Determining The Molar Mass Of Butane Lab Answers

Okay, so you've been in the lab. You've probably got some numbers staring back at you. And those numbers? They're all about butane. Specifically, you're trying to figure out its molar mass. Sounds kinda… science-y, right? But trust me, it's way more interesting than it sounds. Think of it like a treasure hunt, but the treasure is the true weight of a bunch of butane molecules.
Butane. What even is butane? It's that stuff in your lighter. Yeah, the little clicky thing you use to light candles. Or maybe it's the fuel for your camping stove. It's pretty common, actually. And figuring out its molar mass is like learning its secret identity. It tells us how much one mole of butane weighs. A mole, by the way, is just a huge number of things. Like, a ridiculous amount. So, the molar mass is the weight of that giant, unimaginable pile of butane molecules.
Why is this fun? Because science is a giant puzzle. And every piece you figure out is a little victory. Plus, you get to play with cool equipment. Bunsen burners? Check. Beakers? Check. Scales that go down to, like, super tiny amounts? Double check. It’s like being a detective, but your suspect is an invisible gas.
So, you've done the experiment. You've measured stuff. You've maybe watched some bubbles. Now comes the moment of truth: the calculation. This is where your lab answers come into play. You're not just plugging numbers into a formula; you're unlocking the secrets of butane. It’s like cracking a code. And the code is written in grams per mole. Pretty neat, huh?
Let's talk about the lab itself. What did you do? Did you heat up some butane? Did you measure the volume of gas it produced? Maybe you collected it over water. That’s a classic! It looks all fancy, with the gas bubbling up and displacing the water. It’s a bit like a science experiment from a retro movie. You probably felt pretty accomplished watching it happen. Admit it, you probably grinned a little.
The Nitty-Gritty: What’s Actually Happening?
So, what are we even calculating? Molar mass. It's literally the mass of one mole of a substance. And a mole? It's Avogadro's number. Yep, that guy again. It’s about 6.022 x 10^23. That’s a 6 followed by 23 zeros. Just imagine that many tiny butane molecules. It’s mind-boggling. And we want to know how much that massive pile weighs. Our lab is giving us the clues to figure that out.
For butane, the chemical formula is C4H10. This tells us that each molecule of butane has 4 carbon atoms and 10 hydrogen atoms. Simple, right? But then we need to know the atomic masses of carbon and hydrogen. You’ve probably got a periodic table handy. Carbon is roughly 12.01 grams per mole, and hydrogen is about 1.008 grams per mole. Easy peasy.
So, if you have 4 carbons, that’s 4 times 12.01. And if you have 10 hydrogens, that’s 10 times 1.008. Add those two numbers together, and BAM! You’ve got the theoretical molar mass of butane. This is your baseline, your perfect-world answer. But in the lab, things are rarely perfect, are they?
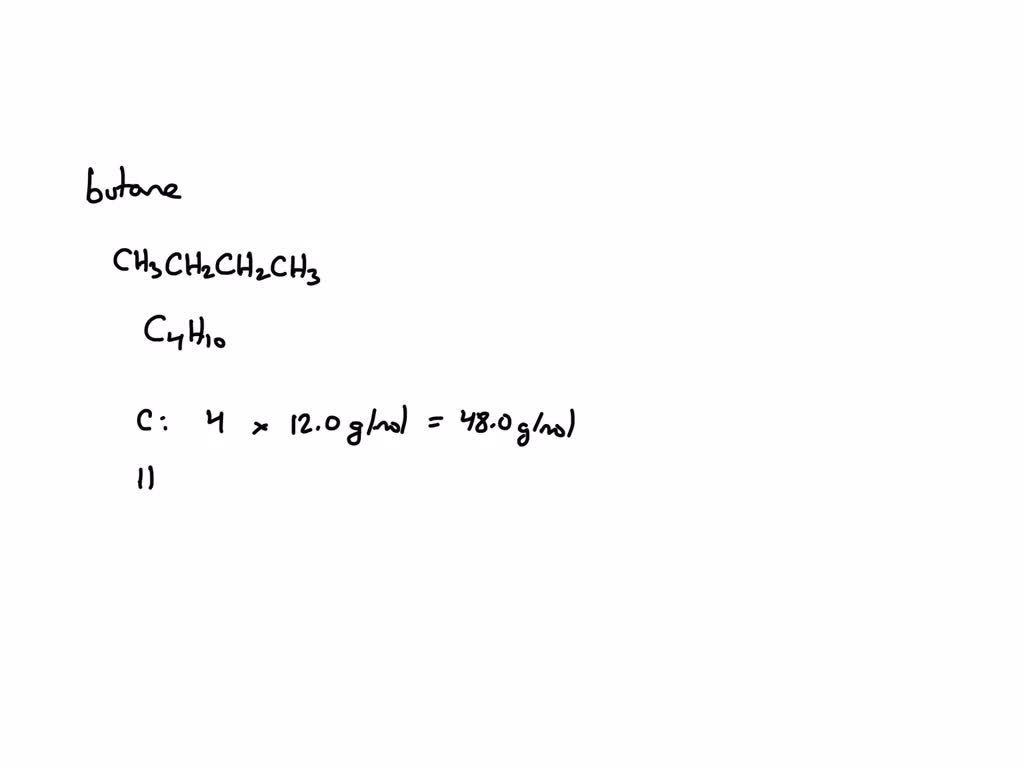
Why Your Lab Answers Might Be a Little Different
