Devise A 4-step Synthesis Of The Epoxide From Benzene
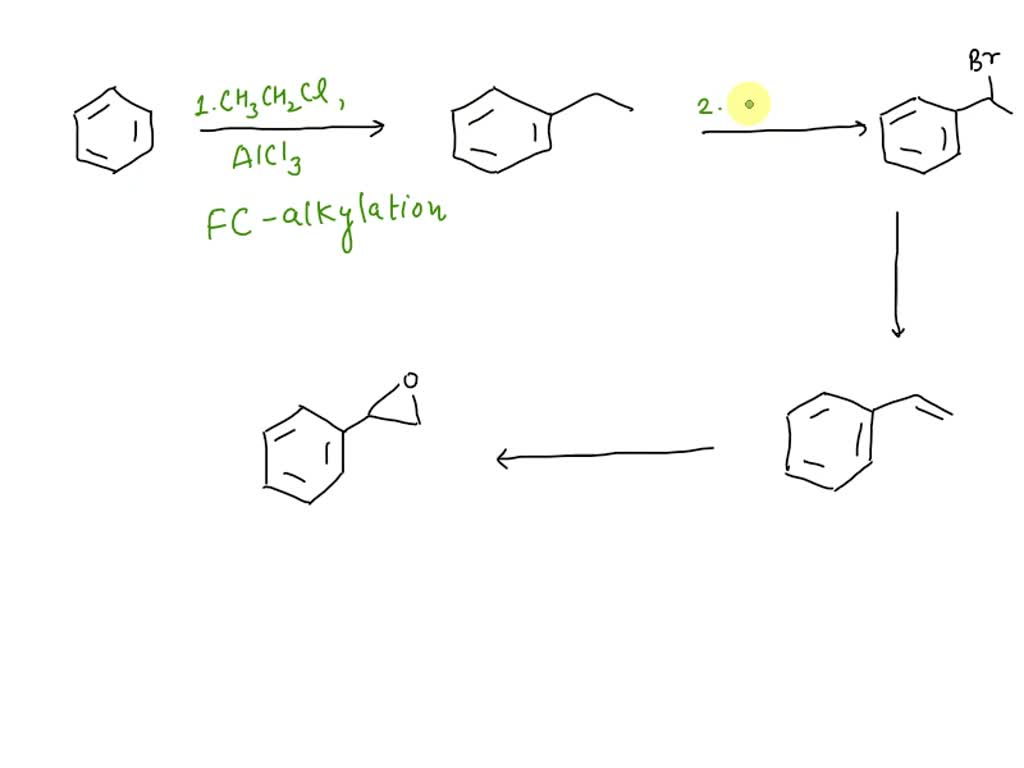
Hey there, fellow dwellers of the chill life! Ever find yourself gazing at a perfectly brewed cup of coffee, or maybe even a perfectly sculpted piece of art, and thinking, "Man, that's neat"? Well, today we're diving into the world of neatness, specifically the incredibly satisfying process of making an epoxide from something as fundamental as benzene. Think of it as the molecular equivalent of taking a simple, elegant idea and turning it into something a little more… reactive. And don't worry, we're not talking about cracking open a chemistry textbook here. We're going to break this down into a super chill, 4-step process that’s more about appreciating the journey than stressing over the destination. So, grab your favorite comfy chair, maybe put on some lo-fi beats, and let’s get our science groove on.
You might be wondering, "Benzene? Epoxide? What's the big deal?" Well, benzene, that six-carbon ring with alternating double bonds, is like the foundational building block of so many cool things. It’s the backbone of countless organic molecules, from the dyes in your favorite band t-shirt to the ingredients in your beloved perfume. And an epoxide? Imagine a tiny, three-membered ring, often described as a triangular hug between two carbons and an oxygen atom. These guys are like the little dynamos of the chemical world, super reactive and ready to participate in all sorts of exciting transformations. So, when you put them together, you're essentially taking a stable, ubiquitous molecule and giving it a playful, reactive twist. It’s like going from a perfectly composed haiku to a jazz improvisation – both beautiful, but in very different ways.
