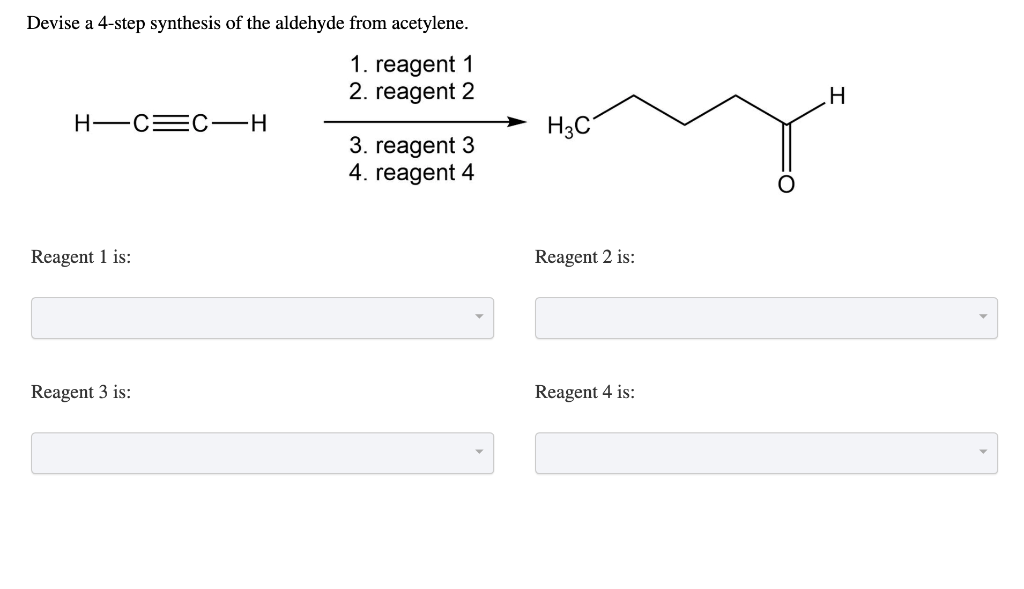
Devise A 4 Step Synthesis Of The Aldehyde From Acetylene
Hey there, curious minds! Ever wonder how some of the amazing smells we love – like freshly cut grass or that lovely hint of vanilla – come to be? Or perhaps you've heard of something called "acetylene" and thought, "Is that like, a fancy welding gas?" Well, today, we're going to dive into a little bit of chemistry magic, but don't worry, it's going to be more like a fun kitchen experiment than a stuffy lecture. We're going to explore a super neat way to make a type of molecule called an aldehyde starting from that very same acetylene. And trust me, it's way cooler than it sounds!
So, why should you, the everyday awesome person, care about making aldehydes from acetylene? Think of it this way: aldehydes are like the building blocks for so many things we encounter daily. They're the reason why perfumes smell so delightful, why some foods have that distinctive aroma (think of that yummy roast chicken smell!), and they even play a role in how some medicines are made. They're practically the unsung heroes of our sensory world. And learning how they're made from something as simple as acetylene is like unlocking a secret recipe for a lot of the good stuff around us.
Let's talk about our starting material: acetylene. You might have seen it used by welders, making those sparks fly. It's a gas, and its chemical formula is C₂H₂. Imagine two carbon atoms holding hands really, really tightly, and then each of them is also holding hands with a hydrogen atom. They're pretty enthusiastic about sticking together! Acetylene is a simple molecule, but it's packed with energy, which is why it's so useful.
Now, our goal is to turn this energetic gas into an aldehyde. What's an aldehyde? It's a molecule with a special little group of atoms attached, often described as a "carbonyl group" bonded to a hydrogen atom. Think of it as a little "O" double-bonded to a carbon atom, and that carbon atom is also attached to a hydrogen atom. This little "CHO" group is what gives aldehydes their unique properties and their often pleasant smells. For instance, acetaldehyde (which we'll be hinting at making!) is a simple aldehyde that smells a bit like green apples. Yum!
So, how do we get from our "welding gas" acetylene to these fragrant aldehydes? We're going to do it in four simple, elegant steps. Think of it like a well-choreographed dance, where each step leads smoothly to the next, resulting in a beautiful final product.
Step 1: Get the Party Started with a Little Help from Water!
Our first move is to introduce our acetylene to something familiar: water (H₂O). But we can't just toss them together like a casual meet-up. We need a little nudge, a facilitator, if you will. Imagine you're trying to get two shy friends to talk to each other. You might introduce them or break the ice with a shared activity. In our case, the facilitator is an acid, and to be specific, we often use something like sulfuric acid (H₂SO₄). And to make sure things move along at a good pace, we often add a touch of mercury salt, like mercury(II) sulfate (HgSO₄).
