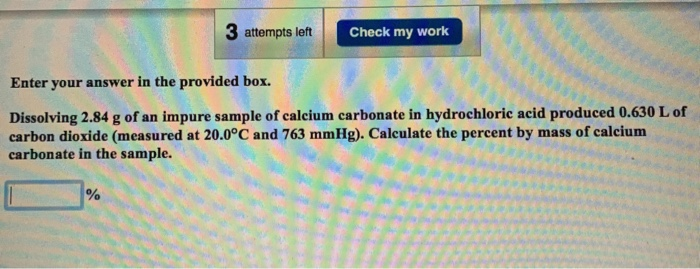
Dissolving 3.00 G Of An Impure Sample Of Calcium
Hey there, science enthusiasts and curious minds! Ever wondered what happens when you take a little bit of stuff and make it disappear? Well, get ready for a surprisingly fun ride because we're diving into the exciting world of dissolving a 3.00 g impure sample of calcium. Now, I know what you might be thinking: "Dissolving? Sounds…wet." And you're not wrong! But trust me, there's more magic happening here than you might imagine.
Imagine you've got this tiny little pile, just 3.00 grams, of something that's supposed to be calcium. But here's the twist – it's not pure! Think of it like a cookie that’s got a few extra chocolate chips you weren't expecting, or maybe a few too many sprinkles. It’s still a cookie, but it's got its own little personality. That’s our impure sample of calcium. It’s mostly calcium, but with a few friendly (or not-so-friendly!) impurities tagging along for the ride.
So, what do we do with this intriguing little sample? We’re going to dissolve it! Now, dissolving isn't just about making things vanish into thin air. It's like unlocking a secret. You’re taking something solid and introducing it to a liquid buddy, and together, they become a whole new thing – a solution! It’s a bit like making hot chocolate. You’ve got your solid cocoa powder and your milk, and when they hang out, they create that delicious, smooth drink. Pretty neat, right?
Our calcium, bless its impure heart, needs a special liquid friend to get the job done. And that friend is usually something like a dilute acid. Don’t let the word "acid" scare you! We’re talking about the gentle kind here, the kind that’s happy to make friends with metals. Think of it as a friendly handshake that’s so firm, it helps the calcium break apart and become part of the liquid.
When our 3.00 g impure sample of calcium meets the dilute acid, it’s like a little party starting. The calcium atoms, which were all snuggly packed together in their solid form, start to get a little antsy. The acid molecules come in, saying, "Hey, calcium! Want to come out and play?" And the calcium, especially the pure calcium bits, are all like, "Sure, sounds fun!" They start to let go of each other and get surrounded by the acid and water molecules. It’s a beautiful breakup, really, but a necessary one for our scientific adventure.
![[ANSWERED] 58 Igofan impure sample of calcium carbonate containing no](https://media.kunduz.com/media/sug-question-candidate/20210607074509220814-1461613.jpg?h=512)
But what about those impurities? Ah, that’s where the real fun begins! Because our sample isn't pure, not everything will dissolve. Some bits might be like the shy guests at the party, not wanting to mingle with the acid. They might just sit there, unmoved, or maybe they’ll do their own thing. This is what makes dissolving an impure sample so much more interesting than dissolving something perfectly pure.
Imagine you're sorting through a bag of candy. Some pieces you gobble right up, others you might eye suspiciously, and some you might just set aside. The impurities in our calcium are like those "set aside" candies. They don't react with the dilute acid in the same way the calcium does, or maybe they don't dissolve at all.

So, as our 3.00 g impure sample of calcium does its dissolving dance, a couple of things can happen. The calcium itself might transform into something soluble, like calcium ions floating happily in the liquid. This is where the visible action is – the solid chunk gets smaller and smaller until, poof, it's gone! But the impurities? They might stay solid, forming a little sediment at the bottom of the container. Or, they might be gases that bubble up, like tiny effervescent cheers from the unreactive bits.
Watching this happen is surprisingly satisfying. It's like a mini-mystery unfolding right before your eyes. You put in your 3.00 grams, add the liquid, and then you observe. You see the solid disappear. You might see bubbles. You might see a little bit of leftover stuff. Each observation is a clue, helping you understand what was really in that impure sample of calcium.
