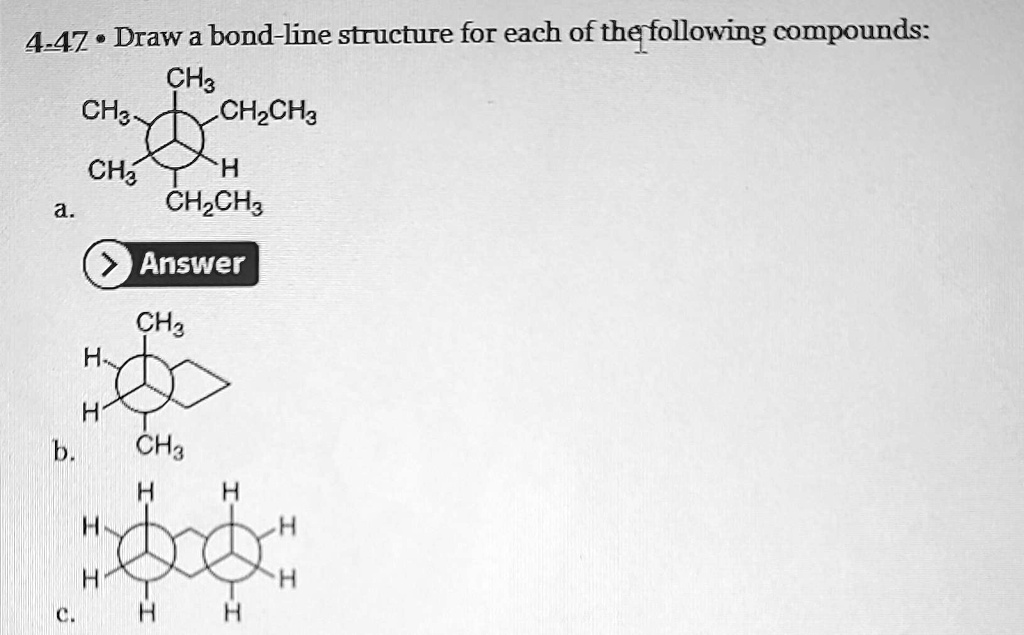
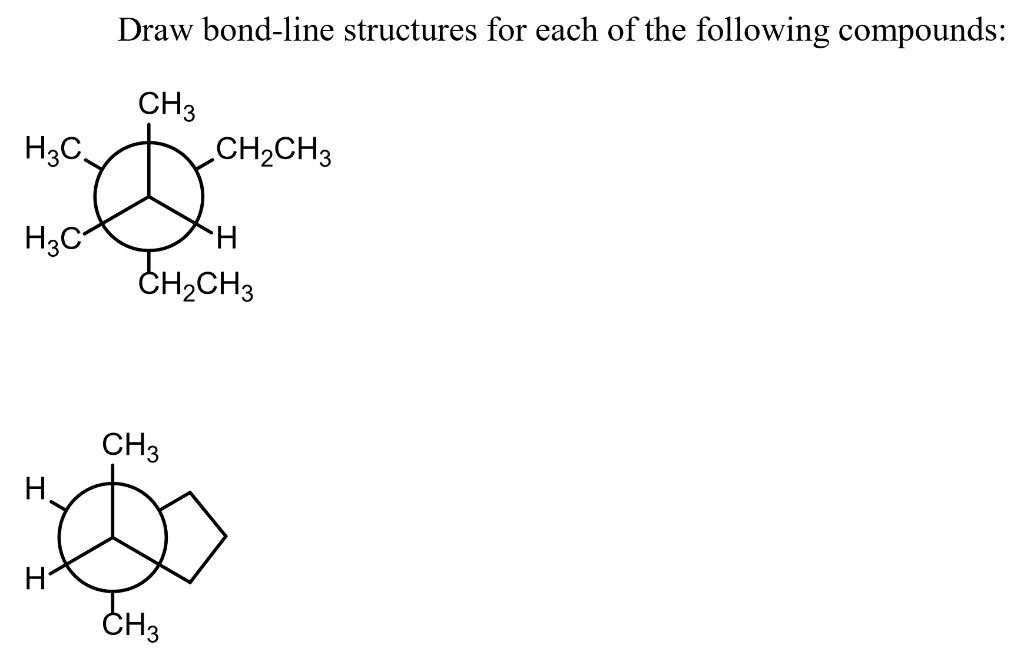
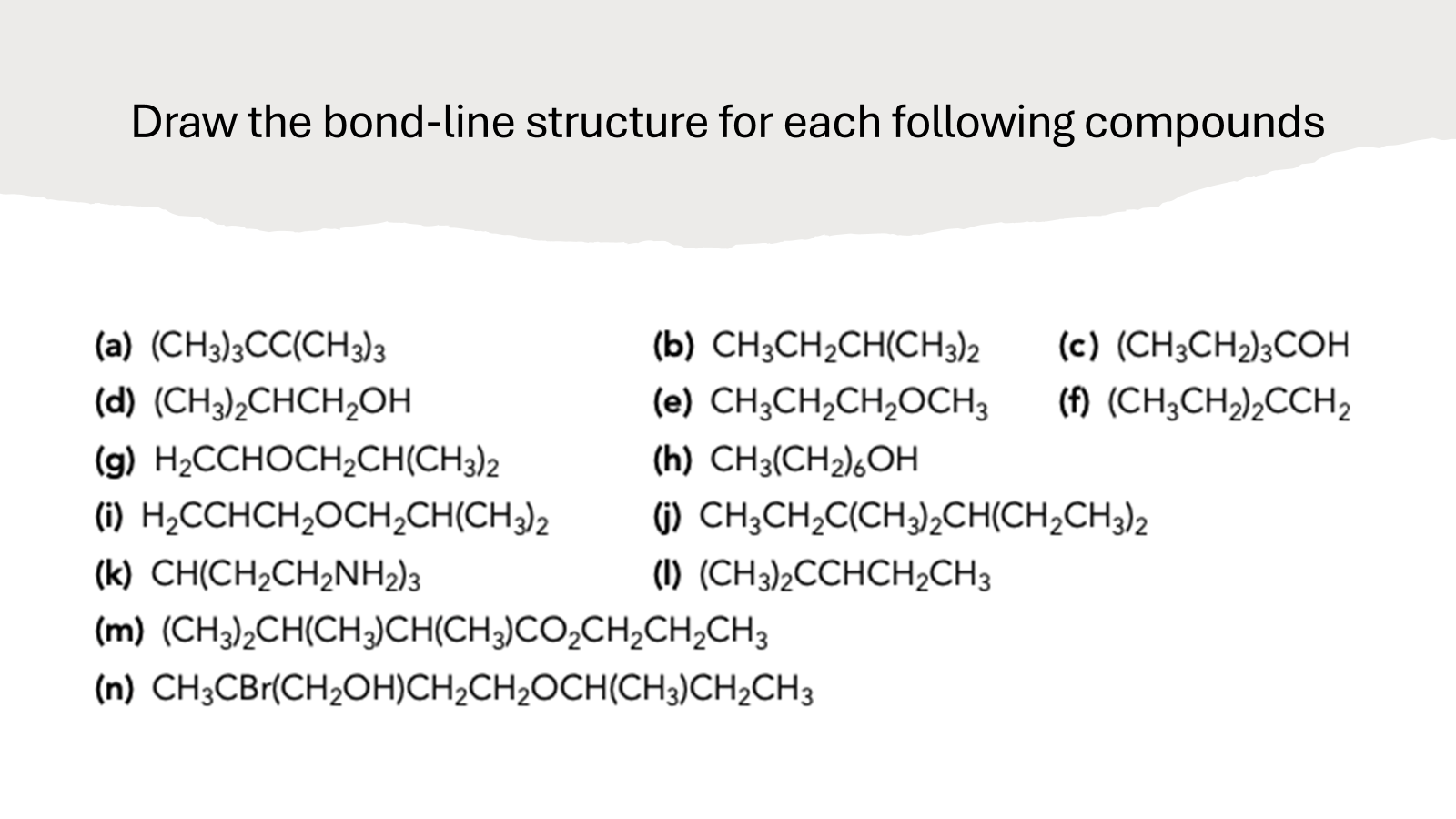
Draw A Bond-line Structure For Each Of The Following Compounds
You know, I was recently rummaging through my grandmother's attic – a place that, let me tell you, is a treasure trove of forgotten wonders and questionable fashion choices from the 70s. Amongst moth-eaten cardigans and stacks of National Geographic magazines featuring more puffins than I ever thought existed, I stumbled upon a box of her old art supplies. There were dried-out paint tubes, charcoal sticks that crumbled at the slightest touch, and then, tucked away in a velvet pouch, a set of beautiful, old-school pencils. They felt substantial in my hand, not like those flimsy ones they give you at dentists' offices. It got me thinking about drawing, about representing things, and how sometimes, the simplest way to show something is to get rid of all the unnecessary bits.
