Does Lead Melt Into Individual Atoms Or Molecules

Ever stared at a dripping faucet and wondered what’s really going on at the tiniest level? Or maybe you’ve seen a metal sculpture and marveled at its solid form. We interact with materials all the time, and understanding how they behave when things get a bit… warmer… is pretty fascinating. Think about it: how do things go from solid to liquid? It’s like a magic trick of nature! And when we talk about metals, especially common ones like lead, the process of melting is a classic example of this transformation. It’s not just about making something gooey; it’s about understanding the fundamental building blocks of matter and how they interact. This curiosity about the unseen world of atoms and molecules is what makes science so cool, and exploring what happens to lead when it melts is a perfect gateway into that world.
The Great Lead Melt: Atoms or Molecules?
So, when we talk about lead, and we heat it up until it turns from a solid chunk into a shiny, flowing liquid, what’s actually happening? Are we breaking it down into its tiniest, individual pieces, like lonely little atoms? Or are these atoms somehow sticking together in small groups, forming tiny molecules that then flow around? This is where things get really interesting, and thankfully, the answer is pretty straightforward once you break it down.
The key to understanding this lies in the nature of lead itself. Lead is a metal. And metals, when they’re pure, are made up of only one type of atom. Think of it like a big LEGO set where all the bricks are exactly the same shape and color. In the case of lead, these identical building blocks are lead atoms. These atoms are held together in a rigid, organized structure when lead is solid. They vibrate in place, but they don’t generally move around and switch places freely.
Now, what happens when we apply heat? Heat is essentially energy, and when we add energy to a substance, its particles start to move more vigorously. For lead, this added energy causes the lead atoms to vibrate even more intensely. Eventually, they gain enough energy to overcome the forces that are holding them in their fixed positions in the solid structure. They start to break free from their rigid arrangement and begin to slide past each other. This is what we observe as melting. The lead has transitioned from a solid state to a liquid state.
So, when lead melts, it’s the individual lead atoms that are gaining enough energy to move around. They aren’t forming new combinations or grouping up into molecules. The lead atoms remain lead atoms. They’re just no longer neatly arranged in a solid lattice. Instead, they are free to move independently of each other, bumping and jostling within the liquid, which allows the lead to flow.
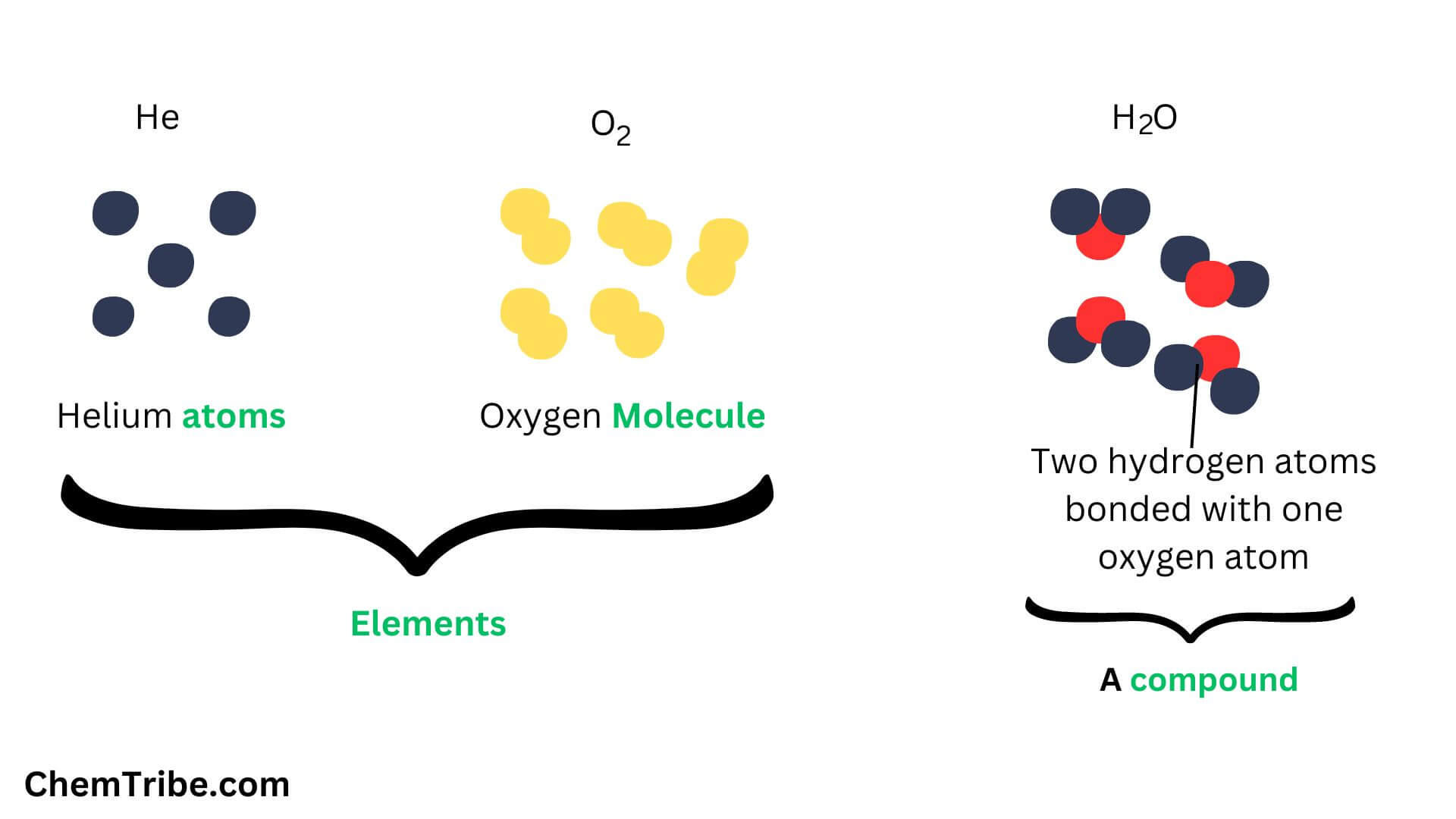
Atoms, Elements, Molecules, and Compounds - ChemTribe
This is a crucial distinction. In some substances, like water (which is made of H₂O molecules), melting involves breaking bonds between these molecules, allowing the molecules themselves to move more freely. But in pure elements like lead, which are made of only one type of atom, melting is about the atoms themselves becoming mobile. The bonds that hold the atoms together in a metallic structure are broken, but the atoms themselves don't combine to form different entities.
The benefits of understanding this are far-reaching. For scientists and engineers, knowing whether a substance melts into individual atoms or molecules is fundamental to designing processes involving heating, cooling, or chemical reactions. For example, in metallurgy, understanding the atomic behavior of metals during melting is crucial for casting, welding, and creating alloys. It helps predict how the metal will flow, how it will solidify, and what its final properties will be.

For the curious mind, it’s simply about appreciating the intricate dance of matter. It’s about realizing that even something as familiar as melting lead is a complex and elegant process governed by fundamental scientific principles. The next time you see something melt, whether it's ice, chocolate, or even a piece of metal (from a safe distance, of course!), you can impress yourself and maybe others with the knowledge that you’re witnessing a fascinating display of atomic freedom.
In short, when lead melts, it's the individual lead atoms that are set free to roam. They don't team up to form molecules. They just become a lot more mobile, and that's what makes liquid lead so… well, liquid!