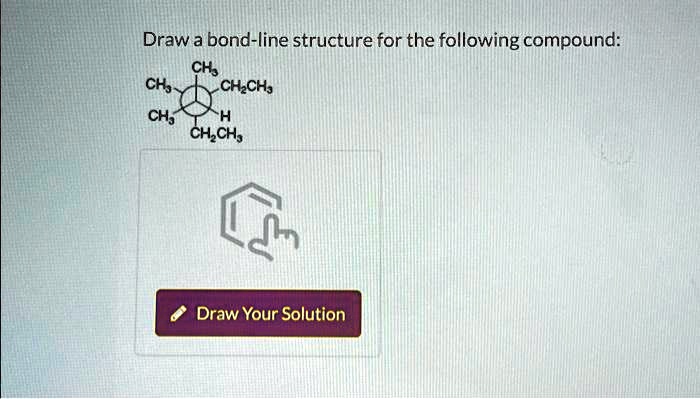
Draw A Bond Line Structure For The Following Compound
Okay, so you’ve probably seen those weird drawings in chemistry class, right? The ones that look like a secret code from a spy movie. They’re called bond-line structures. And sometimes, just sometimes, looking at them feels like trying to solve a really, really complicated puzzle. You know, the kind where you’re missing half the pieces and the picture on the box is smudged.
Today, we’re diving into one of these little mysteries. It’s a game of sorts, a visual scavenger hunt. You're given a name, and you have to draw this special kind of picture. It's like a molecular fingerprint, but way more zig-zaggy.
So, the compound we’re tackling is… drumroll please… 2-methylbut-2-ene. Sounds fancy, doesn't it? Like a secret agent’s code name or a very specific type of car exhaust. But hey, it’s just a molecule doing its thing.
Now, before we get our drawing hands dirty, let’s acknowledge the elephant in the chemistry lab. For many of us, chemistry was a subject that sometimes felt like trying to understand a foreign language spoken backwards. Right? We’d stare at these diagrams, and our brains would go, "Is this supposed to look like a pretzel? Or a really abstract piece of art?"
My unpopular opinion? These bond-line structures, while super useful for chemists, can sometimes feel like they were designed by someone who really loved geometry class and hated people actually understanding things at first glance. Like, "Oh, you want to see the atoms? Nah, let's just draw some lines and pretend you know what's at the corners."
But here’s the fun part, and I’m genuinely serious about this: once you get the hang of it, it’s actually kind of cool. It’s like unlocking a cheat code. You look at the name, and then poof, the structure appears in your mind’s eye. Or, you know, on your paper. After a bit of head-scratching, of course.
Let’s Break Down 2-methylbut-2-ene, Shall We?
The name itself is a set of instructions. It’s like a recipe, but for building molecules. First, we see the “but” part. This tells us the backbone of our structure. Think of it as the main road, the longest chain of carbon atoms we’re going to have. And “but” means four. So, we’re starting with a chain of four carbon atoms. Easy peasy, right? Well, as easy as anything in chemistry can be without a calculator and a strong cup of coffee.
Next, we have the “-ene” ending. This is a big clue. It means there’s a double bond somewhere in that four-carbon chain. Double bonds are like the exciting shortcuts or detours on our molecular road. They make things a bit more reactive and interesting. Think of them as the spicy salsa in the otherwise plain burrito of the carbon chain.
Now for the “2-ene” part. This tells us where that double bond is. It’s connecting the second and third carbon atoms in our four-carbon chain. So, it’s not at the very beginning or the very end, but smack-dab in the middle. Like the most important conversation happening in the middle of a party. Everyone’s paying attention.
But wait, there’s more! We also have “2-methyl.” This is like an extra topping for our molecular pizza. A “methyl” group is simply a carbon atom with three hydrogens attached to it. It’s a single carbon atom, looking for a place to party. And this one is attached to the second carbon atom in our main four-carbon chain.
So, to recap: a four-carbon chain, a double bond between carbons two and three, and a little methyl group hanging out on carbon two. It’s starting to sound like a tiny molecular traffic jam, isn’t it?
The Zig-Zag Dance of Drawing
Now, the fun part: the drawing. Forget drawing little circles for atoms. That’s for beginners. In bond-line structures, each corner and the end of each line represents a carbon atom. And all the hydrogen atoms are just… implied. They’re there, doing their hydrogen thing, but we don’t usually draw them unless they’re attached to something unusual.