This is where the fun really starts. Your experimental molar mass is probably not going to be exactly the theoretical molar mass. And that’s okay! In fact, it's expected. It’s like baking a cake. You follow the recipe, but sometimes it comes out a little puffier, or a little flatter. There are so many reasons why your results might differ.
Think about it. Were there any leaks in your setup? Did all the gas actually get collected? Maybe some of the butane evaporated before you could measure it. Or perhaps your measurements of volume or temperature weren't perfectly accurate. Even the purity of your butane could be a factor. Was it 100% pure, or were there some other gases mixed in? These are the little quirks of the real world that make science so fascinating.
You might have gotten a slightly higher molar mass. This could mean there were some heavier impurities in your sample. Or maybe some air got into your collection system. If you got a lower molar mass, it could indicate that some of your butane escaped, or perhaps there were lighter impurities.
And don’t forget about the temperature and pressure. Gases are sensitive little things. They expand when they’re hot and contract when they’re cold. They also get squished when the pressure is high. So, if your temperature or pressure readings weren’t spot on, it’s going to affect your calculations. It’s all about understanding these variables.
Putting Your Answers to the Test
So, you’ve got your experimental molar mass. How do you present it? This is where you compare your findings to the theoretical value. You’ve probably calculated a percent error. This is basically a measure of how far off you were. And guess what? A small percent error is a good thing! It means your experiment was pretty darn accurate.
Think of it like this: if you were aiming for a bullseye and you landed a few millimeters away, that’s a great shot. If you landed across the room, well, you might need a bit more practice. The same goes for your lab. A small error shows you did a good job managing all those tricky variables. A larger error? It’s an opportunity to learn. What went wrong? What could you do differently next time? It’s a chance to troubleshoot and become a better scientist.

Sometimes, the lab might involve using the ideal gas law: PV=nRT. That’s a mouthful, right? But it's a super useful equation. P is pressure, V is volume, n is the number of moles, R is the ideal gas constant, and T is temperature. You use this to figure out ‘n’, the number of moles. Once you have ‘n’ and you know the mass of your butane, you can easily calculate the molar mass (mass/moles).
It’s like having a secret decoder ring for gases. You plug in your measurements, and out pops the answer. And when your calculated molar mass is close to the theoretical value, it feels like you’ve won. You’ve conquered the invisible world of molecules and emerged victorious with a number that tells you something fundamental about butane.
So, don't just stare at your lab answers. Dive into them! Understand where they came from. Celebrate the close calls and learn from the ones that are a bit wild. Because determining the molar mass of butane isn't just about a number; it's about understanding the building blocks of the world around us. It’s about the thrill of discovery, even if that discovery is about the stuff that fuels your lighter. Pretty cool, right?
