Diels Alder Reaction Of Furan And Maleic Anhydride
Hey there, fellow explorers of the delightfully complex! Ever feel like your everyday life could use a little… oomph? A touch of magic? Well, what if I told you that a little bit of kitchen chemistry, a dash of everyday ingredients, and a whole lot of ingenuity could unlock some seriously cool reactions? Today, we're diving headfirst into the wonderfully chill world of the Diels-Alder reaction, specifically the one between furan and maleic anhydride. Think of it as a molecular handshake that’s way more exciting than your average "how do you do?"
You might be thinking, "Diels-Alder? Sounds a bit… intimidating." And I get it. The name itself has a certain academic weight to it. But trust me, when you break it down, it's really just about molecules finding their perfect partners. It’s like the universe’s very own dating app for atoms, where the right conditions bring the right compounds together for a beautiful, transformative union.
The Star Players: Furan and Maleic Anhydride
So, who are these VIPs of our molecular soirée? First up, we have furan. Picture this: it’s a five-membered ring, kinda like a tiny, slightly lopsided pentagon, with one oxygen atom sniffing around in there. It's found in all sorts of places, from the aroma of toasted bread (seriously, the smell of freshly baked sourdough has furan to thank!) to some essential oils. It’s a pretty common and agreeable molecule, always ready to mingle.
And then there’s maleic anhydride. This one’s a bit more… zesty. It’s also a ring, but this time it’s a six-membered one with a double bond that's got a bit of a strained personality. Imagine a square with a smiley face, but the smile is a little bit puckered. This slight tension makes it a fantastic participant in reactions. It’s a chemical workhorse, used in the production of plastics, resins, and even some food additives (though we’re not cooking with it today, thankfully!).
The Big Meet-Cute: How They Get Together
The Diels-Alder reaction is essentially a [4+2] cycloaddition. Don't let the numbers scare you! It just means we have a molecule with four pi electrons (that's furan in its aromatic dance) and another with two pi electrons (our eager maleic anhydride). They come together, no fuss, no muss, and form a brand new six-membered ring. It’s a concerted reaction, which is a fancy way of saying it all happens in one smooth step. Think of it like a perfectly choreographed dance move – everything shifts and rearranges simultaneously, creating something entirely new.
This isn't just some random bumping into each other. Furan, with its aromatic stability, actually has a less electron-rich pi system compared to many other dienes. Maleic anhydride, on the other hand, is what we call a dienophile – it’s "eager to be a diene's partner." The electron-withdrawing groups on the maleic anhydride make it particularly attractive to the electron-rich pi system of the furan. It's a classic case of opposites attracting, with a molecular twist!
What's really neat is that this reaction often proceeds without the need for harsh catalysts or extreme temperatures. You can often get a good yield just by mixing them and maybe applying a little gentle heat. It’s the kind of reaction that makes you feel like a chemistry wizard, even if you’re just in your garage with some beakers. It’s like finding a shortcut to a delicious recipe – efficient and rewarding!

Why We Care (Besides the Cool Factor)
Okay, so it’s a cool reaction, but what’s the big deal? Well, this reaction is a cornerstone of organic synthesis. It's incredibly versatile and allows chemists to build complex molecules with high precision. Think about it: being able to reliably create cyclic structures opens up pathways to all sorts of interesting compounds, including many pharmaceuticals, natural products, and advanced materials. It's like having a Lego brick that can snap together in very specific ways to build intricate structures.
The product of the furan and maleic anhydride reaction is a bicyclic compound. This means it has two rings fused together. It’s a bit like a molecular hug, with the two original rings now sharing some of their atoms. This new structure is called an adduct, and it’s a valuable intermediate for further chemical transformations. You can imagine it as a finished piece of furniture, ready for the next step of decorating or being incorporated into a larger design.
A Little Help From Our Friends: Solvents and Temperature
While the Diels-Alder reaction can be quite forgiving, a little bit of thoughtful planning can elevate your results. The choice of solvent can play a role. For furan and maleic anhydride, common solvents include things like ethyl acetate, toluene, or even just letting the reaction happen neat (without a solvent), especially if you’re heating it. Different solvents can influence the reaction rate and the solubility of your reactants and products.
Temperature is another key player. As mentioned, a gentle warmth is often all that's needed. Too much heat, however, can sometimes lead to side reactions or even cause your product to undergo a reverse Diels-Alder reaction, where it breaks back apart! It’s a delicate balance, like finding the perfect temperature for a perfectly brewed cup of tea. You want it warm enough to be enjoyable, but not so hot that it burns.
