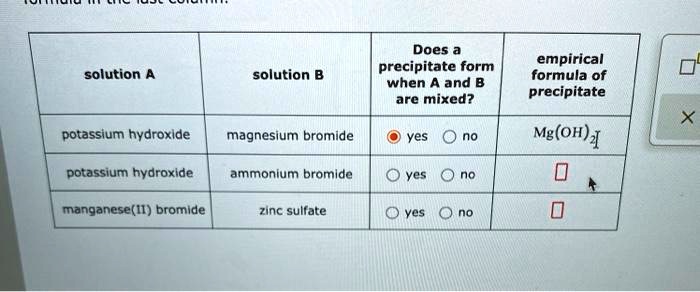
Does A Precipitate Form When A And B Are Mixed

Alright, settle in, grab your latte, and let's chat about something that sounds super serious but is actually, dare I say it, a little bit dramatic. We're talking about what happens when you throw two things together, let's call them 'A' and 'B', and then, bam! Something unexpected pops out. Specifically, does a
Think of it like this: you've got two perfectly nice, clear liquids. They're chilling, minding their own business. Maybe liquid A is feeling a bit lonely, and liquid B is looking for a party. So, they mix. It's like a blind date set up by Mother Nature. And sometimes, these dates go spectacularly well, resulting in a beautiful, homogeneous solution. Other times? It's a total disaster. They get together, realize they have nothing in common, and one of them just throws up its hands (or, you know, molecules) and solidifies. That solid stuff? That's our
Now, why would this happen? It's all about the
Let's get a bit more specific. Imagine you have a solution of, say, silver nitrate. Clear as day. Then, you add another solution, this one containing sodium chloride. Now, normally, you'd expect these to just sort of... blend. But here's where the magic (and by magic, I mean chemistry) happens. The silver ions (Ag+) and the chloride ions (Cl-) are like soulmates who just haven't met yet. When they get the chance to interact in the same space, they realize, "Whoa, you're the one!" and they immediately ditch their original partners (nitrate and sodium, who are perfectly happy staying dissolved) to form a new, very stable, and insoluble compound: silver chloride (AgCl).
And this silver chloride? It doesn't want to stay dissolved. It’s like that one friend who always gets way too much attention at parties and ends up making a scene. It clumps together, forming solid little particles that you can see. These particles, these little rebels, are the
This isn’t just a weird laboratory phenomenon, either. Precipitation is happening all around us, and it’s pretty important. Think about
Or consider

So, how do we know if a precipitate is going to form? Well, thankfully, we don't have to just guess and hope for the best. Chemists have these handy-dandy
There are even some hilarious exceptions. Like, you might think, "Okay, so if something's insoluble, it'll always be a solid." But sometimes, the chemistry gods are playing a prank. You might get a gelatinous precipitate, which is basically a wobbly, semi-solid that’s more like a really thick Jell-O than a distinct solid. Or, you could get a

The amount of precipitate you get is also a big deal. Sometimes, you get a tiny dusting, like someone sneezed glitter. Other times, it’s a thick sludge that could choke a drain. This depends on how much of the reactive ingredients you have. If you add a tiny bit of, say, sodium chloride to your silver nitrate, you'll get a small amount of silver chloride precipitate. But if you dump in a whole shaker? You'll get a significant amount of that white, powdery stuff. It’s like adding a pinch of salt to a soup versus adding the whole box – the results are, shall we say, dramatically different.
In essence, when you mix A and B, and a
