Diffusion And Osmosis Worksheet Answers Page 2

Hey there, science fans (and curious cats)! So, you've been wrestling with a diffusion and osmosis worksheet, and maybe you've even peeked at Page 2 for those elusive answers. No judgment here! Sometimes, the best way to truly understand something is to see how it plays out, and those answer keys can be like little cheat codes for your brain. But what’s so fascinating about this stuff anyway? It’s not exactly rocket science, right? Well, believe it or not, diffusion and osmosis are happening all around us, and inside us, all the time. They're like the tiny, invisible ballet dancers of the microscopic world, constantly moving and shaping everything.
Think about it. Have you ever walked into a room where someone’s baking cookies? That amazing smell doesn't just magically appear where you are, does it? Nope! Those delicious cookie molecules are diffusing through the air, spreading out from the oven until they reach your nose. It’s kind of like when you spray a little perfume in one corner of a room, and pretty soon, the whole place smells like a spring meadow (or, you know, whatever your preferred scent is). That’s diffusion in action – molecules spreading from an area where there are a lot of them to an area where there are fewer. Simple, right? But also, kind of… elegant?
So, What's the Big Deal with Diffusion?
Diffusion is all about things moving to find their happy place, a state of equilibrium. Imagine a bunch of hyperactive kids in a classroom. They’re all crammed together at the start, but eventually, they’ll spread out to take up all the available space. Molecules are kind of like that. They’re always on the move, bouncing around, and they’ll keep moving until they’re evenly distributed. It’s a fundamental principle in nature, and it’s crucial for so many processes.
On your worksheet, you might have seen questions about diffusion rates. What affects how fast those molecules spread? Things like temperature (hotter means faster movement, like a hot pan making water boil quicker), concentration gradient (a bigger difference in the number of molecules means faster spreading, like a crowded elevator emptying out faster), and the size of the molecules (smaller molecules zip around faster). It's all about those tiny particles having a good old dance party.
And it’s not just about smells. Diffusion is super important for things like:

- How plants get nutrients from the soil.
- How oxygen gets from your lungs into your bloodstream.
- How waste products leave your cells.
Pretty cool for something that seems so basic, huh?
Now, Let's Talk About Osmosis: Diffusion's Fancy Cousin
Osmosis is like diffusion’s more particular, special-occasion sibling. It’s a specific type of diffusion, but it only involves water (or other solvent) molecules, and it happens across a selectively permeable membrane. What’s that, you ask? Think of it as a bouncer at a club. It lets some things in (like water), but it’s a bit picky about what else gets through. Cell membranes are the ultimate bouncers for our cells!

So, osmosis is the movement of water from an area of high water concentration (which means low solute concentration, like pure water) to an area of low water concentration (which means high solute concentration, like salty water). Why? Again, it’s all about reaching balance. The water is trying to dilute the concentrated side, to even things out.
Osmosis in Real Life: From Raisins to Your Own Cells
Ever put a dried-out raisin in a glass of water and watched it plump up? That’s osmosis! The water from the glass, which has a higher water concentration, moves into the raisin, where the sugars are more concentrated. Your worksheet might have had questions about this. Maybe you had to draw diagrams or explain what happens to a cell in different solutions.

And here’s where it gets really personal: osmosis is absolutely vital for your body. Your red blood cells, for instance, have to be in just the right environment. If they’re placed in pure water (a hypotonic solution), water rushes into them, and they might swell and burst – not ideal! If they’re placed in super salty water (a hypertonic solution), water rushes out, and they shrivel up like tiny, sad raisins. You want them to be in an isotonic solution, where the water concentration inside and outside the cell is balanced, and everything stays happily plump.
Think of it like a tug-of-war. The water molecules are the rope, and the membrane is the center line. The side with more “stuff” (solutes) on it is pulling the water towards it. Osmosis is the natural tendency for that water to move and try to make the “pull” equal on both sides. It’s a constant, silent negotiation happening at a cellular level.
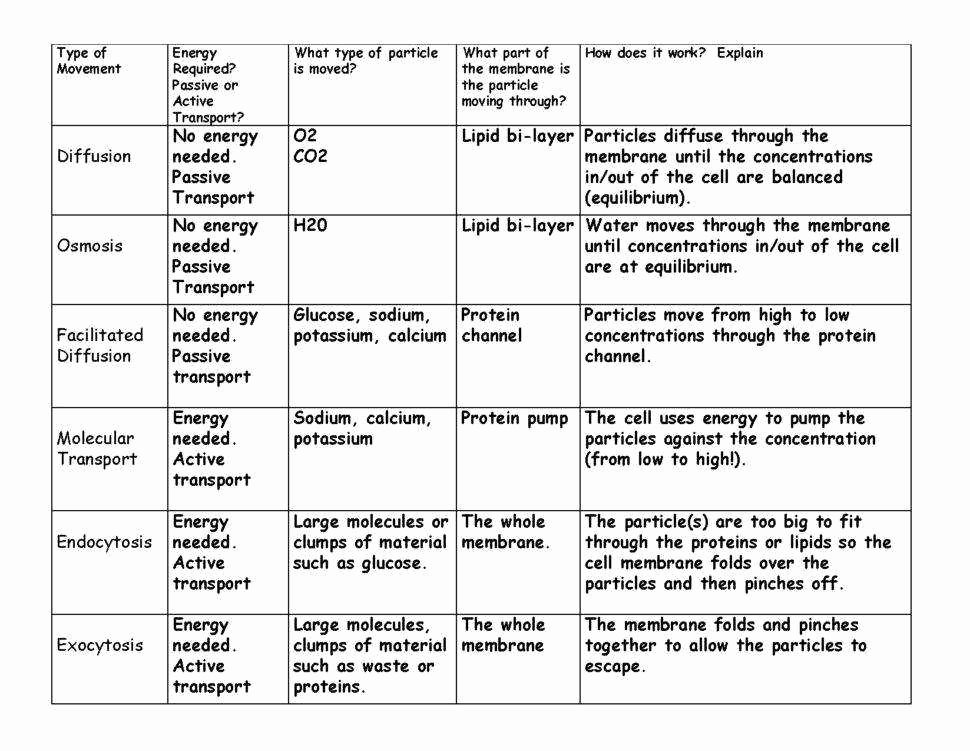
Page 2 and Beyond: Making Sense of the Answers
So, when you’re looking at those answers on Page 2, don’t just see them as correct or incorrect. See them as explanations. Each correct answer is a little lightbulb moment, showing you why things happen the way they do. Did you get a question wrong about red blood cells in a hypotonic solution? The answer probably explained that water moves in, causing the cell to swell. Now you know!
It’s like learning a new language. At first, the grammar rules might seem confusing, but once you start seeing how sentences are formed and how words connect, it all starts to click. Those worksheet answers are your sentence examples. They show you the pattern, the underlying logic. They’re not just about memorizing facts; they’re about understanding a fundamental process that governs life itself.
And honestly, isn’t that amazing? That these simple-sounding concepts of molecules moving around have such profound implications? From the freshness of your produce to the very functioning of your organs, diffusion and osmosis are the unsung heroes. So, the next time you’re working on a worksheet, or even just enjoying a glass of water, take a moment to appreciate the invisible world of molecular movement. It’s a tiny universe at play, and it’s pretty darn cool.