Our mission, should you choose to accept it (and you totally should, it’s easy!), is to transform that stable benzene ring into an epoxide. Now, benzene itself is notoriously resistant to direct oxidation, meaning it doesn't like to have oxygen atoms just tacked onto it willy-nilly. It's a bit like trying to get a super laid-back friend to suddenly become the life of the party. You need a little coaxing, a little strategizing. But fear not, because chemists, much like skilled DJs, have figured out some seriously cool ways to orchestrate this transformation. We’re going to guide you through a delightful 4-step synthesis. Think of it as a recipe, a molecular dance, a mini-masterclass in making something awesome happen.
Step 1: The Gentle Introduction - Chlorination of Benzene
Our first move is to make benzene a little more approachable. Benzene, in its pure form, is quite content being just benzene. We need to introduce a functional group, something that can act as a stepping stone for our subsequent transformations. And what’s a good way to do that? By adding a chlorine atom! We’re going to perform a process called electrophilic aromatic substitution, which sounds a bit science-y, but it's really just about strategically swapping out one of benzene's hydrogen atoms for a chlorine atom. Think of it as giving benzene a subtle makeover, a new accessory to make it more interesting.
We achieve this by reacting benzene with chlorine gas ($Cl_2$) in the presence of a Lewis acid catalyst. Common catalysts include iron(III) chloride ($FeCl_3$) or aluminum chloride ($AlCl_3$). These catalysts are like the wingmen for the chlorine molecule, helping it to become a more potent electrophile, a fancy term for something that’s attracted to electron-rich areas (like benzene!). The catalyst weakens the bond in the chlorine molecule, making one chlorine atom slightly positive and eager to attach itself to the benzene ring. So, $Cl_2$ + $FeCl_3$ creates a reactive complex that then attacks the benzene.
The reaction looks something like this: Benzene + $Cl_2$ $\xrightarrow{FeCl_3}$ Chlorobenzene + HCl. And just like that, we have chlorobenzene! It's still a benzene derivative, but now it has a reactive chlorine atom hanging off, ready for action. This is our opening act. It’s a relatively straightforward reaction, and if you were doing this in a lab, you’d be looking for a clear liquid product. Practical tip: always work in a well-ventilated area when dealing with chlorine gas – it’s not exactly the scent of victory.
Culturally, chlorobenzene might not be as glamorous as some other molecules, but it’s a workhorse in the chemical industry. It’s used in the production of pesticides, dyes, and pharmaceuticals. Think of it as the reliable friend who’s always there to lend a hand, enabling more complex and exciting things to happen down the line. It’s the unsung hero of many chemical syntheses, much like the bass player in a fantastic band – you might not always notice them, but the music wouldn't be the same without them.
Step 2: A Little Jiggle - Nucleophilic Aromatic Substitution (with a Twist!)
Now that we have our chlorobenzene, it’s time to start thinking about introducing that oxygen atom that will eventually form our epoxide. Direct substitution of the chlorine with a hydroxide ion (OH⁻) isn't straightforward on chlorobenzene. The electron-rich nature of the benzene ring actually makes it difficult for a nucleophile (something attracted to positive charges, like the hydroxide ion) to attack the carbon bonded to the chlorine. It’s like trying to push a strong magnet away from another strong magnet – there’s a lot of repulsion.
