Determine The Type Of Alcohol Represented By Each Structure.
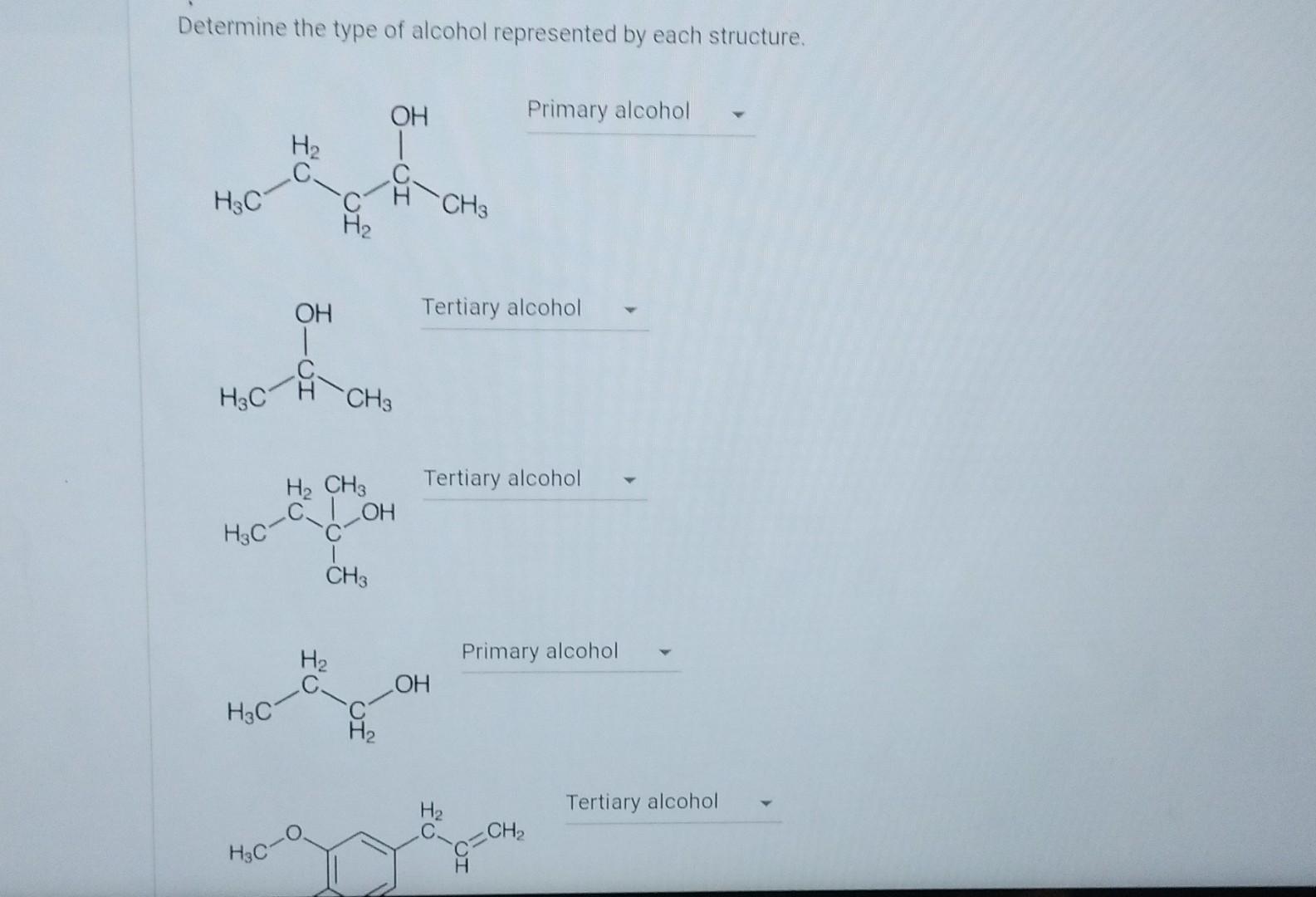
Ever looked at a bunch of little drawings that look like LEGOs stuck together and wondered, "What on earth is this supposed to be?" Well, get ready to have your mind slightly blown, because those drawings are actually the secret code to the amazing world of alcohol! Yup, the stuff that makes celebrations pop and sometimes leads to questionable dance moves. But we're not talking about the bubbly stuff in your glass here. We're diving into the super cool science behind it, and trust me, it's way more interesting than it sounds. Think of it like a fun puzzle, where each piece is a tiny atom, and when you put them together in just the right way, you get a whole different personality – a different type of alcohol!
Imagine you have a bunch of building blocks. You can use the same blocks to build a car, a house, or even a wobbly tower that's destined to fall. It's the same idea with these alcohol structures. They're all made of the same basic ingredients: carbon atoms (like the stuff in pencils!), hydrogen atoms (the most common thing in the universe!), and a special little group called a hydroxyl group. This hydroxyl group is like the "magic wand" of the alcohol world. It's an oxygen atom holding hands with a hydrogen atom (O-H). It's this tiny O-H buddy that really makes an alcohol an alcohol.
So, how do we tell these guys apart? It's all about where that hydroxyl group decides to hang out. Think of the carbon atoms as a little chain, like beads on a necklace. The simplest alcohol, the king of the small fry, is methanol. It's super tiny, just one carbon atom with its friends and that essential hydroxyl group. It's like the baby of the alcohol family. Super important, but you wouldn't want to take it to a party, if you catch my drift. It's pretty potent stuff and can be quite dangerous.
