Determine The Percent Composition Of Fructose C6h12o6
Hey there, science curious friends! Ever find yourself staring at a juicy piece of fruit, or maybe indulging in a sweet treat, and wondering, "What exactly is all this goodness made of?" Well, today, we're diving into a super fun, surprisingly delightful topic: figuring out the percent composition of fructose, that naturally sweet stuff found in fruits and honey. Sounds a bit like homework, right? But trust me, this is the kind of "homework" that can actually make your life a little sweeter and a whole lot more interesting!
So, what’s this "percent composition" jazz? Think of it like this: when you’re baking a cake, you use different ingredients – flour, sugar, eggs, butter. Each ingredient adds its own flavor and texture, and they all combine to make that delicious final product. Percent composition is kind of the same idea, but instead of cake ingredients, we're talking about the tiny building blocks that make up a molecule. We're going to find out what percentage of fructose is made up of carbon, what percentage is hydrogen, and what percentage is oxygen. Pretty neat, huh?
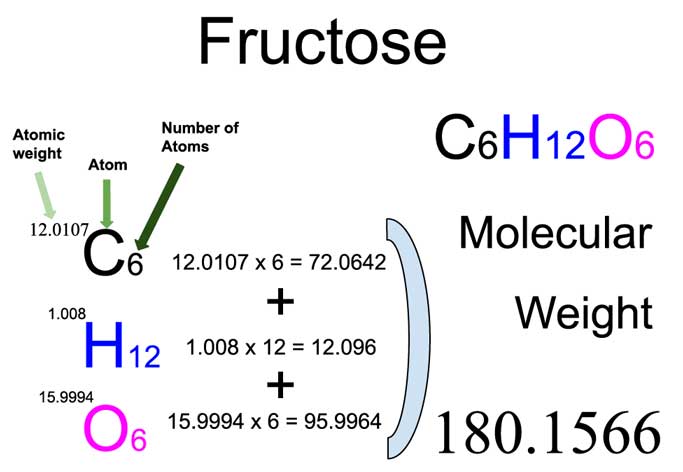
Our star of the show today is fructose, with the chemical formula C6H12O6. Now, don't let those letters and numbers intimidate you! This formula is like a secret code telling us exactly how many atoms of each element are in one single molecule of fructose. See the little '6' next to the 'C'? That means there are 6 carbon atoms. The '12' next to the 'H' tells us there are 12 hydrogen atoms. And the '6' next to the 'O' means we have 6 oxygen atoms. Simple as that!
Why is this cool? Because understanding the building blocks of things around us, from the food we eat to the air we breathe, is like unlocking a secret level in the game of life! It gives you a deeper appreciation for the world and can even spark some amazing ideas. Maybe you'll become a super-chef inventing new recipes, or a brilliant scientist creating healthier sweets, or just someone who can impress their friends with cool science facts at your next picnic. The possibilities are as endless as a summer day!
Alright, let's get down to business. To figure out the percent composition, we need two main things: the atomic masses of each element and the molar mass of the entire molecule. Atomic masses are basically the "weights" of individual atoms. You can find these on the periodic table – your friendly neighborhood guide to all the elements. For our calculation, we'll use approximate values:
Atomic Masses (approximate):
- Carbon (C): 12.01 g/mol
- Hydrogen (H): 1.01 g/mol
- Oxygen (O): 16.00 g/mol
Now, let’s calculate the molar mass of fructose (C6H12O6). This is simply the sum of the masses of all the atoms in one mole of fructose. Remember our secret code? We just multiply the atomic mass of each element by the number of atoms of that element in the molecule and add them all up. Let's do this together, step-by-step. You got this!
Calculating Molar Mass of Fructose:
- Carbon: 6 atoms * 12.01 g/mol = 72.06 g/mol
- Hydrogen: 12 atoms * 1.01 g/mol = 12.12 g/mol
- Oxygen: 6 atoms * 16.00 g/mol = 96.00 g/mol
Add these together, and… voilà! The molar mass of fructose is 72.06 + 12.12 + 96.00 = 180.18 g/mol. High five! We've just calculated the total weight of a mole of fructose. This is our denominator for our percentage calculations, the whole pie we're going to slice up.

Now comes the fun part – figuring out the percentage of each element. We're going to take the total mass contributed by each element in the molecule and divide it by the total molar mass of fructose. Then, we multiply by 100 to get our percentage. Ready to see how much sweetness comes from each element?
Percent Composition Calculation:
Percent Carbon:
We found that 6 carbon atoms contribute 72.06 g/mol to the fructose molecule. So, the percent carbon is:
(72.06 g/mol / 180.18 g/mol) * 100% = 40.00% (approximately)
Isn't that neat? About 40% of fructose is made up of carbon. That’s the backbone of this sweet molecule!
Percent Hydrogen:
And for hydrogen, we calculated 12.12 g/mol. Let's see its percentage:
(12.12 g/mol / 180.18 g/mol) * 100% = 6.73% (approximately)
So, a little over 6% of fructose is hydrogen. It might seem like a small percentage, but hydrogen atoms are super important for creating those delicious chemical bonds!
Percent Oxygen:
Finally, let’s look at oxygen, which contributed 96.00 g/mol. What percentage does it make up?

(96.00 g/mol / 180.18 g/mol) * 100% = 53.27% (approximately)
And there you have it! Over half of fructose, by mass, is oxygen. This makes sense because oxygen is a pretty heavy atom compared to hydrogen, and we have just as many oxygen atoms as carbon atoms, but six times as many hydrogen atoms.
So, to sum it up, the percent composition of fructose (C6H12O6) is roughly:
- Carbon: 40.00%
- Hydrogen: 6.73%
- Oxygen: 53.27%
If you add these percentages up, you should get very close to 100% (40.00 + 6.73 + 53.27 = 100.00%). Isn't that a satisfying feeling? It's like solving a little puzzle and seeing all the pieces fit perfectly!

Why should you care about these percentages? Well, knowing the percent composition of different molecules helps scientists understand their properties. It can tell us a lot about how a substance will behave, what it might be used for, and even how it interacts with our bodies. For fructose, its high oxygen content contributes to its ability to dissolve easily in water, which is why it’s so refreshing in fruit juices.
Plus, think about the bragging rights! The next time you're enjoying a sweet peach or a drizzle of honey, you can casually drop a fact like, "You know, did you realize that about 53% of this deliciousness is oxygen?" It’s a fantastic conversation starter, and honestly, it just makes appreciating food a little more fun when you understand the science behind it.
This whole process of determining percent composition is a fundamental skill in chemistry, but it’s also a gateway to understanding so much more. From the complex molecules in our DNA to the fuel that powers rockets, the principles are the same. It’s about breaking down the complex into its fundamental parts and understanding the proportions. It's a way of seeing the hidden order in the universe.
So, don't stop here! This is just the beginning of your scientific adventure. Pick another molecule – maybe sugar (sucrose) that you put in your coffee, or even water! See if you can figure out its percent composition. The periodic table and a calculator are your trusty companions on this journey. Every calculation you make, every formula you decode, is a step towards a richer, more informed understanding of the world around you. Embrace the curiosity, have fun with the numbers, and let the wonders of chemistry inspire you to keep learning and exploring!
