Dissolved Oxygen And Aquatic Primary Productivity Lab Answers

Alright, gather 'round, you lovely landlubbers, and let me tell you a tale of tiny titans and underwater oxygen capers. We're diving deep, not into a murky swamp (though sometimes it feels like it), but into the glorious world of Dissolved Oxygen and Aquatic Primary Productivity. Yeah, I know, it sounds like something straight out of a ridiculously boring documentary narrated by a robot with a cold. But trust me, this is where the real drama unfolds. This is the stuff that keeps our watery friends breathing and the tiny green machines of the ecosystem humming.
So, imagine this: you’re a microscopic plant, a phytoplankton, chilling in a nice, sun-drenched pond. Your job? To photosynthesize like your life depends on it. Because, well, it does. You’re basically the microscopic equivalent of a fast-food chef, churning out energy for the whole neighborhood. And what’s the secret ingredient to your culinary success? Sunshine and… you guessed it… dissolved oxygen. Yep, even the tiny plants need their O2 fix. It’s like they’re running a marathon in slow motion and need to breathe.
The Great Oxygen Mystery
Now, the lab that probably sent you scrambling for your notes (and maybe a strong cup of coffee) was all about figuring out just how much of this magical dissolved oxygen is floating around and how it relates to these little green powerhouses. Think of it as a detective story, but instead of a shadowy figure, we’re chasing down elusive oxygen molecules. Our main suspects?
First up, we have our phytoplankton. These guys are the MVPs of primary productivity. They’re the ones taking sunlight, water, and CO2 and transforming them into glorious, life-sustaining food. And as a fantastic byproduct of their hard work, they spit out oxygen. It’s like they’re little green factories with incredibly efficient ventilation systems. The more they photosynthesize, the more oxygen they pump into the water. It’s a win-win, a real feel-good story for the aquatic community.
But then, there’s the shady character: respiration. Everything that lives in the water, from the tiniest bacteria to your pet goldfish (if you’re brave enough to have one), needs to breathe. And when they breathe, they gobble up oxygen. So, it's a constant tug-of-war. Plants make it, critters take it. It’s like a really polite, silent battle for survival happening 24/7.

The Lab Shenanigans: What Did We Actually Do?
So, the lab was designed to catch these guys in the act. We probably had bottles. Lots of little glass bottles, looking all innocent. And we probably filled them with water from a pond, a lake, or maybe even a suspiciously green swimming pool. The real trick came next. We took our water samples and then we did… science.
One set of bottles likely sat out in the glorious, life-giving sunlight. These were our "photosynthesis in action" samples. We expected these to show a nice, healthy increase in dissolved oxygen, right? Because, you know, plants making oxygen. Duh. It’s like leaving cookies out for Santa, you expect a reward.
The other set of bottles? These were probably shoved into a dark box, or a cupboard, or maybe just hidden under a pile of textbooks. These were our "respiration only" samples. In the dark, no sunlight means no photosynthesis. So, the only thing happening here is the critters in the water having a big ol’ oxygen feast. We expected these bottles to show a decrease in dissolved oxygen. It's like turning off the oven; the cookies are still there, but no new ones are being made, and the existing ones might get a bit stale (or in this case, consumed).
And then, the moment of truth! We probably used some fancy gizmo – a dissolved oxygen meter, perhaps? – to measure the O2 levels in each bottle. This is where the magic (or the madness) happened. We were looking for a difference. A significant, quantifiable difference between the sunlit bottles and the dark bottles.
The Glorious Results (and Why They Matter)
If your sunlit bottles showed a healthy jump in dissolved oxygen, and your dark bottles showed a dip, congratulations! You've witnessed primary productivity in action. These are the signs of a happy, healthy aquatic ecosystem. It means the phytoplankton are doing their jobs, feeding the world, and keeping the water nice and breathable for everyone else. Think of it as the ecosystem giving you a thumbs-up. A tiny, bubbly thumbs-up.