This got me thinking about chemistry, specifically how we draw molecules. We’ve all seen those super detailed, almost overwhelming diagrams with every single atom and every single bond meticulously drawn out. It's like trying to describe a person by listing every single hair on their head and every single freckle. You could do it, but is it the most efficient way? Probably not, right? Especially when you’re dealing with, say, a molecule as big and complicated as, I don't know, a protein. Imagine trying to draw every single bond in a protein! My hand would cramp just thinking about it. Fortunately, chemists, being the clever bunch they are, have figured out a much cooler, much more streamlined way to do it. And that, my friends, is where the wonderful world of bond-line structures comes in.
So, What Exactly Are Bond-Line Structures?
Think of it like this: instead of drawing every single atom, we make some educated guesses. Or, more accurately, we follow some very well-established conventions. A bond-line structure, also known as a skeletal structure or line-angle formula, is a way of representing organic molecules where carbon atoms are represented by the vertices (the corners) and ends of lines, and hydrogen atoms bonded to carbon are generally not shown. You’re probably thinking, “Wait, what? We just… omit things?” Yes, you do! It sounds a bit radical at first, doesn't it? Like throwing out half the ingredients in a recipe. But trust me, it works, and it’s incredibly useful.
The beauty of these structures lies in their simplicity and clarity. They allow us to focus on the functional groups and the overall shape of the molecule, which are often the most important aspects when discussing reactivity and properties. It's like looking at a silhouette of a person instead of a detailed portrait. You still recognize them, but you get the overall form instantly. No need to count every eyelash, you know?
Here’s the golden rule: Each line segment represents a chemical bond. And where two lines meet, or where a line ends, that’s where a carbon atom usually is. Pretty neat, right? You’ve probably seen them before, maybe even in textbooks or online, and thought, “Huh, that looks… minimal.” Well, that minimalism is its superpower!
Let's Break Down the Rules (Don't Worry, They're Not Scary)
Alright, so before we dive into drawing some examples (which is the fun part!), let's get a handle on the basic rules. Think of these as the unspoken agreements among chemists that make these drawings universally understandable. It’s like learning the handshake of your favorite club – once you know it, you're in!
- Carbon is King (and Invisible): As I mentioned, carbon atoms are implied at every vertex and at the end of every line. You don’t draw a 'C'. The only time you would draw a 'C' is if it's bonded to something other than carbon or hydrogen – but we’ll get to that.
- Hydrogen is Shy: Hydrogen atoms directly attached to carbon atoms are also implied. The number of hydrogens attached to a carbon is determined by assuming that each carbon forms a total of four bonds. So, if a carbon vertex is connected to two other atoms (say, two other carbons via lines), it’s assumed to have two hydrogens attached (4 total bonds - 2 bonds = 2 hydrogens). If a carbon is at the end of a line and connected to only one other atom, it’s assumed to have three hydrogens attached (4 total bonds - 1 bond = 3 hydrogens).
- Heteroatoms (The Non-Carbon, Non-Hydrogen Stars): Atoms other than carbon and hydrogen – like oxygen (O), nitrogen (N), chlorine (Cl), etc. – must be explicitly drawn. And any hydrogen atoms attached to these heteroatoms also need to be shown. This is where we see explicit Hs, like in -OH groups.
- Double and Triple Bonds Matter! Single bonds are just single lines. But double bonds are shown as two parallel lines, and triple bonds are shown as three parallel lines. This is super important for understanding the molecule's geometry and reactivity.