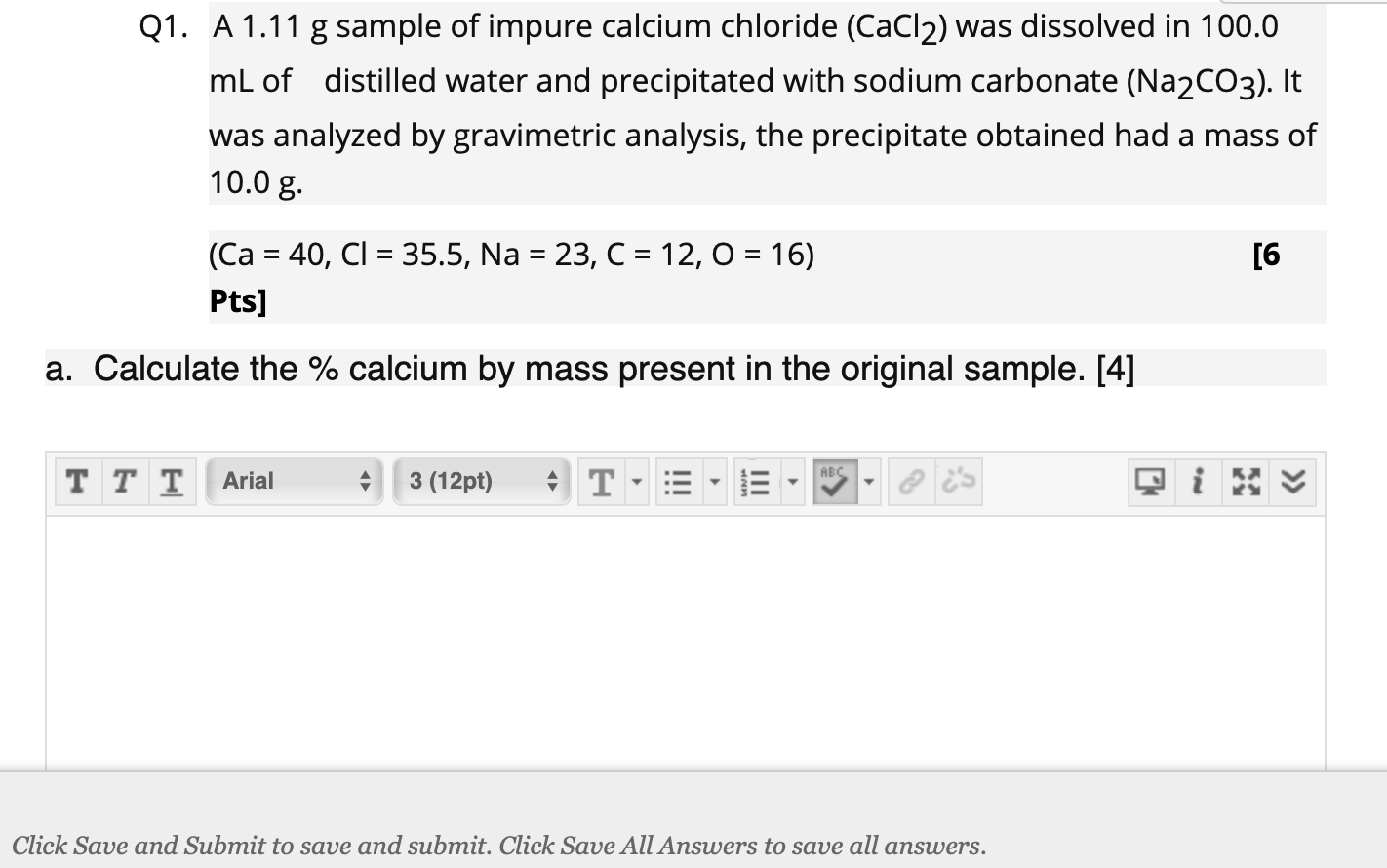
Why is this so special, you ask? Because it's a window into the real world! In labs and in nature, things are rarely perfectly pure. Everything has a story, a history, and often, a few extra ingredients. By dissolving an impure sample, we're getting a hands-on lesson in how to deal with the messy, imperfect, but ultimately fascinating reality of chemistry. It’s not just about making something disappear; it’s about learning what’s there, what reacts, and what's left behind. It’s about discovery!
And the best part? You don't need a super fancy lab coat to appreciate this. You can visualize it, you can think about the reactions, and you can understand the basic principles. It’s a little snippet of science that’s accessible, visually engaging (if you imagine it!), and teaches you something fundamental. So next time you hear about dissolving a 3.00 g impure sample of calcium, don't just picture a wet mess. Picture a scientific investigation, a tiny adventure, and a peek into the wonderful, imperfect world of matter!
