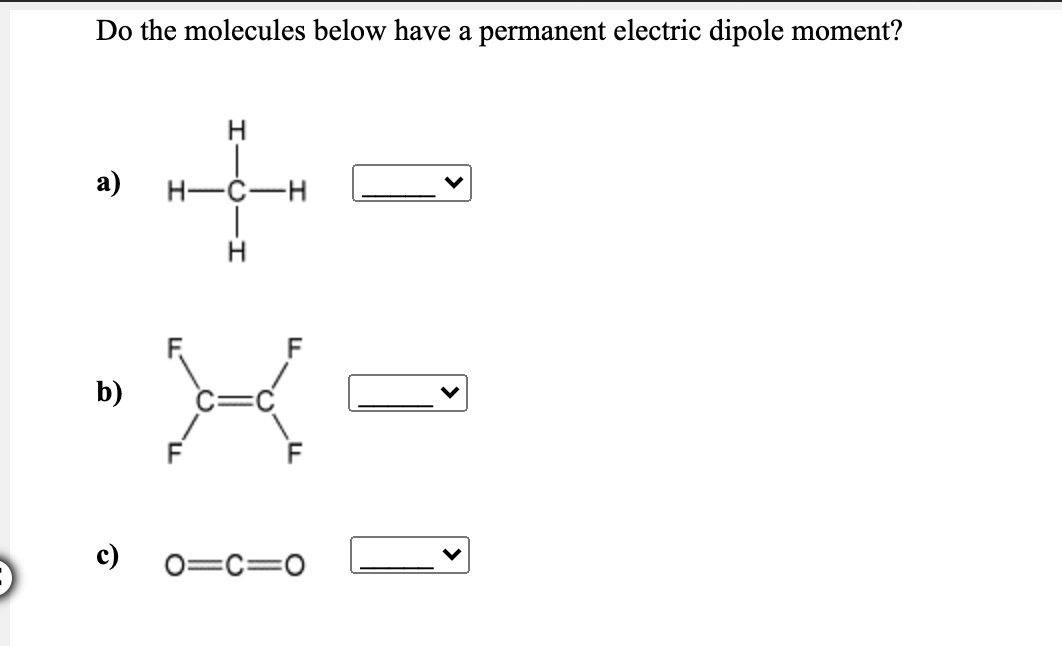
Do The Molecules Below Have A Permanent Electric Dipole Moment
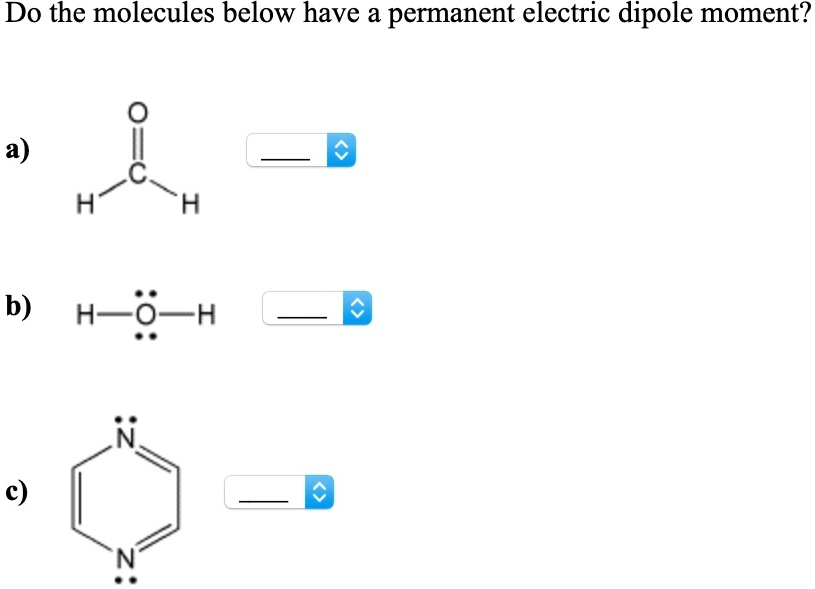
Hey there, lovely people! Ever find yourself pondering the tiny invisible forces that shape our world? We’re not talking about the cosmic kind here, but the super-duper small stuff that’s happening all the time, even in that mug of perfectly brewed coffee you’re sipping right now. Today, we’re diving into a topic that sounds a little science-y, but trust me, it’s as chill and relevant as your favorite playlist. We’re talking about electric dipole moments – and whether some of your everyday molecules are rocking them.
