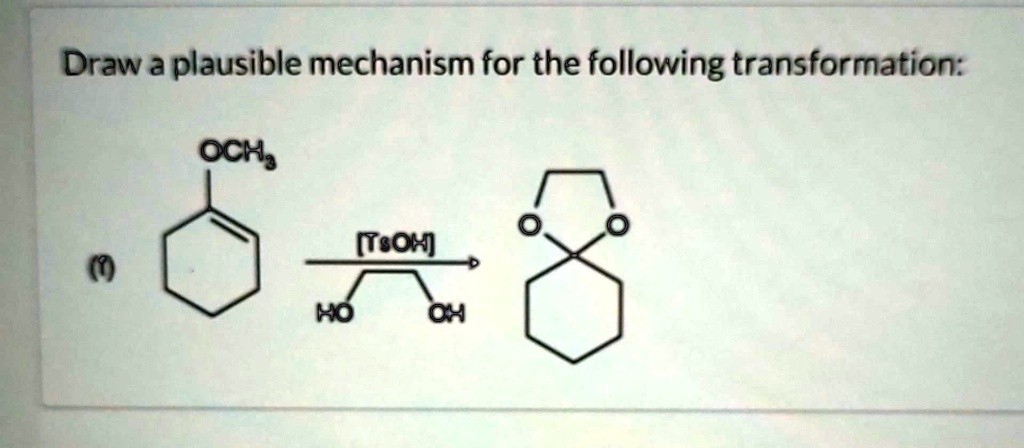
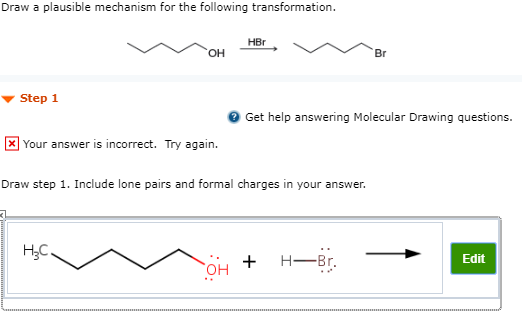
Draw A Plausible Mechanism For The Following Transformation.
Ever looked at a chemical reaction and thought, "Whoa, how did that happen?" Sometimes, the way molecules do their magic can feel like a secret handshake. It's all about drawing out the story, step by step.
Think of it like a detective case for atoms. You've got your starting players, the reactants, and your finished goods, the products. Your job is to figure out the exact sequence of events that got us from A to B.
This isn't just scribbling on a whiteboard. It’s about understanding the hidden forces at play. It's like predicting the moves in a super-fast, microscopic chess match.
So, what’s the big deal? Why should you care about drawing out a plausible mechanism? It’s surprisingly fun! It’s like unlocking a puzzle where the pieces are tiny, energetic particles.
Imagine you're given a really cool transformation. Let's say we’re turning one type of sugary treat into another, but with a few extra sprinkles. That's the kind of challenge that sparks joy.
The beauty of it is that it’s not just random. There are rules! These rules are based on how electrons behave, and boy, do they love to move around.
When you’re drawing a mechanism, you're essentially showing where those electrons are going. You're drawing little arrows that show the dance of the electron cloud. It's a visual narrative of molecular choreography.
It’s like saying, "Okay, this atom is feeling a bit lonely for electrons, so it's going to steal some from its neighbor." And poof, a new bond forms!
What makes it so entertaining? It’s the thrill of prediction. You look at the starting materials and the end goal, and you start to brainstorm the possibilities.
You might think, "Could this atom push its electron this way?" Or, "Maybe this group needs to grab a proton first?" Each question is a potential clue.
And the best part? Often, there's more than one way to get there! It’s like having multiple routes on a map. Some are more direct, some are more scenic, but they all lead to the same destination.
This is where the "plausible" part comes in. A plausible mechanism is one that makes chemical sense. It follows the established principles of how molecules interact.
We don't just invent steps out of thin air. We lean on established theories, like the idea that electrons like to go from areas of high electron density to areas of low electron density. It’s like a molecular game of "follow the leader."
So, when you're presented with a transformation, say, something like turning a plain old alcohol into a super-reactive halide, you start to think. What makes alcohols tick? What makes halides so… well, reactive?
You might recall that the hydroxyl group (that’s the -OH bit) on an alcohol isn’t the best leaving group. It’s a bit clingy. For the reaction to happen, we need to make it easier for it to depart.
This is where clever chemistry comes in. Perhaps we can add an acid, like HBr. That acid has a hydrogen ion, a lone proton just waiting to find a home.