Then we get to ethanol. Aha! This is the star of the show, the one you know and love (or at least recognize from wine bottles and beer cans). Ethanol has two carbon atoms linked together. That hydroxyl group is attached to one of those carbons. It’s this little extra carbon that makes all the difference. Ethanol is the reason we have cocktails, craft beers, and that cozy feeling after a warm mug of mulled wine. It’s the magic behind the buzz, the laughter, and the slightly off-key karaoke sessions. It’s the approachable, fun-loving member of the alcohol club.
But the party doesn't stop there! As you add more carbon atoms to that chain, things get more interesting. Take propanol. It's got three carbon atoms. Now, here's where it gets a little more detailed, and this is where the drawings become really useful. The hydroxyl group can attach to the end carbon, or it can attach to the middle carbon. This might seem like a tiny difference, but it changes the alcohol's personality. It's like having two twins who look almost identical but have completely different hobbies. One might be a daredevil, and the other might prefer reading books. These slightly different structures lead to different properties, like how easily they mix with water or how they behave in chemical reactions. They're both propanol, but they're like cousins with unique quirks.
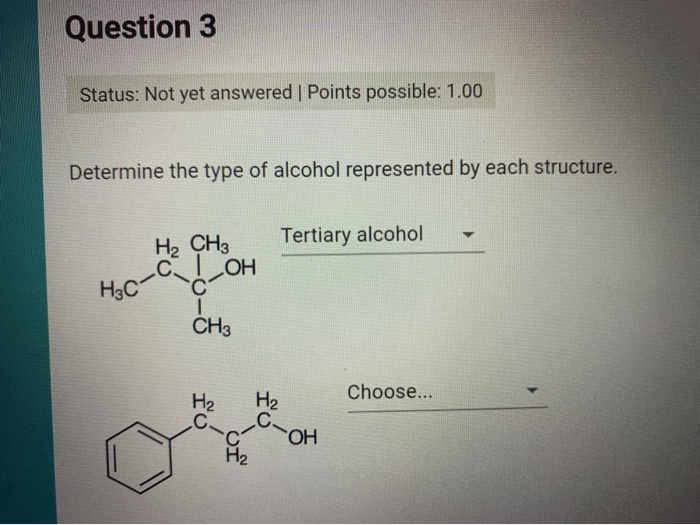
And it keeps going! You can have butanol (four carbons), pentanol (five carbons), and so on, all the way up to super long chains. As these chains get longer, the alcohols start acting less like the familiar spirits and more like oils or waxy substances. They become less soluble in water, meaning they don't mix as easily with your drink. They’re like the quiet relatives who show up to the family reunion but prefer to hang out in the background. The longer the carbon chain, the more the "alcohol" part of the molecule starts to get overshadowed by the "fatty" part of the molecule. It's a fascinating transition.
So, when you see those little stick figures of atoms, remember they're not just random doodles. They're the blueprints for different kinds of alcohol, each with its own special characteristics. You've got your super potent, industrial methanol. You've got your party-starting, celebratory ethanol. And then you have a whole spectrum of other alcohols, from the slightly different propanols to the long-chain molecules that have completely different jobs. It's like a whole universe of molecular personalities, and just by looking at how the atoms are arranged, scientists can figure out exactly what kind of alcohol they're dealing with. It's a beautiful dance of atoms, and understanding it is like unlocking a secret code to the world around you. Pretty neat, right?
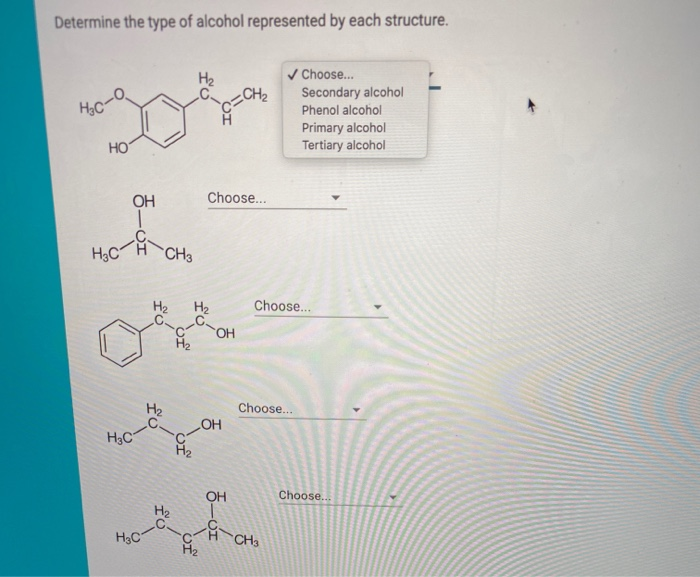
It's like a secret handshake between atoms, and when you know the handshake, you know the molecule!
The shape of the molecule matters! The way those atoms connect and bend dictates everything from how something feels on your tongue to how it reacts with other substances. It’s this structural diversity that makes the world of chemistry so endlessly captivating. Even small changes in arrangement can lead to huge differences in properties. Think of it as the difference between a perfectly tailored suit and one that’s a bit baggy. Both are suits, but one fits beautifully and does its job perfectly, while the other is… well, a bit off. So next time you see one of these chemical diagrams, don't just see lines and circles. See the potential for a party, the promise of a chemical reaction, or the building blocks of something entirely new. It's a tiny world with huge possibilities!