Think of it like this: atoms are the building blocks, and when they team up to form molecules, they create little teams. Sometimes, these teams are super balanced, like a perfectly symmetrical yoga pose. Other times, one atom in the team is a bit more clingy with its electrons, giving it a slightly negative vibe, while another is a little more generous, giving it a slightly positive vibe. This uneven distribution of charge within a molecule? That’s what we call a permanent electric dipole moment.
Why should you care? Because this little charge imbalance is the secret sauce behind so much of our daily lives! It influences how substances interact, how they dissolve, how they behave in fields – basically, a whole lot of the magic that makes chemistry, and by extension, our world, work.
So, let’s get down to the nitty-gritty. We’re going to look at a few common molecules and suss out whether they're sporting a dipole moment like a designer accessory or keeping things totally neutral, like a minimalist chic vibe.
The Usual Suspects: Let's Take a Peek!
We’ve all heard of these guys, they’re practically celebrities in the molecular world. First up, the undisputed champ, the universal solvent, the lifeblood of our planet: Water (H₂O)!
Water is like the supermodel of molecules. It’s got that distinctive bent shape. Oxygen is a real electron-lover, meaning it pulls those shared electrons closer to itself. This makes the oxygen side of the water molecule a little bit negative, and the hydrogen sides, well, a little bit positive. Because of this bent geometry, these positive and negative poles don't cancel each other out. It’s like having a tiny, built-in battery!
So, does water have a permanent electric dipole moment? You bet it does! This is why water is such an amazing solvent for so many things – it can interact with and dissolve polar substances. It’s also why water molecules stick together so nicely, forming those lovely dew drops on your morning newspaper or creating that satisfying splash when you dive into a pool. It’s all thanks to that little dipole!
Next on our molecular catwalk, we have Carbon Dioxide (CO₂). This is the stuff we breathe out, the stuff that makes fizzy drinks go fizz. It’s made of one carbon atom and two oxygen atoms, all lined up in a straight line: O=C=O.
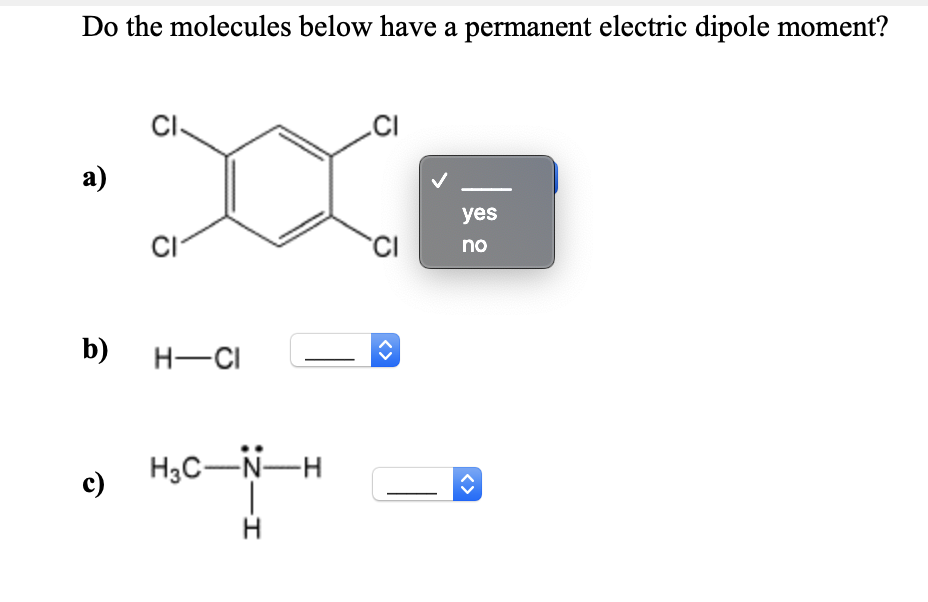
Now, oxygen is still an electron-lover, so it pulls electrons from the carbon. This gives the oxygen atoms a slightly negative charge and the carbon atom a slightly positive charge. BUT! And this is a big but, CO₂ is a linear molecule. Imagine two equally strong tug-of-war players pulling in opposite directions on a central point. The pulls cancel each other out perfectly.