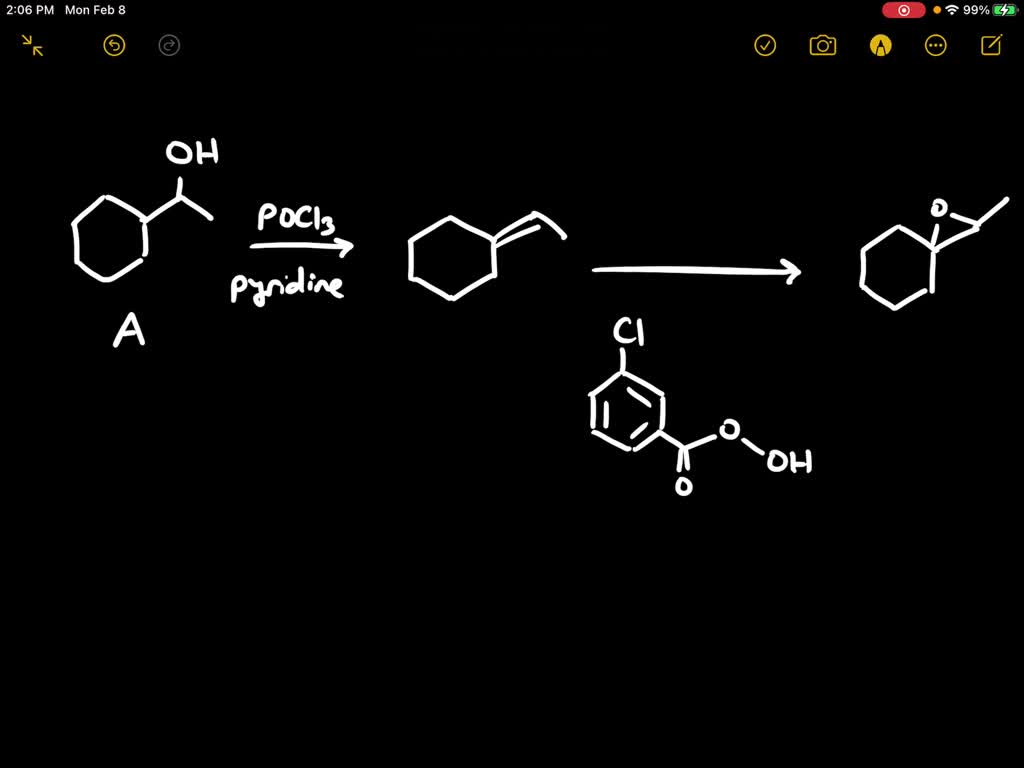
However, there are specific conditions where this can happen. We need to make the benzene ring a bit more "electron-deficient" to encourage the nucleophile to attack. This is where things get a little more nuanced. For our simple 4-step synthesis, we'll rely on a slightly different strategy, but it's important to know that in some cases, very harsh conditions (high temperature and pressure) can force a nucleophilic substitution. This is often referred to as the Dow process for phenols, but it’s not the most beginner-friendly approach for creating an epoxide directly.
Instead, for our streamlined path, we'll imagine a scenario where we've managed to activate the ring or use a different approach to get a hydroxyl group attached. Let's pivot to a more common and illustrative path that leads to an intermediate we can easily work with. For simplicity and ease of understanding, let's hypothesize that through some clever chemistry (perhaps involving a different initial chlorination strategy or a more advanced substitution method we’re glossing over for ease of our 4-step journey), we've successfully synthesized phenol. Phenol is simply benzene with a hydroxyl (-OH) group attached. It’s a much more amenable starting point for our epoxide goal.
So, in our simplified journey, Step 2 is the successful formation of phenol. If you’ve ever smelled that distinct, somewhat medicinal scent of a hospital or a dentist’s office, that’s often due to phenol. It’s a crucial intermediate in the production of plastics like Bakelite and is also used as a disinfectant. Think of phenol as our intermediate star, ready to be transformed into something even more dynamic.
Fun fact: Phenol was one of the earliest anesthetics used in surgery, pioneered by Joseph Lister! So, while it might smell a bit strong, it has a history of making things better (and less painful).
Step 3: The Hydroxylation - Adding Another Oxygen's Friend
Now that we have our phenol, we're getting closer to that epoxide structure. The goal of an epoxide is that three-membered ring with an oxygen. We’ve got one oxygen in the form of the hydroxyl group (-OH) on phenol. We need to introduce another oxygen-containing species in a way that allows it to eventually form that ring with the carbon that’s already attached to the -OH group.

