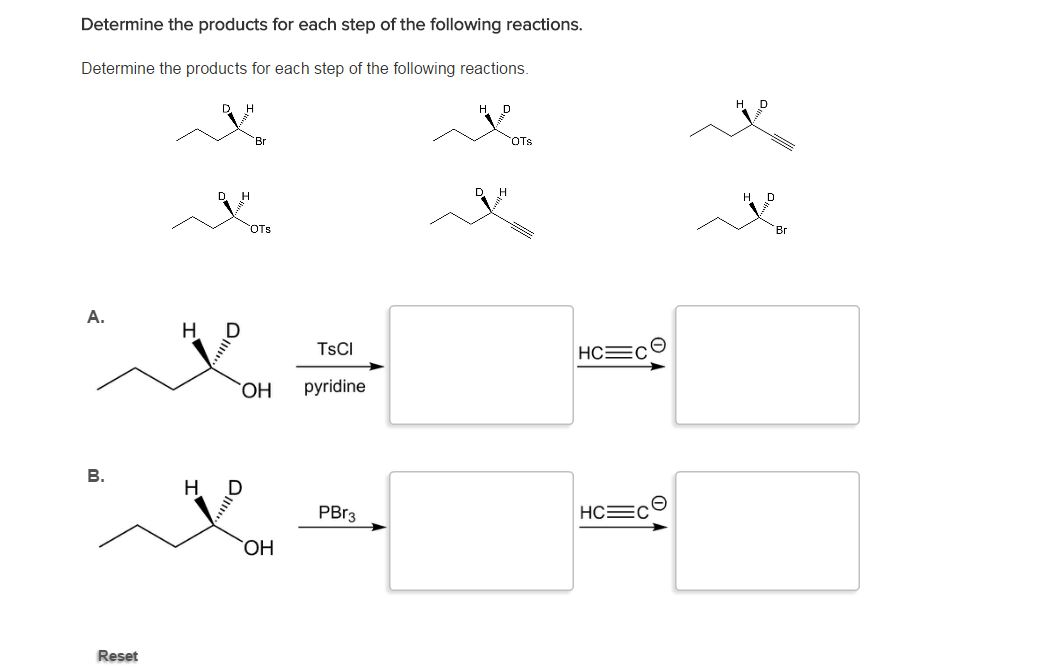
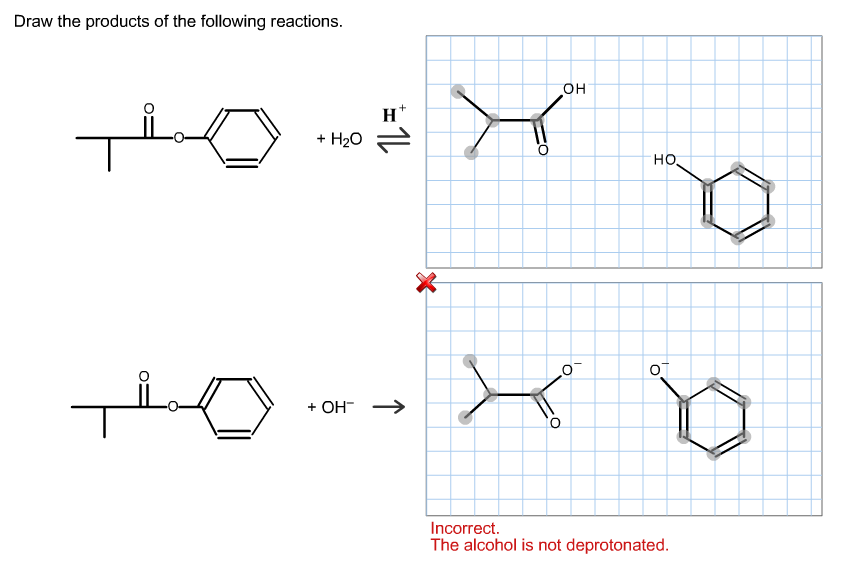
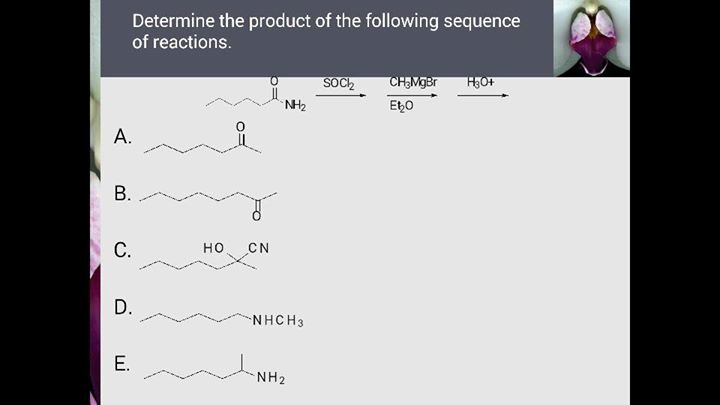
Determine The Products For The Following Reactions
Alright, settle in, grab a cuppa. We're about to embark on a little adventure. It’s the kind of adventure that involves squiggly lines and letters.
Now, I know what you’re thinking. "Chemistry? Ugh." But bear with me. This isn't your dusty textbook. This is chemistry with a wink and a nudge.
Think of it like a fun guessing game. We've got some ingredients, and we need to figure out what delicious, or at least interesting, things they'll turn into. Easy peasy, right? (Okay, maybe not always easy, but definitely entertaining!)