Now, this might sound a bit industrial, but think of it this way: the acid is like the encouraging friend, and the mercury salt is like the fun icebreaker. Together, they help the acetylene and water get really friendly. What happens is that the water molecule starts to "add" itself across the triple bond in acetylene. It's like the water is finding a place to slot in and make things a bit more stable. This first step creates a molecule called enol. The enol molecule is a bit like a chameleon; it has the potential to change its form.
Imagine acetylene as a tightly wound spring. Water, with the help of our acid and mercury catalyst, comes along and helps to uncoil that spring, but not all the way. It creates a temporary, wobbly structure. This wobbly structure, the enol, is crucial because it's ready for the next transformation.
Step 2: The Great Enol-to-Keto Tussle!
Our enol molecule, while interesting, isn't quite the aldehyde we're after. It's like having a lump of dough – it's got potential, but it needs shaping. This is where the second step, a process called tautomerization, comes in. It's a fancy word for a molecule rearranging itself. In our enol's case, it's going to shift its atoms around to become more stable. Think of it like a dancer doing a quick, elegant spin to land in a much better pose.
The enol form has a double bond between two carbon atoms and a hydroxyl group (-OH) attached. The keto form, which is what we're aiming for in this step, has a double bond between a carbon atom and an oxygen atom (our carbonyl group!) and the hydrogen atom has moved to the carbon that was part of the original double bond. It’s a subtle but significant change.
This transformation is usually driven by the presence of the acid we used in step one. The acid helps to make this rearrangement happen easily. So, our wobbly enol molecule takes a deep breath, does a little internal jig, and transforms into a more stable form that's much closer to our aldehyde. It’s like a caterpillar transforming into a butterfly, but much, much faster!
This step is really important because the keto form is where the aldehyde magic really starts to happen. The enol is like a fleeting moment, and tautomerization helps us capture that potential into a more usable structure.
Step 3: Introducing Our Very Own "Hydration" Friend!
So far, we've got our acetylene, we've added water with some help, and we've rearranged things to get closer to our goal. Now, we need to refine our molecule a bit further. If we were making a cake, this step would be like adding the specific flavorings. We've already got the basic structure, but we want to ensure we get exactly the aldehyde we're looking for.
In some variations of this process, especially if we want to be really precise about making a specific aldehyde, we might introduce another water molecule, or in essence, perform a second "hydration" step. This is where things can get a little more specific depending on what kind of aldehyde we want to end up with. However, a very common and direct route from acetylene often leads us to a molecule called acetaldehyde (CH₃CHO) directly after the tautomerization. Acetaldehyde is a simple aldehyde, and it's the one we often get when we just follow steps 1 and 2 with the right conditions.
Think of it like this: step one and two are like adding flour and eggs to make a batter. This step is like deciding whether you want a vanilla cake or a chocolate cake. For acetylene, the most straightforward path after the enol-keto shift directly gives us acetaldehyde, which is a fantastic starting point for many other things, or a useful aldehyde on its own!
Step 4: Purification and Enjoyment!
We've done the hard chemical work! Our acetylene has been transformed into an aldehyde. But just like when you bake a cake, you often need to let it cool and maybe frost it before you can truly enjoy it. This final step is all about purification. We want to isolate our beautiful aldehyde from any leftover starting materials or byproducts. Think of it like sifting out the lumps from your cake batter or straining the pulp from fresh juice.
Chemists have all sorts of clever ways to do this. They might use techniques like distillation, which separates liquids based on their boiling points. Imagine heating a mixture, and the substance with the lower boiling point turns into vapor first, and you can collect that vapor separately. It’s like catching steam from a teapot.
Once purified, our aldehyde is ready! We can then use it for all sorts of wonderful applications. If we made acetaldehyde, it's a key ingredient in making acetic acid (the stuff that makes vinegar!), and it's also used in making plastics and other chemicals. It's the end of our journey, but the beginning of so many new possibilities!
So, there you have it! A four-step journey from the humble acetylene gas to the versatile and fragrant world of aldehydes. It’s a testament to how simple molecules can be transformed through clever chemistry into compounds that enrich our lives in countless ways. The next time you catch a whiff of a lovely scent or enjoy a tasty dish, you might just be experiencing the magic that started with a reaction very much like the one we've explored today. Pretty neat, right?