- Stereochemistry (The Fancy Footwork): For molecules with chiral centers (carbons bonded to four different groups), we can use wedges and dashed lines to show the 3D arrangement of atoms. Wedges sticking out of the page, dashed lines going behind. We’re not going to get too deep into this for this particular set of examples, but it’s good to know it’s there!
See? Not so bad, right? It’s like learning a secret code that unlocks a whole new way of seeing molecules. Now, let’s put this into practice!
Let's Draw Some! (Prepare for Maximum Coolness)
Alright, time to get our hands dirty – or rather, our pencils (virtually speaking, of course). We’re going to tackle a few compounds and translate them into these sleek bond-line structures. Don’t be intimidated; just follow the rules we just discussed. It’s like a guided meditation for your inner chemist.
1. Propane
First up, a classic: propane. This is a simple alkane, meaning it only contains carbon and hydrogen, and all the bonds are single bonds. Let’s think about its molecular formula: C₃H₈. So, we have three carbon atoms and eight hydrogen atoms.
If we were to draw it the old-fashioned way, it would look like: H₃C-CH₂-CH₃. Three carbons in a row, with hydrogens attached.
Now, for the bond-line version:
- We need three carbon atoms connected in a chain.
- So, we draw a line. That’s our first carbon at the end and our second carbon at the other end of the line.
- Then, we draw another line from the second carbon. This line represents the bond to the third carbon.
- So, we have two connected line segments. The ends and the point where they connect are our three carbon atoms.
- No hydrogens are shown because they are implied to complete the tetravalent nature of each carbon. The end carbons have one bond to another carbon, so they’re assumed to have 3 hydrogens each (3+1=4). The middle carbon has two bonds to other carbons, so it’s assumed to have 2 hydrogens (2+2=4).
Your structure will look like a zigzag with two angles. Simple, clean, and instantly recognizable as a three-carbon chain. Boom! Propane, conquered.
2. Butane
Let’s step it up a notch. Next is butane. Molecular formula: C₄H₁₀. Again, all single bonds. We can have a straight chain or a branched structure. For this exercise, let’s assume the straight-chain isomer, often called n-butane.
The full structure is: H₃C-CH₂-CH₂-CH₃.
For the bond-line structure:
- We need four carbon atoms in a row.
- So, we draw a line (carbon 1 to carbon 2).
- Then, another line from carbon 2 (carbon 2 to carbon 3).
- And a third line from carbon 3 (carbon 3 to carbon 4).
- This gives us a zigzag with three connected line segments. The two ends and the two interior vertices represent the four carbon atoms.
- Again, all hydrogens are implied. Each end carbon has 3 implied Hs, and each interior carbon has 2 implied Hs.
Your structure will be a slightly longer zigzag than propane. It’s like adding another link to the chain. You might also encounter isobutane, which is a branched isomer. In isobutane, one of the carbons is bonded to three other carbons. How would that look? You'd have a central carbon with three lines extending from it, each representing a carbon atom. The end of each of those lines would be another carbon atom. It looks a bit like a Y or a tripod. See how the connectivity is immediately obvious?
3. 2-Methylpropane (Isobutane)
Let’s draw that isobutane we just talked about. The name tells us a lot: “propane” means a three-carbon chain, and “2-methyl” means there’s a methyl group (CH₃) attached to the second carbon of that propane chain.

So, we start with our propane chain (three carbons in a row). Let's draw that zigzag with two corners.
Now, we need to attach a methyl group to the middle carbon. A methyl group is just a CH₃. In bond-line structures, CH₃ is a single line extending from a carbon. So, from the middle vertex of our propane zigzag, we draw a single line sticking up (or down, the direction doesn't fundamentally change the molecule's connectivity). That line represents the carbon atom of the methyl group, and it’s implied to have its three hydrogens.
The structure will look like a Y. The stem of the Y is the three-carbon chain, and one of the arms is the methyl group. The central vertex is the carbon bonded to three other carbons. The two ends of the Y-arms and the end of the stem are the carbons bonded to three hydrogens. This is why these structures are so powerful – they show the branching pattern at a glance!
