Determining Density Via Water Displacement Answer Key

Ever find yourself staring at a shiny new gadget, a beautiful piece of pottery, or even just a particularly interesting rock and wonder, "What's this thing really made of?" It's that innate curiosity, isn't it? The desire to peel back the layers, to understand the substance beneath the surface. Well, you're in luck, because the universe has handed us a wonderfully simple, almost magical, way to get a peek into an object's density, and it all starts with something we have in abundance: water. That's right, we're talking about the delightfully straightforward method of determining density via water displacement. And don't worry, this isn't some stuffy, lab-coat-required affair. Think of it more like a fun, kitchen-counter chemistry experiment that unlocks a little bit of everyday science.
So, what exactly is density? In a nutshell, it's how much "stuff" is packed into a given space. Imagine two identical boxes. One is filled with feathers, the other with rocks. Which one is heavier? The rock-filled box, of course. That's because rocks are denser than feathers. They have more mass packed into the same volume. Density is your secret weapon for understanding why some things float and others sink, why a balloon filled with helium floats but a regular balloon filled with air doesn't, and even why those fancy, solid-looking glass sculptures are surprisingly heavy. It’s a fundamental property that whispers secrets about an object’s composition and its place in the world.
Now, for the star of our show: water displacement. This is where Archimedes, the ancient Greek mathematician and inventor, would wink at you from across the ages. Legend has it he discovered this very principle while trying to figure out if a king's crown was pure gold or mixed with cheaper metals. He needed a way to measure the crown's volume without melting it down. The story goes that he jumped into a bathtub, noticed the water level rise, and had his "Eureka!" moment. Pretty cool, right? It’s a story that’s been passed down through generations, reminding us that profound scientific discoveries can sprout from the most mundane of observations.