So, does carbon dioxide have a permanent electric dipole moment? Nope! It’s nonpolar. Even though there are polar bonds within the molecule, the symmetry of the molecule means the overall charge distribution is even. Think of it as a perfectly balanced seesaw – no lean in either direction.
This is why you can add carbon dioxide to water, and it won’t dissolve as readily as salt or sugar. It just chills on the surface, waiting for a bit of pressure to get things going. It’s a bit like a lone wolf in the molecular world, not easily mingling with the polar crowd.
Let’s talk about Ammonia (NH₃). This one smells… distinctive. Think cleaning products, or maybe that quirky fertilizer smell your gardener neighbor uses. Ammonia has a central nitrogen atom bonded to three hydrogen atoms. The nitrogen is a pretty strong electron-lover.
The nitrogen atom pulls the electrons towards itself, creating a negative charge on the nitrogen end and positive charges on the hydrogen ends. Ammonia isn't linear; it has a trigonal pyramidal shape, sort of like a tiny pyramid or a three-legged stool. This shape means the charges don't cancel out. There's a clear negative pole and a clear positive pole.
So, does ammonia have a permanent electric dipole moment? Absolutely! It's a polar molecule. This polarity is a big reason why ammonia is soluble in water and can form hydrogen bonds, which are super important in many biological and chemical processes. It’s like the social butterfly of the polar molecules.
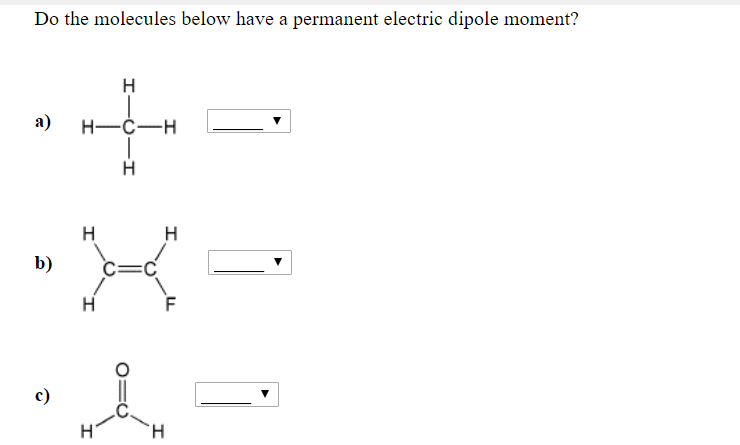
Now, for something a bit different: Methane (CH₄). This is the primary component of natural gas, the stuff that heats our homes and cooks our food. Methane has a central carbon atom bonded to four hydrogen atoms. The shape is wonderfully symmetrical – tetrahedral, like a perfect pyramid with a base.
Carbon is slightly more electronegative than hydrogen, so it does pull the electrons a tiny bit. This would create small partial charges. However, because the molecule is so symmetrical, and the carbon-hydrogen bonds are arranged evenly in space, these tiny charges perfectly cancel each other out.
So, does methane have a permanent electric dipole moment? No, it does not. It's a nonpolar molecule. This is why natural gas, being nonpolar, doesn't mix well with water. It’s the molecular equivalent of oil and water, always keeping its distance. This nonpolar nature is also why it burns so cleanly – it’s a simple, efficient fuel.
Why Does This Even Matter in Our Chill Life?
You might be thinking, "Okay, so some molecules are a bit lopsided in their electron distribution, big deal!" But here’s the cool part: this seemingly small difference has HUGE implications for how we live.
