Diffusion And Osmosis Beaker Worksheet Answer Key
Alright, science explorers and curious cats! Ever feel like you’ve been staring at a science worksheet, particularly one about diffusion and osmosis, and felt a tiny bit… bewildered? Like you’ve been transported to a land where tiny particles have their own secret dance moves? Well, take a deep breath and un-squint those eyebrows! Because today, we’re diving headfirst into the magical world of the “Diffusion And Osmosis Beaker Worksheet Answer Key,” and let me tell you, it’s not as scary as it sounds. In fact, it’s more like unlocking the cheat codes to a super fun, microscopic game!
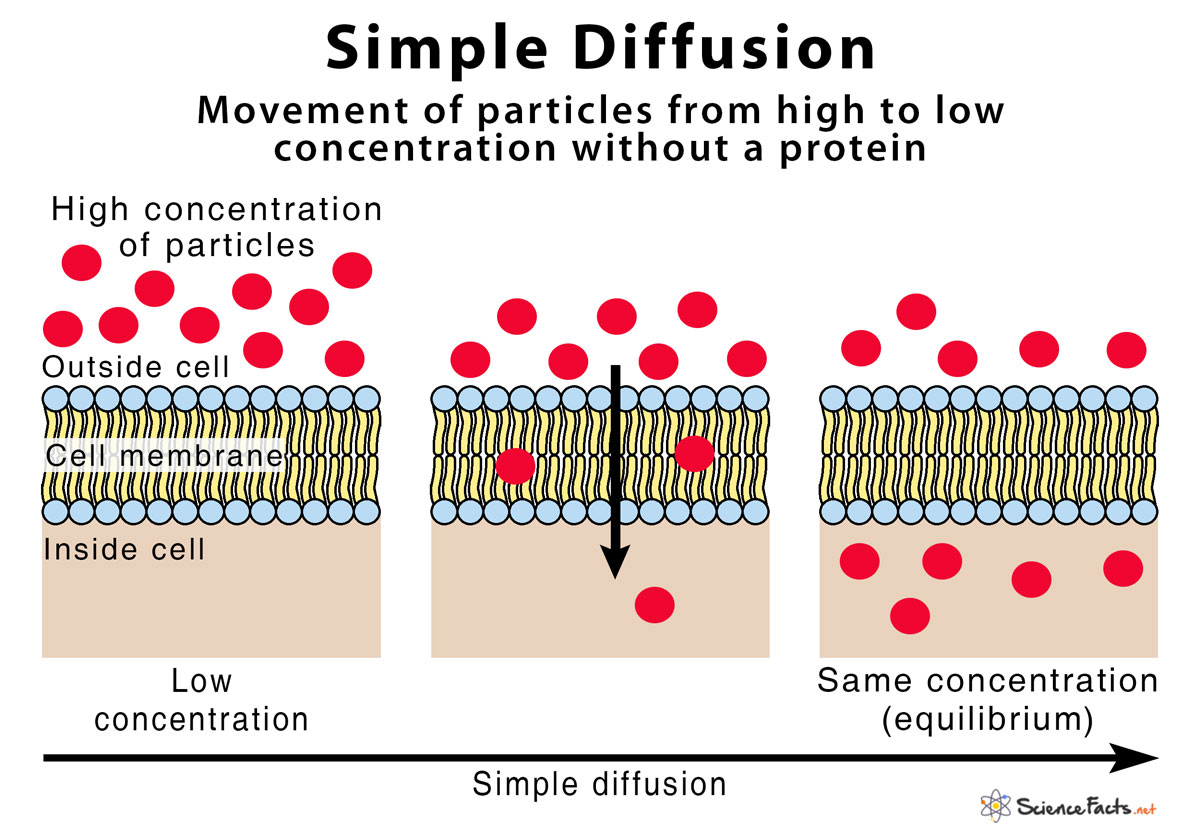
Imagine your favorite comfy couch. Now, imagine someone spilled a tiny drop of super-concentrated, extra-delicious chocolate syrup right in the middle of it. What happens? Even if you don’t touch it, that rich, chocolatey goodness starts to spread out, right? Slowly but surely, it moves towards the edges of the couch, making everything… well, more chocolaty. That, my friends, is the essence of diffusion! It’s basically the universe’s way of saying, “Hey, let’s even things out!” Molecules, like our yummy chocolate syrup, just can’t help but spread from where they are super crowded to where there are fewer of them. Think of a packed concert crowd slowly dispersing into the open street – that’s diffusion in action, no ticket required!