4. Pentanol
Okay, now we’re getting a bit more interesting because we’re introducing a heteroatom: oxygen, and a hydrogen attached to it. Pentanol means a five-carbon chain (pent-) with an alcohol functional group (-ol), which is a hydroxyl group (-OH). There are different positional isomers of pentanol, but let's draw 1-pentanol, where the -OH group is on the first carbon.
Molecular formula for 1-pentanol: C₅H₁₂O.
The full structure of 1-pentanol would be: CH₃-CH₂-CH₂-CH₂-CH₂-OH.
Let's draw it as a bond-line structure:
- We need a five-carbon chain. So, draw a zigzag with four connected line segments. This gives us five carbons at the ends and vertices.
- Now, the crucial part: the -OH group. Remember, heteroatoms must be shown. So, from one of the end carbons, we draw a line connecting to an 'O'.
- This 'O' is a vertex in our drawing.
- The 'O' is bonded to an 'H', so we must draw that 'H' explicitly attached to the 'O'.
Your structure will look like a five-segment zigzag with a line extending from one end that terminates in an 'O' with an 'H' attached. You can draw the -OH group sticking up, down, or even extending outwards from the end of the chain – as long as it’s clearly attached to the end carbon. This is where the structure starts to look less like just carbons and more like a molecule with distinct parts.
5. Propanone (Acetone)
Let’s try a ketone. Propanone, commonly known as acetone. This has a carbonyl group (C=O) in the middle of a three-carbon chain. The formula is C₃H₆O.
The full structure is: CH₃-C(=O)-CH₃. Notice the double bond between the central carbon and the oxygen.
For the bond-line structure:
- We need a three-carbon chain. Draw a zigzag with two connected line segments.
- The central carbon is the one involved in the carbonyl group. So, from the middle vertex, we draw a line to an 'O'.
- This bond to the oxygen is a double bond. So, we need to draw two parallel lines between the central carbon vertex and the 'O'.
- The oxygens are not implied, and neither are the hydrogens attached to them (if any). In this case, the oxygens don't have hydrogens attached.
Your structure will look like the propane zigzag, but the middle vertex will have two lines extending upwards (or downwards) to an 'O'. This visually screams “ketone!” It’s so much quicker than writing out all the Hs and Cs. It’s like you’ve just distilled the essence of the molecule.
6. 1-Bromopropane
Last one for this set! Let’s do an alkyl halide. 1-Bromopropane. A three-carbon chain with a bromine atom attached to the first carbon.
Molecular formula: C₃H₇Br.
Full structure: CH₃-CH₂-CH₂-Br.
Bond-line structure:
- Start with your three-carbon chain zigzag (two line segments).
- Attach the bromine atom. Bromine is a heteroatom, so we write 'Br' explicitly.
- Draw a line from one of the end carbons to the 'Br'.
This will be the familiar propane zigzag with a 'Br' attached to one of the end carbons. Easy peasy! It’s like adding a little flag to the end of your molecular ribbon.
Why Bother With All This Simplification?
Honestly, when I first learned this, I remember thinking, “Why can’t we just draw it all out?” But then I started working with more complex molecules, and let me tell you, the full Lewis structures would become incredibly cluttered. Bond-line structures are the elegant shorthand of organic chemistry. They let you see the carbon skeleton, the functional groups, and the branching patterns of a molecule at a single glance.
This is crucial for understanding things like:
- Reactivity: Where are the electron-rich or electron-poor areas? What functional groups are present that can participate in reactions?
- Isomerism: How can the same atoms be arranged differently in space to form different molecules?
- Molecular Shape: While bond-line structures don't perfectly represent 3D shape, they give you a good starting point and are often drawn to reflect the basic geometry.
Think of it like learning to read musical notation. You could try to write out a symphony by describing every single note, every single instrument, every single dynamic. Or, you can learn to read the notes on a staff, and suddenly, complex music becomes accessible. Bond-line structures are the musical staff of organic chemistry.
So, the next time you see one of these minimalist drawings, don't dismiss it as incomplete. Appreciate it for its efficiency and clarity. It’s a testament to the ingenuity of chemists in finding the most effective ways to represent complex ideas. It’s a language, really. And once you learn its grammar, you unlock a whole new level of understanding. Happy drawing!
