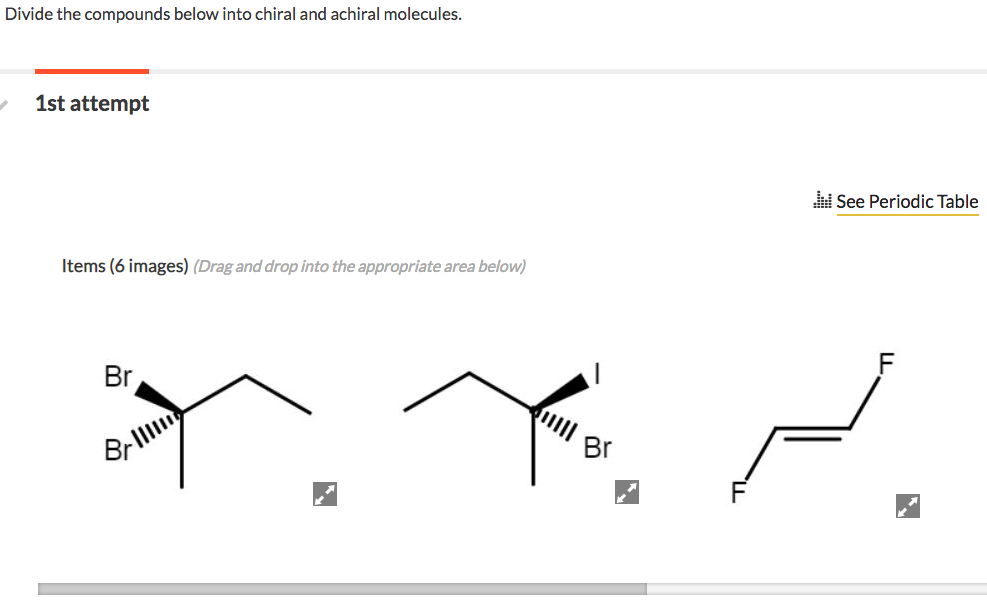
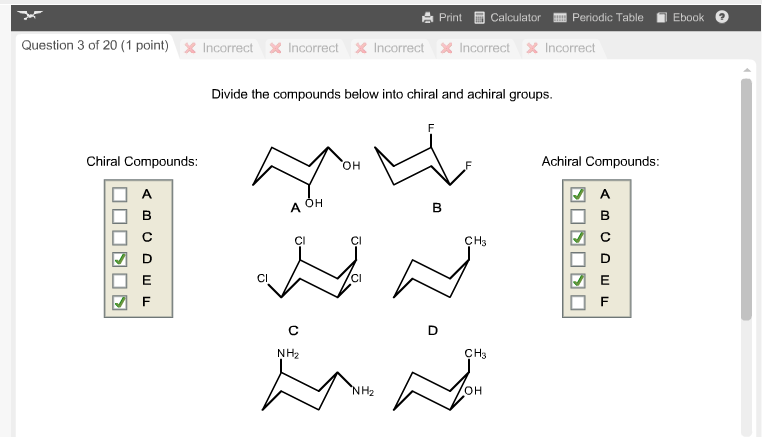
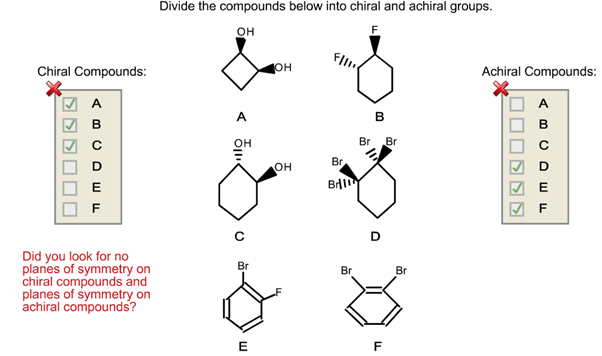
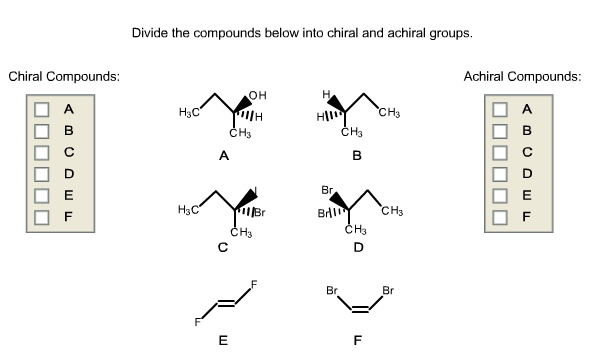
Divide The Compounds Below Into Chiral And Achiral Molecules.
Hey there, science enthusiasts and curious minds! Ever looked at a molecule and thought, "Is this thing a lefty or a righty?" Well, get ready to have your world a little bit… mirrored! Today, we’re diving into the wonderfully quirky world of chirality, and trust me, it’s way more fun than it sounds. Think of it as a molecular fashion show, where some molecules strut their stuff with a distinct sense of style, while others are happy to be a bit… well, generic.
So, what’s the big deal? It all boils down to something called a chiral center. Imagine you have a carbon atom (the superstar of organic chemistry, as you probably know!) bonded to four different things. This little setup is like the molecule’s version of having hands. Just like you have a left hand and a right hand that are mirror images of each other but aren’t quite the same, a molecule with a chiral center can exist in two forms, called enantiomers. These enantiomers are non-superimposable mirror images. Pretty neat, huh?