Our First Culinary Concoction
Let’s start with something that looks a bit intimidating. We have Sodium Chloride. Yep, that's just fancy talk for salt.
And what are we mixing this humble salt with? We're throwing in some Silver Nitrate. Imagine tiny, sparkling, silver specks of disappointment.
When these two dance together, something rather predictable happens. It’s like a bad date where you knew it wouldn’t work out.
Our products here are Sodium Nitrate and Silver Chloride.
The sodium, feeling a bit left out, pairs up with the nitrate. And the silver, bless its heart, decides it'd rather be alone with the chloride.
It’s a classic case of swapping partners. Nothing too dramatic, just a polite regrouping.
A Fiery Affair
Next up, let's get a little more energetic. We've got Magnesium. Think of it as a shiny, metal party starter.
And its dance partner? A big, enthusiastic puff of Oxygen. It’s like Magnesium is the grill, and Oxygen is the flame.
When these two get together, things get… bright. Very, very bright. Seriously, don't stare.
The result? Pure, unadulterated Magnesium Oxide.
It’s a solid, white powder. The byproduct of a very energetic reunion. It's the clean-up crew after a wild party.
Think of it as Magnesium finally settling down with a really stable, slightly boring relationship.
A Slightly Acidic Situation
Now, let's add a bit of a bite. We're dealing with Hydrochloric Acid. It’s got a reputation, this one.
And it’s meeting up with Calcium Carbonate. This is basically chalk, or the shell of a very surprised egg.
When these two have a heart-to-heart, there's a lot of fizzing. It’s like they're arguing, but in a bubbly way.
The outcome is Calcium Chloride, Water, and Carbon Dioxide.
Calcium and Chloride form a stable compound. Water is always happy to join the party. And Carbon Dioxide? That's the gas, the "bah humbug" of the chemical world.
It's the effervescence of a spirited debate. A lot of sound and fury, signifying… well, something new!
The Delicate Dissolution
Let's try something a bit more gentle. We have Potassium Iodide. Imagine tiny, pale yellow crystals.

And we're dissolving it in Water. The universal solvent. Water is like the chill friend who just goes with the flow.
When Potassium Iodide meets Water, it doesn't really react in the dramatic sense. It just… chills out.
The result is simply an aqueous solution of Potassium Iodide.
The Potassium ions and Iodide ions are now floating around, happily dispersed. They're just having a lovely swim.
It's less of a reaction and more of a spa day. Very relaxed, very tranquil.
A Explosive Introduction (Not Really)
Okay, let's add a bit of drama. We've got Hydrogen Peroxide. You know, that stuff that bubbles when you pour it on a cut?
And it’s being "helped" by Potassium Iodide again. Our old friend, the yellow crystal.
This time, with the Potassium Iodide acting as a catalyst, things get a bit more exciting. It’s like Hydrogen Peroxide was trying to decompose slowly, and KI said, "Nope, we're doing this now!"
The products are Water, Oxygen, and Potassium Iodide (which is regenerated).
The Hydrogen Peroxide breaks down. Water is formed. And then there’s a big, enthusiastic release of Oxygen gas.
It's the "volcano experiment" you did in third grade, but less messy. A controlled burst of energy.
The Balancing Act
Now, sometimes, the trick isn't just what is made, but how much of it is made. This is where balancing equations comes in.
Imagine you have two hungry people and only one sandwich. You need to give each person half, or you've got a problem.
In chemistry, we need the same number of atoms on both sides of the reaction. It's all about cosmic fairness.
So, for Hydrogen Peroxide decomposition, it’s 2 H₂O₂ → 2 H₂O + O₂.
We need two molecules of Hydrogen Peroxide to make two molecules of Water and one molecule of Oxygen. It's a perfectly balanced equation.
It’s the chemical equivalent of making sure everyone gets a fair share of the pizza. No one's left out.
A Slightly More Complicated Dance
Let's try a slightly more intricate waltz. We have Methane (that’s the main stuff in natural gas) reacting with Oxygen.
This is a combustion reaction. Think fire. Lots of heat and light.
![[ANSWERED] Determine the products of the following reactions and - Kunduz](https://media.kunduz.com/media/sug-question/raw/53058854-1659267093.4438064.jpeg?h=512)
When these two get really fired up, they don't just produce a single thing. It's a bit more of a production.
The products are Carbon Dioxide and Water.
The carbon from Methane joins up with Oxygen to make Carbon Dioxide. And the hydrogen from Methane teams up with Oxygen to make Water.
It’s a complete transformation. The fuel is gone, and its elements are repurposed. Like a phoenix from the ashes, but with less drama.
And if we want to balance this fiery fiesta? We need CH₄ + 2 O₂ → CO₂ + 2 H₂O.
See? One carbon in, one carbon out. Four hydrogens in, four hydrogens out (two water molecules each have two). And four oxygens in, four oxygens out. It all adds up.
It's the ultimate tidy-up operation. All the atoms are accounted for, doing their part in the grand scheme of things.
So, there you have it. A little peek into the world of chemical reactions. It's not so scary when you approach it with a sense of humor, is it?
Just remember, every reaction has its products. Some are predictable, some are surprising, but all of them tell a little story. And isn't that kind of fascinating?
Until our next chemical escapade! Keep those beakers clean and your curiosity sharp!