Now, the actual numbers are where things can get a bit… nuanced. Did your sunlit bottle go from practically a vacuum to overflowing with oxygen? Awesome! Did it only go up by a smidgen? Well, maybe that pond is a bit… less enthusiastic. Or perhaps there are a LOT of oxygen-guzzling critters working overtime. It’s a delicate balance, you see. Like a tightrope walker juggling flaming torches while riding a unicycle.
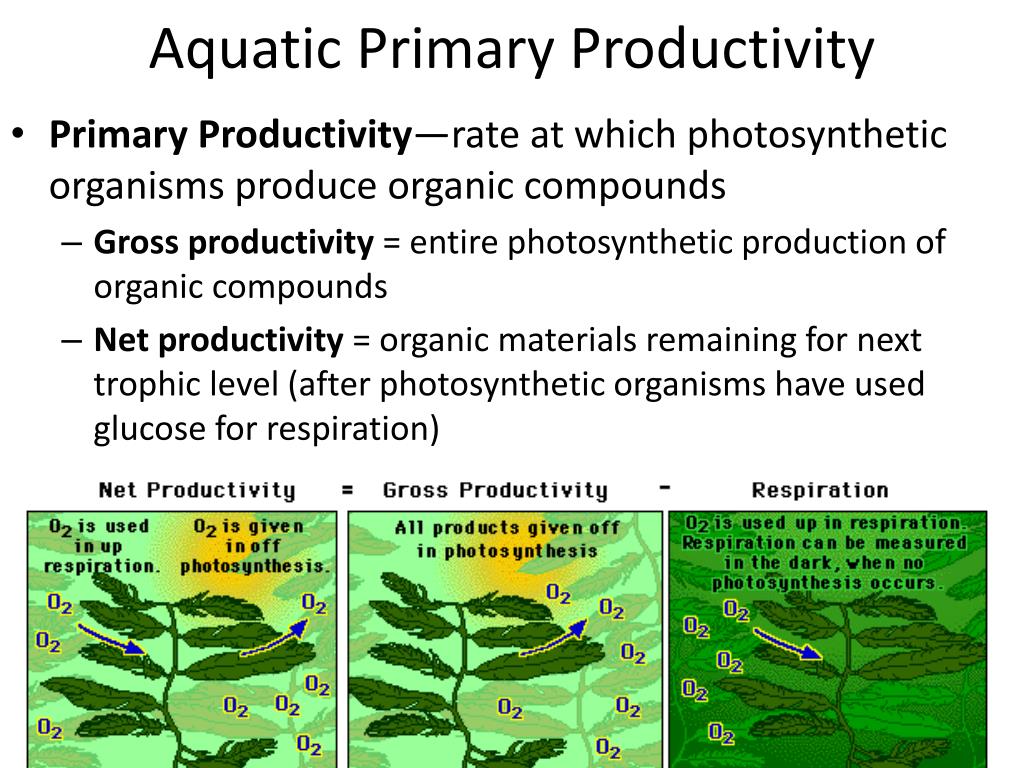
The difference between the initial dissolved oxygen levels and the final levels in the sunlit bottle gives you an idea of the net primary productivity. It’s the oxygen produced by the plants, minus the oxygen used by them (and anything else respiring). The difference between the initial and final levels in the dark bottle tells you about the respiration rate. And if you do some clever math (which, let’s be honest, is the real challenge for most of us), you can even estimate the gross primary productivity – the total oxygen produced by photosynthesis, before any of it gets eaten back up.
The Surprising Truths and the Silly Bits
Here’s a fun fact for you: Phytoplankton are responsible for producing somewhere between 50% and 85% of the oxygen on Earth. Yes, you read that right. Those microscopic specks of green are basically our planet's lungs. So, next time you take a deep breath, spare a thought for your little phytoplankton pals out there, working their microscopic butts off.

Another thing to consider: Water temperature plays a HUGE role. Colder water can hold more dissolved oxygen than warmer water. It’s like a cozy blanket for oxygen molecules. So, if your pond is super warm, it might have a harder time holding onto its O2, even if the plants are working overtime. It’s a bit like trying to pack a suitcase for a trip to the tropics – things just don't fit as snugly.
And let’s not forget about the other players. Algae blooms? While they look pretty (sometimes), a massive bloom can lead to a huge spike in productivity, followed by a massive die-off. When all those dead algae decompose, they consume a TON of oxygen, leading to an oxygen sag. This is bad news for fish, who can suffocate and make a dramatic exit. It's the aquatic equivalent of a party gone wrong.
So, when you were staring at those numbers, trying to make sense of them, you were essentially peering into the very engine of aquatic life. You were seeing the ebb and flow of oxygen, the silent symphony of production and consumption. You were getting answers to the lab questions, yes, but you were also glimpsing something much grander: the intricate, often hilarious, and absolutely vital dance of life in our waters. And that, my friends, is way cooler than it sounds.