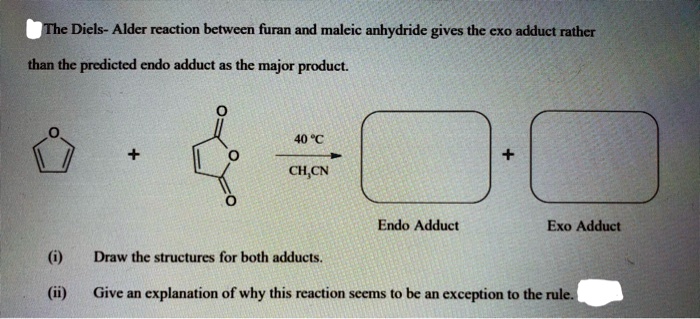
The "endo" versus "exo" selectivity of the Diels-Alder reaction is also a fascinating detail. In the case of furan and maleic anhydride, the reaction typically favors the endo product. This means the anhydride part of the maleic anhydride ends up pointing under the newly formed ring. It's a bit like how you might naturally orient a book on a shelf – there's a preferred, more stable position. This regiochemistry (where things end up) and stereochemistry (the 3D arrangement) are crucial in building complex molecules, and the Diels-Alder reaction provides a predictable way to achieve them.
Practical Tips for Your Own Molecular Adventure
So, how can you get your hands dirty with this? While I wouldn't recommend trying this in your kitchen without proper safety precautions and a strong understanding of chemistry, the principles are quite accessible. If you have access to a chemistry lab or are involved in a chemistry club, this is a fantastic introductory reaction.
Safety First! Always wear appropriate personal protective equipment, like gloves and safety glasses. Work in a well-ventilated area or under a fume hood. Handle chemicals with respect and know their properties. This isn't a TikTok challenge; it's science!
Ingredient Sourcing: Furan can be a bit trickier to source for home enthusiasts, but it's a common lab reagent. Maleic anhydride is more readily available. If you're interested in exploring related Diels-Alder reactions, look for other conjugated dienes and dienophiles. Many are much easier to obtain and work with.

The Setup: Typically, you'd dissolve your reactants in a suitable solvent in a flask. You might then heat the mixture using a hot plate or a water bath. Stirring is important to ensure good mixing. The reaction can take anywhere from a few minutes to a few hours, depending on the specific conditions.
Isolation and Purification: Once the reaction is complete, you'll need to isolate your product. This might involve cooling the mixture to allow the product to crystallize, followed by filtration. Purification techniques like recrystallization can be used to get a pure sample. Observing the melting point of your solid product is a good way to assess its purity. Is it sharp and consistent, like a perfectly cut diamond, or all over the place, like a Jackson Pollock painting?
Cultural Connections and Fun Facts
The Diels-Alder reaction is so fundamental that it even has its own Nobel Prize! Otto Diels and Kurt Alder were awarded the Nobel Prize in Chemistry in 1950 for their work on this very reaction. It’s a testament to how significant this molecular dance has been for the advancement of chemistry.
Think about the impact! From the life-saving drugs we take to the durable plastics that make up so many of our everyday objects, the Diels-Alder reaction has played a role. It’s a quiet hero in the background of modern life. It's like discovering that your favorite song was written by someone who also invented a really comfy chair – a double win!
And here’s a fun thought: imagine if your favorite fictional characters were chemists. Would Sherlock Holmes use the Diels-Alder reaction to synthesize a rare poison or an antidote? Would Hermione Granger whip up a batch to create a new potion ingredient? The possibilities are as endless as the molecules themselves!
A Daily Dose of Molecular Inspiration
So, what’s the takeaway from this deep dive into furan and maleic anhydride’s love story? It’s a reminder that even in the seemingly mundane, there’s incredible complexity and beauty. The world around us, down to the molecular level, is constantly in motion, forming new bonds, and creating new possibilities.
Just like the Diels-Alder reaction brings together seemingly simple molecules to create something more intricate and useful, our own lives are a series of reactions. We meet new people, we learn new skills, we combine experiences, and we transform. Every interaction, every lesson, every moment is a chance to build something new, something better, something uniquely us.
So, the next time you’re enjoying that toasty bread or using a plastic item, take a moment to appreciate the invisible world of chemistry. And remember, with a little understanding and the right conditions, even the most complex-looking processes can lead to something truly remarkable. It’s all about finding your perfect reaction!