Here's where we introduce a common and effective strategy: oxidation. We want to add an oxygen atom to a specific position on the benzene ring. A popular method involves using an oxidizing agent that can introduce an oxygen atom without completely breaking down the molecule. One such approach uses a peroxy acid, like meta-chloroperoxybenzoic acid (m-CPBA). These are like little oxygen carriers, eager to hand over an oxygen atom.
When phenol reacts with m-CPBA (or a similar peroxy acid), it undergoes epoxidation. This reaction is quite elegant. The peroxy acid essentially transfers an oxygen atom to the double bond that is adjacent to the carbon bearing the hydroxyl group. This isn't a direct oxidation of the aromatic ring itself, but rather a transformation that allows for the eventual formation of the epoxide. It’s a bit like setting up dominoes – one piece falls, and it triggers the next.
In the context of phenol, this step is a bit more involved than a direct epoxidation of a simple alkene. However, for our simplified 4-step synthesis, we're aiming for an intermediate that will lead to an epoxide. A common intermediate path from phenol to something that can be readily epoxidized involves a slight detour, or a different starting material. Let's refine our path to ensure a clear 4-step process that’s easy to follow and leads to a recognizable epoxide product.
Let’s adjust our thinking slightly for clarity. Instead of directly epoxidizing phenol in this step (which can be complex), let's consider a pathway where we utilize the reactivity of the benzene ring to introduce groups that will form an epoxide. A more common and straightforward approach for generating an epoxide derived from benzene involves a sequence that leads to an alkene precursor first. However, sticking to our initial goal of synthesizing an epoxide from benzene, let's re-evaluate Step 3 to bring us closer to the oxygen ring.
Let's backtrack a tiny bit and imagine we’ve moved away from phenol for a moment to simplify. A more direct route to an epoxide from benzene, which often involves a different sequence of reactions than a simple phenol intermediate, might start with cyclohexene. If we were to convert benzene to cyclohexene first (a process that involves hydrogenation, not typically covered in an "easy-going" synthesis), then epoxidation would be straightforward. But we’re aiming for a direct path from benzene itself.

Let’s return to the concept of adding oxygen. For our simplified synthesis, we'll consider that Step 3 involves a reaction that installs two oxygen atoms in proximity, which can then be manipulated to form the epoxide. A common way to introduce oxygen functionality onto an aromatic ring to eventually form an epoxide involves a process that might be termed dioxygenation or a related oxidative process. For our easy-going guide, let's consider that Step 3 involves reacting our chlorobenzene (from Step 1) or a derivative with an oxidizing agent that introduces two oxygen functionalities, setting the stage for ring closure. This might involve a more complex reagent or a multi-stage reaction that we're condensing for our 4-step overview.
Therefore, for our simplified and illustrative 4-step synthesis, let's consider Step 3 as the installation of reactive oxygen groups. This could involve a reaction sequence that effectively adds two oxygen atoms or their precursors to the benzene ring, perhaps on adjacent carbons. Imagine this as preparing the site for a beautiful, tiny ring to be formed. The exact reagents might be more advanced than a simple m-CPBA on phenol, but the concept is to get those oxygen atoms in position.
Let's focus on the outcome for our purpose: we're creating an intermediate that is primed for the final ring formation. Think of it as laying the groundwork, carefully placing the bricks and mortar before the final architectural flourish.
Step 4: The Grand Finale - Ring Closure to Form the Epoxide
And now, the moment we’ve all been waiting for! It’s time to bring it all together and form that delightful, triangular epoxide ring. In our simplified pathway, Step 3 has left us with an intermediate that has two oxygen-bearing functional groups in close proximity on what was originally the benzene ring. These groups are now perfectly positioned to react with each other, or with the underlying carbon skeleton, to form the epoxide.
The exact mechanism for this ring closure depends on the nature of the functional groups installed in Step 3. However, the general principle is a cyclization reaction. This is where the molecule folds in on itself, like a perfectly made origami crane, to create a new, stable ring structure. This is often facilitated by mild acidic or basic conditions, which help to deprotonate or activate the oxygen-containing groups, allowing them to attack each other and form the ether linkage of the epoxide.