Now, let’s add a twist to our couch scenario. What if that chocolate syrup was separated from the rest of the couch by a super-special, invisible fence? This fence is like a bouncer at a club, letting some things through but not others. This is where osmosis waltzes in. Osmosis is a special kind of diffusion, but it’s all about water. And this invisible fence? In science terms, we call it a semipermeable membrane. It’s like a sieve that’s really picky about what it lets pass. So, if our chocolate syrup is on one side of this picky fence, and plain old water is on the other, osmosis will kick in. The water molecules, wanting to dilute that super-concentrated chocolate situation and make things less… intense, will sneak through the semipermeable membrane to the side with the chocolate. They’re trying to even out the concentration, just like diffusion, but they’re doing it by moving themselves (the water!), not the solute (the chocolate!).
So, when you’re looking at your “Diffusion And Osmosis Beaker Worksheet Answer Key,” picture these little scenarios. The worksheet might show you a beaker with a U-shaped tube, divided by something that looks suspiciously like our picky fence. On one side, you’ve got a high concentration of something – let’s say, a ridiculously strong fruit punch. On the other side, you’ve got plain water. The answer key will reveal how the water molecules will bravely march across that semipermeable membrane, leaving the fruit punch molecules to grumble about being left behind. They’re just trying to achieve that perfect, balanced, slightly less-sweet-on-one-side beverage!
Think about your own body. Your cells are tiny little factories, and they’re surrounded by these amazing semipermeable membranes. Osmosis is happening all the time to keep your cells happy and hydrated. When you drink water, osmosis helps your body absorb it. When you eat salty snacks (and your body gets a bit concentrated with salt!), osmosis helps your body move water around to dilute that saltiness. It's like a constant, tiny water parade keeping everything running smoothly. Without this amazing process, your cells would either shrivel up like raisins or explode like overfilled water balloons. Nobody wants that!
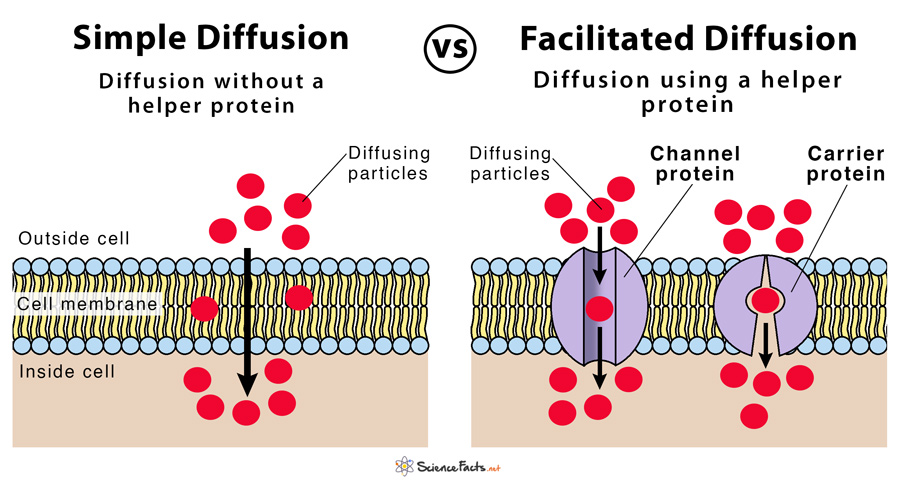
And diffusion? It’s responsible for all sorts of everyday magic. Ever walked into a bakery and instantly smelled those warm, sugary cookies? That’s diffusion! The delicious cookie molecules are spreading through the air from the bakery to your nose. Or when you put a tea bag in hot water, and the color and flavor start to spread out? Diffusion again! It’s the universe’s way of sharing the good stuff. So, those worksheets with beakers and arrows? They're just visual representations of these incredible, invisible processes that are happening all around us and inside us, every single second.
So, when you’re tackling that Diffusion And Osmosis Beaker Worksheet Answer Key, don’t get intimidated by the fancy words. Just remember the chocolate syrup on the couch, the picky fence, and the brave water molecules on a mission. You’ve got this!
The answer key isn't about catching you out; it's about guiding you to understand how these fundamental biological processes work. It’s like having a friendly guide pointing out the hidden wonders of the microscopic world. So, go forth, conquer those worksheets, and impress yourself with how much you’ve learned about the silent, steady, and utterly essential work of diffusion and osmosis. You’re basically becoming a mini-expert in the secret lives of molecules!