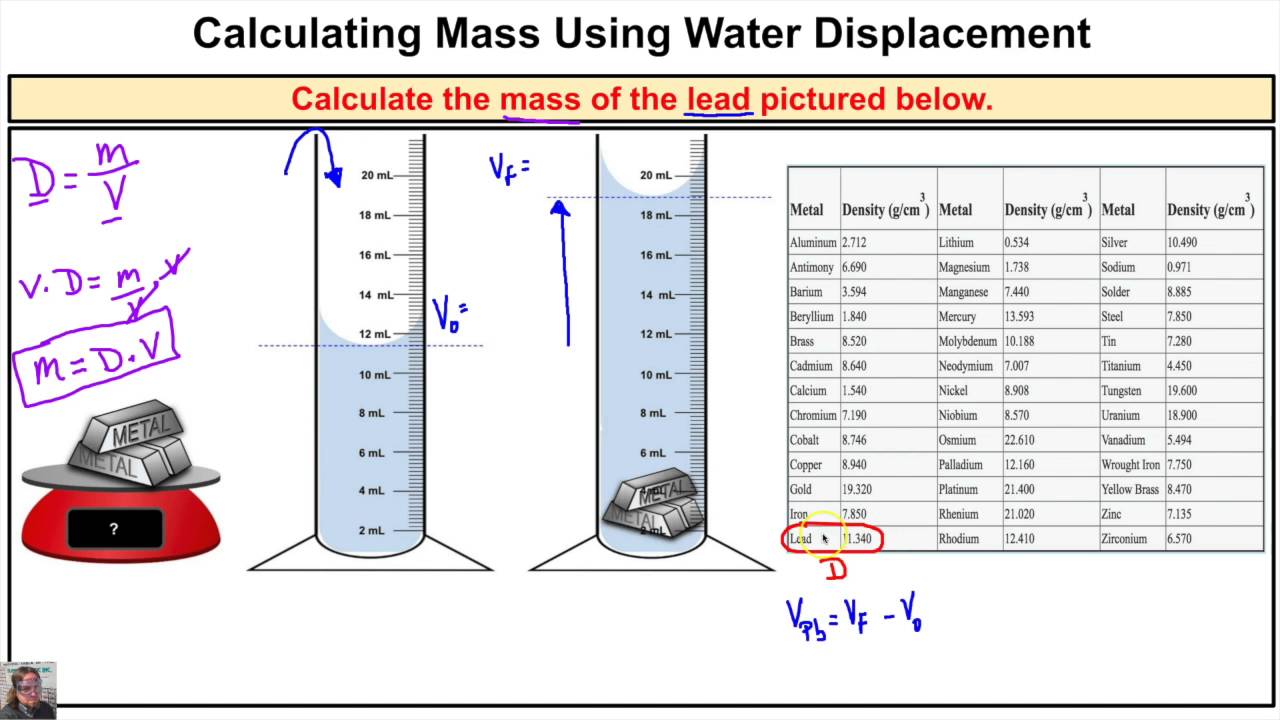
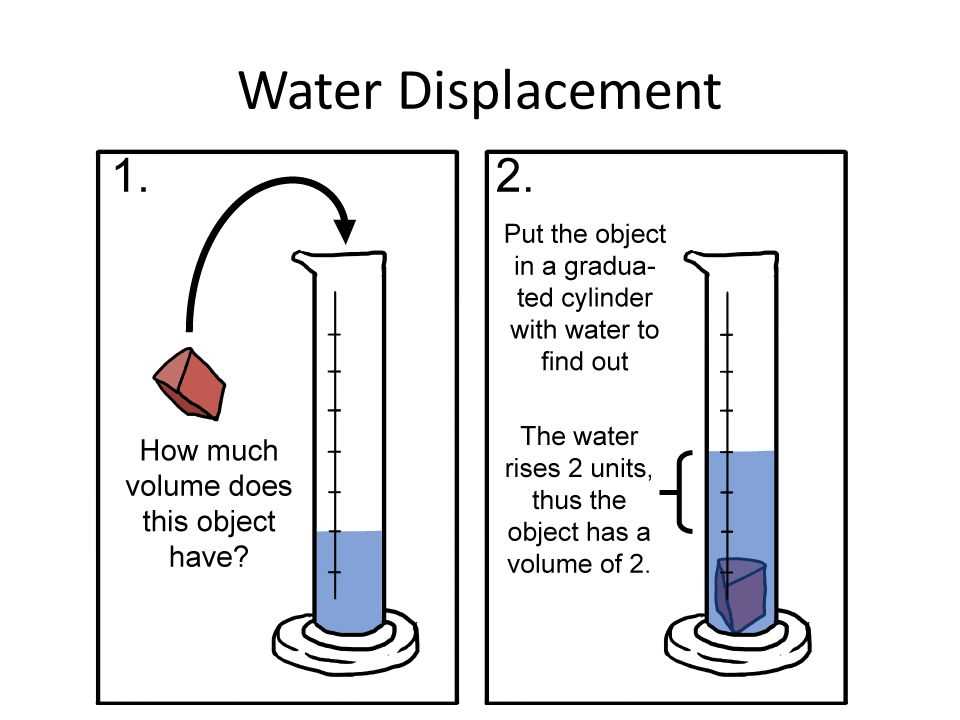
The principle itself is elegantly simple. When you submerge an object in a liquid, it pushes aside, or displaces, an amount of liquid equal to its own volume. Think of it like this: if you’re already crammed into a subway car, and someone else tries to squeeze in, you’re going to have to shift to make space. The amount you shift is like the volume of the object displacing the water. This is the key to unlocking the mystery of an object's volume, and once you have that, the density calculation becomes a breeze.
Let's break down how you'd actually do this at home, turning your kitchen sink or a sturdy bucket into a miniature science lab. You'll need a few things: a container (like a measuring cup with clear markings, a graduated cylinder if you're feeling fancy, or even a clean jar), your object of mystery, and of course, water. First, fill your container with a known amount of water. Be precise here! If you're using a measuring cup, note the starting water level. Let's say you start with 200 milliliters (ml) of water.
Next, carefully submerge your object into the water. Make sure it’s fully underwater. If it floats, you might need to gently push it down with something that won't affect the water level significantly, like a thin rod or a pencil, but be mindful of this for your calculation. Once the object is submerged, observe the new water level. Let’s imagine the water level rises to 250 ml.
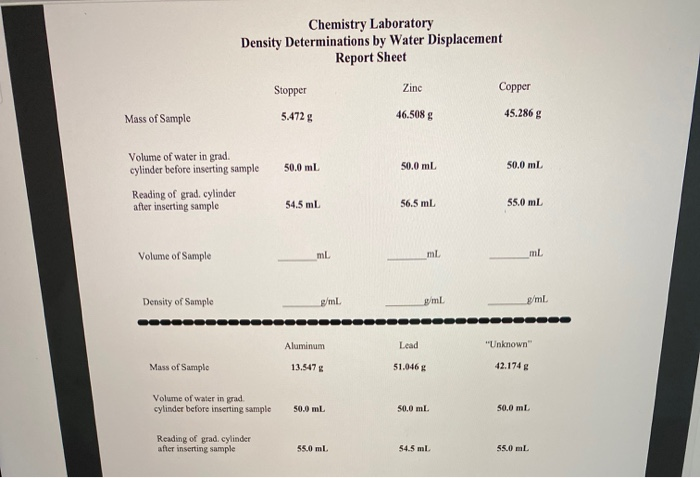
The difference between the final and initial water levels tells you the volume of the object. In our example, that’s 250 ml - 200 ml = 50 ml. So, your mysterious object has a volume of 50 ml. Easy peasy! This is where the magic really starts to happen, because you’ve just measured volume without any complicated tools or formulas. It’s like a secret handshake with the physical world.