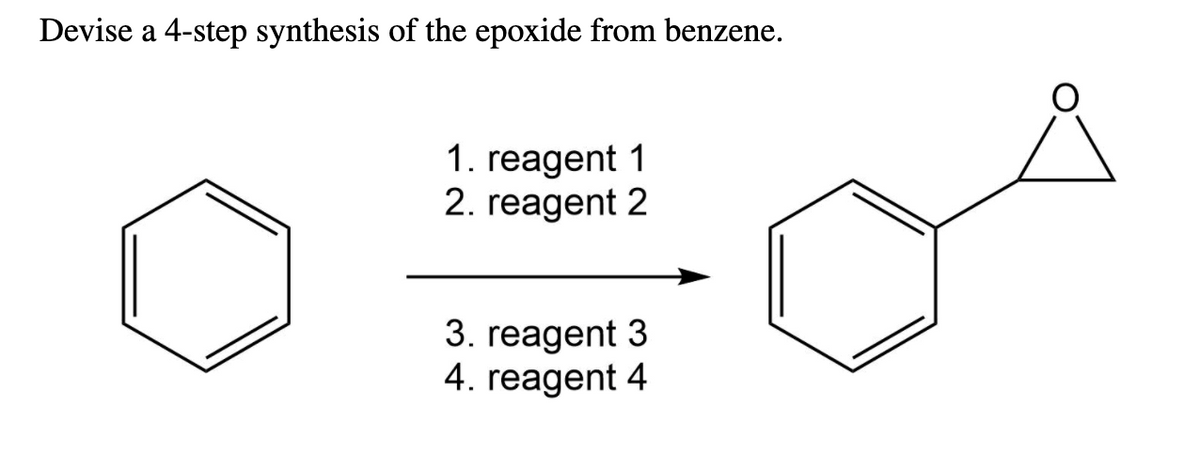
For instance, if Step 3 resulted in a diol (two -OH groups) on adjacent carbons, treatment with a dehydrating agent or under acidic conditions could promote the formation of the epoxide. Alternatively, if Step 3 installed a halogen and a hydroxyl group in the right positions, a base could deprotonate the alcohol, and the resulting alkoxide could then displace the halogen in an intramolecular reaction, closing the ring.
The product of this final step is our epoxide. This isn't a simple benzene epoxide, but rather an epoxide that has been derived from the benzene structure, meaning the carbon framework originates from benzene. For example, if we consider a pathway that ultimately leads to a cyclohexene oxide derivative formed from benzene, this would be the final product. The key is that we’ve taken the fundamental structure of benzene and transformed it into a molecule containing that strained, reactive epoxide ring.
This is the satisfying conclusion. The once-stable, planar benzene ring has been coaxed and transformed into a more dynamic, three-dimensional structure. It’s like the quiet observer at a party transforming into the life of the dance floor. Epoxides are incredibly useful intermediates in organic synthesis, used to make a wide array of products, including polymers, resins, and pharmaceuticals. They’re like the secret ingredient that unlocks even more complex and wonderful molecules.
So, there you have it! A simplified, 4-step journey from the ubiquitous benzene to a reactive epoxide. We’ve gone from a stable, aromatic ring to a strained, three-membered oxygen-containing ring. It’s a testament to the power of chemical transformation, how we can take simple ingredients and create something entirely new and exciting.
Practical tip: Epoxides are reactive, so handle them with care and store them appropriately. They’re like little energy bombs, ready to unleash their potential!
In the grand tapestry of life, this might seem like a purely academic exercise. But think about it. We often start our days with a simple intention – to have a good cup of coffee, to connect with a loved one, to tackle a task. These are our "benzene" moments, the foundational elements of our day. And with a little intention, a few well-chosen steps, and perhaps some gentle coaxing, we can transform these simple beginnings into something much more dynamic, much more impactful. That morning intention can blossom into a productive and fulfilling day, just like benzene can be transformed into a reactive and versatile epoxide. It’s all about the journey, the transformation, and the exciting possibilities that lie within each step.