Now, why should you care about molecular handedness? It turns out, this chirality thing is HUGE in the real world. Think about your body – it’s practically a chiral wonderland! Many of the biological molecules that make you, you, are chiral. Proteins, sugars, even your DNA – they all have this left-handed or right-handed flavor. And guess what? Your body is usually very particular about which hand it prefers. It’s like trying to wear a left glove on your right hand – it just doesn’t feel quite right, does it?
Chiral vs. Achiral: The Molecular Showdown!
Let’s get down to the nitty-gritty, or as I like to call it, the molecular showdown. We’re going to break down some compounds and see who’s rocking the chiral vibe and who’s more of an achiral wallflower. Ready to put on your molecular detective hats?
The Chiral Crew: The Lefties and Righties!
These molecules are the showstoppers, the ones with that special spark. How do we spot them? We look for that chiral center we talked about. It’s usually a carbon atom with four different groups attached. When you find one, BAM! You’ve likely got a chiral molecule on your hands.
Let’s take a classic example: Lactic Acid. Ever heard of it? It’s that stuff that makes your muscles burn after a tough workout. Lactic acid has a carbon atom bonded to a hydrogen atom, a hydroxyl group (-OH), a carboxyl group (-COOH), and a methyl group (-CH3). See? Four different things! This means lactic acid exists as two enantiomers, (R)-lactic acid and (S)-lactic acid. Your body actually uses the (S)-lactic acid more efficiently. Fascinating, right? It’s like your body has a preferred dance partner!
Another cool one is Alanine, one of the building blocks of proteins. Alanine also has a central carbon attached to a hydrogen, a carboxyl group, an amino group (-NH2), and a methyl group. Yep, another chiral center, another pair of enantiomers. The alanine your body uses is typically the L-alanine, which corresponds to the (S) configuration. It’s these subtle differences that can lead to dramatically different effects in biological systems.
Even something as seemingly simple as 2-butanol is chiral. Its central carbon is attached to a hydrogen, a hydroxyl group, a methyl group, and an ethyl group. This seemingly small structural detail means that 2-butanol exists as two mirror-image forms, and they can behave differently in chemical reactions or when interacting with other chiral molecules.
So, the key takeaway for our chiral friends is: look for that carbon with four unique attachments. If you find it, you’re probably dealing with a molecule that has a mirror image that’s not quite the same. It’s like finding a pair of perfectly matched, but distinctly left and right, gloves!
The Achiral All-Stars: The Versatile & The Identical!
Now, let’s talk about the achiral molecules. These guys are the chill ones, the ones who don’t have that distinct handedness. What makes them achiral? Usually, it’s because they have some kind of symmetry. They might have a plane of symmetry, meaning you can slice them in half and both halves are identical. Or, they might have a rotational axis of symmetry where rotating them by a certain angle brings them back to their original appearance.
Think about something super simple, like water (H2O). It's got two hydrogen atoms bonded to an oxygen. There's no way to attach four different things to the oxygen, and there’s plenty of symmetry here. Water is achiral. It’s the same no matter how you look at it, or rotate it.
How about methane (CH4)? That central carbon is bonded to four hydrogen atoms. Since all the attachments are the same, it can’t have four different ones. Methane is achiral. It’s perfectly symmetrical.
Consider ethanol (CH3CH2OH). While it has a hydroxyl group, if you look at the carbon atoms, neither of them is bonded to four different groups. The first carbon is bonded to three hydrogens and another carbon. The second carbon is bonded to two hydrogens, a carbon, and a hydroxyl group. No chiral center here, so ethanol is achiral. It doesn’t have a distinct left or right-handed form.
Let’s take a slightly more complex example, like acetone (CH3COCH3). The central carbon is double-bonded to an oxygen and single-bonded to two methyl groups. Since it's not bonded to four separate groups (it has a double bond and only two types of single bonds), it cannot be a chiral center. Acetone is achiral.
The key for our achiral pals is the lack of a chiral center. They might be symmetrical, or just not have that perfect setup for creating those non-superimposable mirror images. They’re the molecules that are the same forwards and backward, so to speak, from a structural perspective.
Why This Matters (Besides Being Super Cool!)
So, why all this fuss about chirality? It's not just an academic exercise, folks! This stuff has serious real-world implications. Think about pharmaceuticals. Many drugs are chiral, and often, only one enantiomer will have the desired therapeutic effect, while the other might be inactive or even cause harmful side effects! Remember the thalidomide tragedy? It's a stark reminder of how critical chirality can be. One enantiomer was a sedative, while the other caused severe birth defects. Yikes!
In food science, chirality affects taste and smell. For instance, one enantiomer of carvone smells like spearmint, while the other smells like caraway. Isn't that wild? The same molecule, just a different handedness, and a completely different sensory experience!

Even in our environment, chiral molecules play a role. Microorganisms often have a preference for one enantiomer over another when breaking down substances. This can impact how quickly pollutants are degraded or how certain fertilizers work.
Learning about chirality opens up a whole new way of looking at the world around you. It shows you the incredible complexity and elegance of nature at its most fundamental level. It's a peek behind the curtain, revealing that even seemingly identical molecules can have vastly different personalities and properties based on their "handedness."
So, the next time you encounter a molecule, don’t just see a jumble of atoms. See if you can spot that potential chiral center. Can you imagine its mirror image? Is it the same, or is it a unique, non-superimposable twin? This little bit of molecular sleuthing can make chemistry so much more engaging and, dare I say, fun!
Embrace the quirkiest side of chemistry. Dive deeper into the fascinating world of enantiomers and stereochemistry. You might just discover a new passion for the hidden symmetry (or glorious asymmetry!) that shapes our universe. Keep exploring, keep questioning, and keep being amazed by the molecular wonders that surround us!