Now, to get the density, you need one more piece of information: the object’s mass. You can easily find this using a kitchen scale. Weigh your object. Let's say your object weighs 130 grams (g). You’ll want to record this measurement accurately.
With both mass and volume in hand, you can now calculate density. The formula is simple: Density = Mass / Volume. So, in our example, Density = 130 g / 50 ml. That gives us a density of 2.6 g/ml. Congratulations! You've just determined the density of your mystery object. It’s a number that tells you how compact it is. For comparison, water has a density of 1 g/ml. So, our object is 2.6 times denser than water, which is why it sinks.
This method is incredibly versatile. You can use it to test the density of anything from a tiny pebble to a larger, irregularly shaped toy. Imagine comparing the density of a wooden block versus a metal one. Or perhaps you have a collection of marbles – wouldn't it be fun to see if they all have the same density, hinting at their manufacturing consistency? It’s a fantastic way to engage kids in science too. Turn a rainy afternoon into an exploration of what makes things float or sink. It’s hands-on, visual, and requires minimal fuss. Think of it as a playful introduction to the principles that govern everything from shipbuilding to the flight of airplanes.
There are a few practical tips to keep in mind to make your density determination as smooth as possible. First, accuracy matters, especially with smaller objects or when trying to differentiate between objects with very similar densities. Use a measuring tool with fine graduations if possible. If you’re dealing with objects that absorb water, like certain types of wood or porous rocks, you might need to seal them first with a waterproof coating, like clear nail polish or wax, before submerging them. Otherwise, the water absorbed will affect your volume measurement.
Another tip: ensure the object is fully submerged. If it floats, as mentioned, you’ll need to gently hold it down. But be careful not to add any extra volume with your finger! A small, thin rod is a good option. Alternatively, you can tie a string to the object and hold the string, but remember to subtract the volume of the submerged part of the string from your measurement. This is where the "answer key" for your experiment comes in – it's the precise calculation you arrive at!
And what about the cultural side of density? Think about the materials we use in our daily lives. The difference in density between aluminum and steel is why your car is lighter (and more fuel-efficient) than a train. The density of gold is what makes it so precious and resistant to corrosion. Even in art, artists manipulate materials based on their density. Sculptors work with materials that have different weights and strengths, and understanding density is crucial for creating stable, impactful pieces. From ancient Roman concrete to modern-day aerospace materials, density has been a silent, yet powerful, player in human innovation.

Consider the simple act of baking. Why does a cake rise? It's a complex interplay of ingredients, but density plays a role in how air pockets are formed and retained, giving the cake its light, airy texture. Or think about sailing – the density of the boat's materials, combined with the density of the water, determines whether it floats and how it moves. It's a constant, invisible dance of forces governed by density.
Sometimes, you might get a slightly different answer each time you measure. This is perfectly normal, especially when doing it at home. It’s a good opportunity to talk about experimental error. Did you read the water level perfectly? Was the object completely dry before weighing? These small variations are part of the scientific process and teach us about the nuances of measurement. It's not about getting one "perfect" answer, but about understanding the process and the potential for variation.
The "answer key" for density via water displacement isn't a pre-printed sheet of solutions. It's the process itself. It’s the understanding you gain by performing the experiment, by observing the rise of the water, by feeling the weight of the object. It’s about building an intuition for the physical world around you. It’s about realizing that complex scientific principles can be explored with simple tools and a curious mind. It's the satisfaction of solving a little puzzle using your own hands and brain.
Think about your morning coffee. The density of the coffee grounds versus the water, the way the coffee extracts – it’s all a dance of density. Or consider your favorite piece of jewelry. The subtle difference in weight between a silver ring and a gold one is due to their respective densities. This simple experiment helps demystify those everyday observations. It gives you a tangible connection to the properties of the materials that make up your world.
So, next time you’re presented with an object and a bit of water, don’t just see them as separate entities. See them as partners in a scientific discovery. The water is your liquid measuring tape, and the object is your subject of study. It’s a delightful way to spend a few minutes, turning a mundane item into a fascinating scientific specimen. You’re not just measuring; you’re exploring. You’re not just calculating; you’re understanding.
This whole process, from Archimedes’ bath to your kitchen sink, is a testament to the beauty of observation and the power of simple physics. It’s a reminder that science isn’t just for textbooks and laboratories; it’s woven into the fabric of our everyday lives, waiting to be discovered. And the answer key to density? It’s in your hands, literally, whenever you choose to engage with the world in this wonderfully direct way.
Ultimately, understanding density via water displacement is more than just an academic exercise. It's a way of seeing. It’s about appreciating the subtle differences that make objects behave the way they do. It’s about the quiet satisfaction of solving a little puzzle. So go ahead, grab an object, some water, and a measuring cup. The universe is ready to share its secrets, one displaced drop at a time.