The oxygen atom in the hydroxyl group has some lone pairs of electrons, remember? These are like little invitations for positive charges. So, the oxygen kindly offers up a pair to that hydrogen.
Now, the oxygen has a positive charge. This makes the whole -OH group a much better traveler, a better "leaving group." It’s like giving it a ticket to leave the molecule.
Simultaneously, the bromide ion (that's the Br⁻) is hanging around. It’s got a negative charge and is looking for opportunities.
The carbon atom that was attached to the original hydroxyl group is now feeling a bit exposed. With the positively charged oxygen pulling away, it’s becoming more susceptible to attack.
Enter the bromide ion! It sees this opportunity and swoops in. Its negatively charged electrons are attracted to the slightly positive carbon.
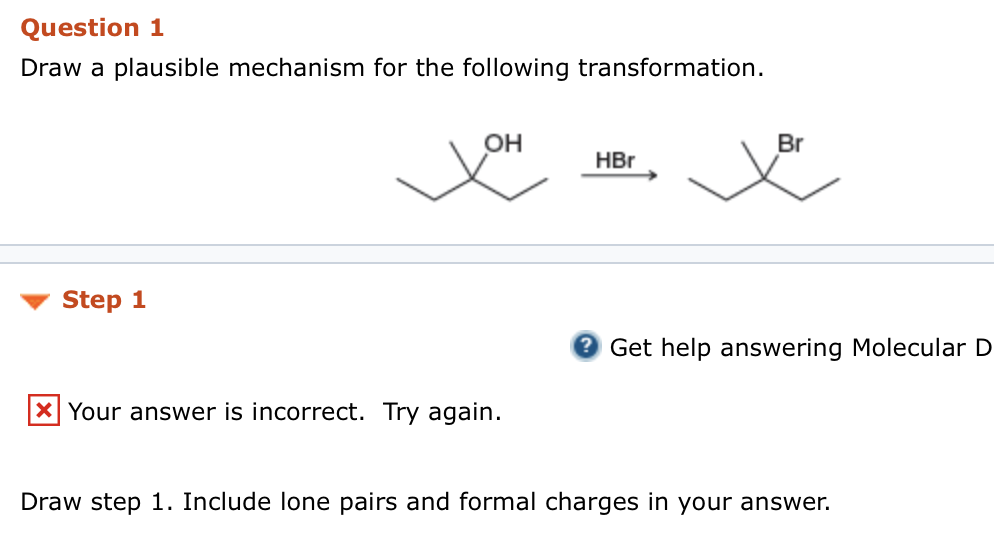
This is a classic move, often seen in reactions called SN2 reactions. It's a concerted process, meaning it happens all in one go. The bromide attacks the carbon at the same time as the water molecule (what the -OH group becomes after protonation) leaves.
It's like a smooth handoff. The bromide comes in from one side, and the water molecule scoots out from the other. This particular mechanism is called inversion of configuration, because the groups around the carbon flip like a pancake.
What makes this so special is the elegance of it. There are no complicated intermediaries that hang around for ages. It's a swift, efficient dance of electrons and atoms.
The ability to visualize these unseen events is incredibly powerful. It’s what allows chemists to design new reactions, to create new materials, and to understand the fundamental processes of life.
It’s also incredibly satisfying when you’ve drawn out the steps and it all makes perfect sense. You feel like you’ve cracked the code. You’ve understood the secret language of molecules.
Sometimes, the reaction might proceed through different pathways. For example, if we were dealing with a tertiary alcohol, the mechanism might be different. It might go through a positively charged intermediate called a carbocation.
In that case, the hydroxyl group would leave first, forming the carbocation. This intermediate is like a temporary, but very reactive, parking spot for a positive charge.
Then, the bromide ion would attack this carbocation. This is characteristic of an SN1 reaction. It's a two-step process where the leaving group departs before the nucleophile attacks.
The beauty of drawing these mechanisms is exploring these nuances. It’s understanding why one alcohol might react differently than another. It’s appreciating the subtle electronic effects that guide molecular behavior.
It’s like being a film director, but for molecules. You're deciding the sequence of shots, the timing of the actions, and the motivations of your microscopic actors.
And when you get it right, when your proposed mechanism perfectly explains the observed transformation, there’s a genuine sense of accomplishment. It’s like solving a really tough riddle.
So, next time you see a chemical transformation, don’t just see the end result. Imagine the journey. Try to draw out the plausible mechanism. You might be surprised by how much fun you have unlocking the secrets of the molecular world.
It's a journey of curiosity and deduction, a testament to the elegant logic that governs the universe at its smallest scales. And that, my friends, is pretty darn entertaining.