So, for our four-carbon chain, we’ll start by drawing a zig-zag line with four points. Imagine drawing a really lazy ‘W’ or a very happy, slightly squashed ‘M’. Each point where the line bends, and the very ends, are your carbon atoms. Let’s number them in our heads, from left to right, 1 to 4.
Now, remember that double bond? It’s between carbon 2 and carbon 3. So, between the second and third points of our zig-zag, we draw a second line. That’s our double bond. So, that section of the zig-zag will look like two parallel lines instead of one.
And what about that “2-methyl” group? It’s a single carbon atom (which we represent with a line) attached to carbon number 2. So, from the second point of our main zig-zag, we draw another little line sticking out. This little line is our methyl group. And since it’s a single carbon, it’s just a little line ending in a point.
It’s like decorating a molecule. You’ve got your main structure, then you add a special feature, and then a little flourish on the side.
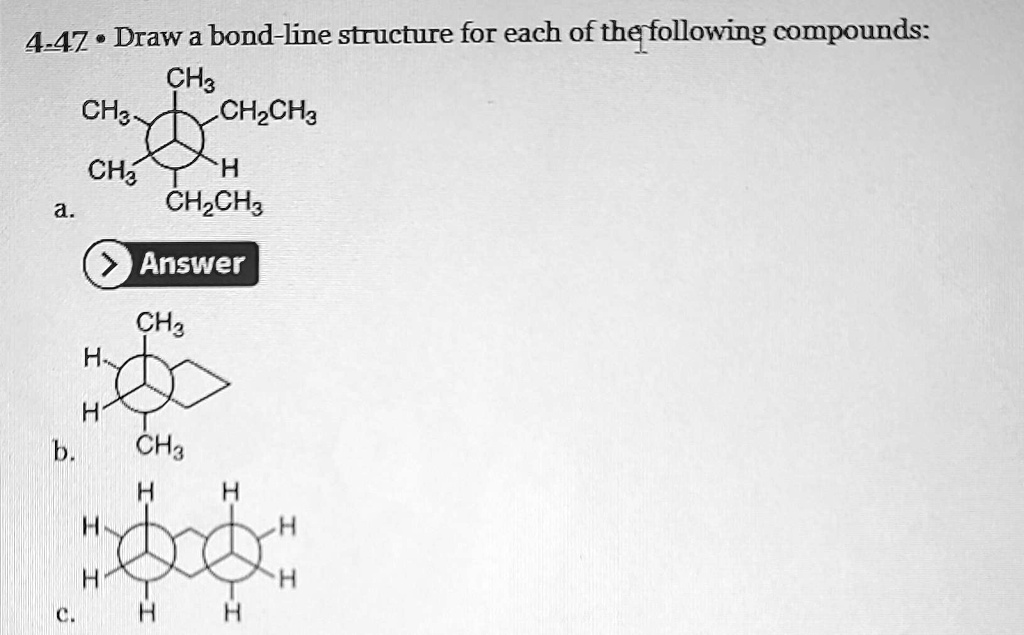
4-47. Draw a bond-line structure for each of the following compounds: a
The beauty, and sometimes the terror, of bond-line structures is their simplicity. They strip away the clutter. You don't see all the H's clinging on for dear life. You just see the carbon skeleton and the important functional groups, like our double bond and our methyl group. It’s the molecular equivalent of a minimalist apartment – very chic, but you have to know what everything is without explicit labels.
So, if you were to draw 2-methylbut-2-ene, you'd have a zig-zag line for the four-carbon chain. The second and third carbons would be connected by two lines. And from the second carbon, a single line would extend outwards, representing the methyl group. Each point and end of a line is a carbon atom, and the hydrogens? They're just assumed to be there, filling up the remaining spots to make each carbon happy with four bonds.
It’s a bit like a secret handshake. Once you know the moves, you can interpret them. And honestly, there’s a certain satisfaction in deciphering these molecular hieroglyphics. It’s like saying, "Aha! I see you, little 2-methylbut-2-ene! You're just a fancy four-carbon chain with a double bond and a side hustle!"
So next time you see one of these zig-zaggy drawings, don't be intimidated. Think of it as a puzzle waiting to be solved, a molecular riddle. And remember, sometimes the simplest drawings hide the most interesting stories. Or at least, the most interesting arrangements of atoms that keep the universe running. And that, my friends, is pretty darn entertaining if you ask me.