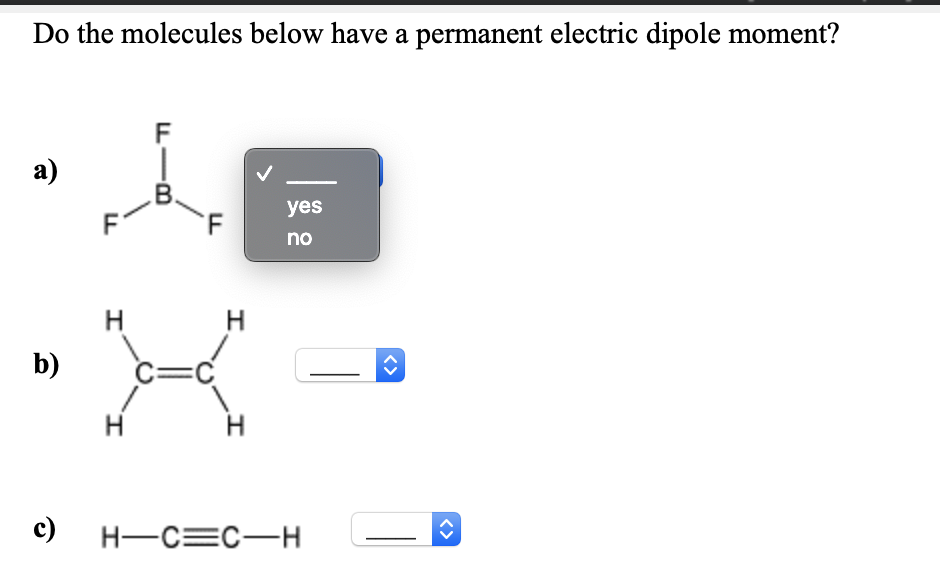
Take solubility. Remember how water, our polar champion, dissolves salt and sugar? That's because the positive ends of water molecules are attracted to the negative ions in salt, and the negative ends of water are attracted to the positive ions. Polar dissolves polar, and nonpolar dissolves nonpolar. This is why you can’t just dump oil into your vinaigrette and expect it to mix; you need an emulsifier to bridge the gap!
Think about biological systems. Your body is a bustling metropolis of molecules, and their interactions are key to everything from digesting your lunch to sending signals from your brain. Proteins, which are made of amino acids, have polar and nonpolar regions. This allows them to fold into complex shapes and interact with other molecules in very specific ways. The water in your cells, being polar, plays a crucial role in keeping these structures stable and functional.
Even something as simple as cleaning relies on dipole moments. Soap molecules are clever little things. They have a polar "head" that loves water and a nonpolar "tail" that loves grease and oil. This allows them to surround dirt and grime, lifting it from your dishes or clothes and allowing it to be washed away by water. It’s like a tiny molecular tugboat!
Consider the difference between cooking oils (largely nonpolar) and vinegar (which contains water and acetic acid, both polar). Mixing them without an emulsifier results in separation. But add a dash of mustard (which has emulsifying properties), and they might just get along for a while. It’s a little bit of molecular diplomacy happening on your salad!
Fun Facts & Cultural Dips
Did you know that the unique properties of water, largely due to its strong dipole moment and hydrogen bonding, are what allow fish to swim and plants to grow? If water were nonpolar, it might all be frozen solid or evaporated into nothingness!
The concept of polarity even creeps into art and design. Think about how artists use contrast. In a way, the contrast between polar and nonpolar molecules is a fundamental aspect of the universe’s "design." It creates diversity and enables complex interactions.
And in pop culture? While not explicitly stated, the idea of things "attracting" or "repelling" based on inherent properties is a recurring theme. Think of opposites attracting – it’s a simplified, romanticized version of molecular interactions!
Quick Recap for the Curious Mind:
- Water (H₂O): Bent shape, uneven electron distribution. Polar molecule with a permanent dipole moment.
- Carbon Dioxide (CO₂): Linear shape, symmetrical electron distribution. Nonpolar molecule, no permanent dipole moment.
- Ammonia (NH₃): Pyramidal shape, uneven electron distribution. Polar molecule with a permanent dipole moment.
- Methane (CH₄): Tetrahedral shape, symmetrical electron distribution. Nonpolar molecule, no permanent dipole moment.
It’s fascinating how these incredibly small, invisible characteristics can dictate the macroscopic world we experience. It’s the silent symphony playing out all around us.
A Little Reflection: The Art of Balance
Thinking about these molecules and their dipoles – their inherent balance or lack thereof – can actually be a pretty neat metaphor for life, right? We all strive for balance, for a sense of equilibrium in our lives. Sometimes we’re perfectly balanced, like a symmetrical molecule, and everything just flows. Other times, we feel a bit… lopsided. One area of our life might be pulling all the "electron-energy," leaving other areas feeling a bit deprived.
Recognizing these imbalances, whether in our molecules or in our own days, is the first step to finding a healthier, more harmonious flow. Just like a skilled chemist can manipulate molecular interactions, we can learn to adjust our own "molecular" composition – our priorities, our habits, our focus – to create a more balanced and fulfilling existence. So next time you’re enjoying a glass of water, or breathing in that crisp air, give a little nod to the amazing world of molecular interactions that’s making it all possible. It’s pretty cool stuff, and it’s all happening, effortlessly, all the time.
