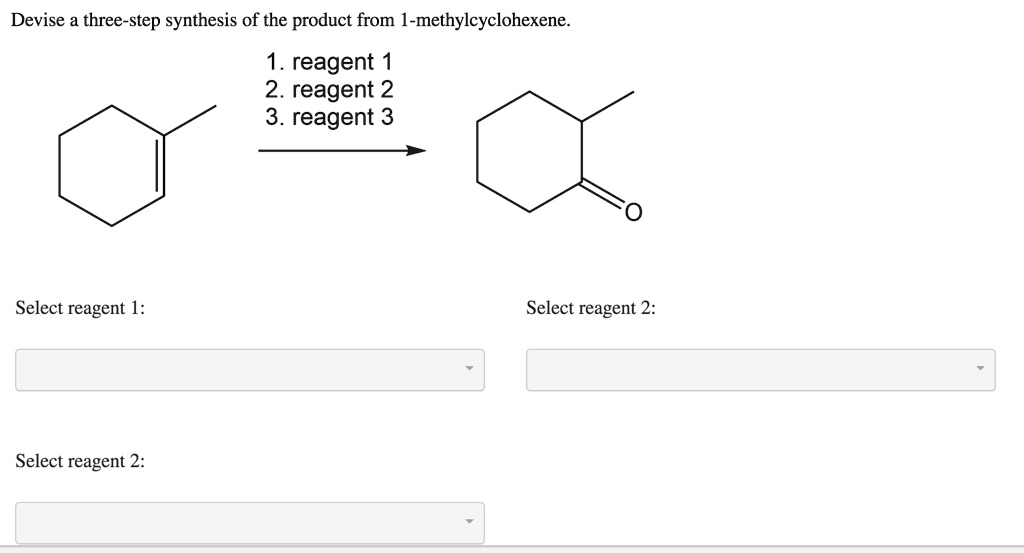
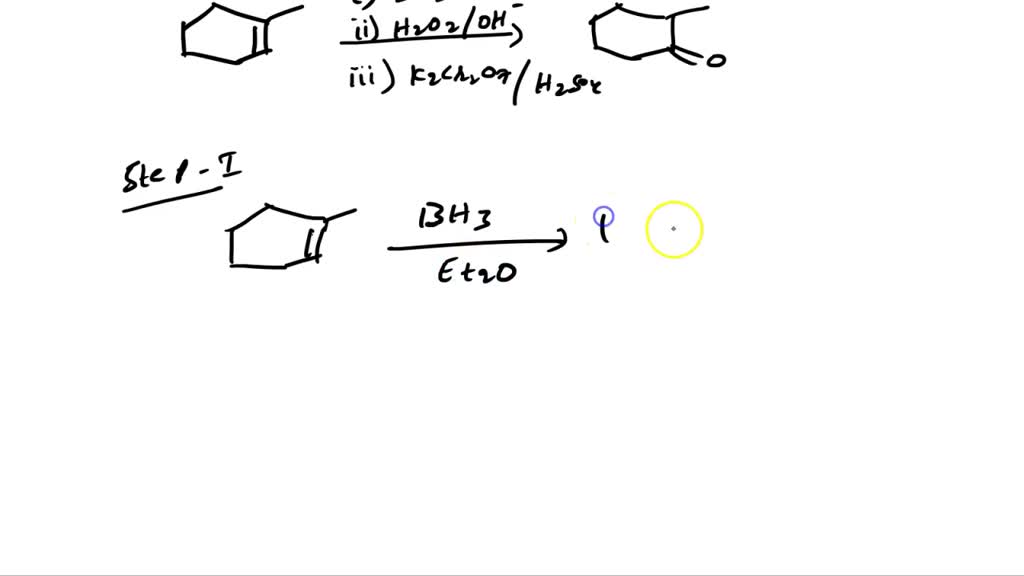
Devise A Three-step Synthesis Of The Product From 1-methylcyclohexene
Ever looked at a complex chemical reaction and thought, "That looks like a masterpiece waiting to happen!"? Well, while the world of organic synthesis might seem daunting, sometimes the most fascinating transformations are born from surprisingly simple beginnings. Today, we’re diving into the creative potential of turning 1-methylcyclohexene into something new and exciting, using a straightforward three-step synthesis. Think of it as unlocking a hidden artistic talent within a humble molecule!
Why should you care about this particular molecular makeover? For the artists among us, this process can be a gateway to understanding the building blocks of new materials, perhaps inspiring unique textures or pigments. Hobbyists who enjoy tinkering and problem-solving will find a satisfying challenge in tracing the molecular journey. And for the casual learner, it’s a chance to demystify a slice of chemistry, revealing that even intricate processes can be broken down into manageable, logical steps. It’s about fostering a sense of curiosity and accomplishment.
So, what can we create? While the possibilities are vast, let’s consider a common and versatile outcome: a functionalized cyclohexane derivative. Imagine using this as a starting point for creating scented compounds – think of the delicate fragrances of lavender or mint. Or perhaps you could envision it as a precursor for creating specialized polymers with unique properties. The beauty lies in the versatility; the final product can be a stepping stone to a whole spectrum of applications, from pharmaceuticals to fine chemicals.
Let’s peek at the three-step journey. First, we might begin with an oxidation reaction. This is like carefully peeling back layers or adding new features. A common approach could involve using something like m-CPBA (meta-chloroperoxybenzoic acid) to form an epoxide. This epoxide is a highly reactive intermediate, much like a blank canvas ready for further artistic intervention. Next, we’d look at ring-opening the epoxide. This is where we introduce a new element. Depending on the reagent we choose, we can attach different groups to our molecule. For instance, using an acid and water could lead to the formation of a diol – two hydroxyl groups attached to the cyclohexane ring. Finally, we might undergo a further functionalization step. This could involve converting one of the hydroxyl groups into something else entirely, like a halide, an ester, or even a ketone. Each choice opens up new avenues for creativity.
Ready to try this at home, metaphorically speaking? The key is patience and understanding. Don't rush the process. Visualize each step: what is happening to the molecule? What new bonds are forming? What bonds are breaking? Focus on the underlying principles rather than just memorizing reagents. Start with simpler examples in textbooks or online tutorials before attempting more complex transformations. And always remember that in a real laboratory setting, safety is paramount.
What makes this exploration so enjoyable is the inherent elegance of chemical transformations. It’s like watching a sculptor chip away at stone to reveal a hidden form, or a painter layering colors to create depth. The three-step synthesis of a product from 1-methylcyclohexene offers a tangible example of how thoughtful manipulation of matter can lead to diverse and useful outcomes. It’s a journey of discovery, a testament to the power of constructive change, and a deeply satisfying pursuit for anyone with a curious mind.
