Determine The Number Of Liters Of Co2 Formed At Stp
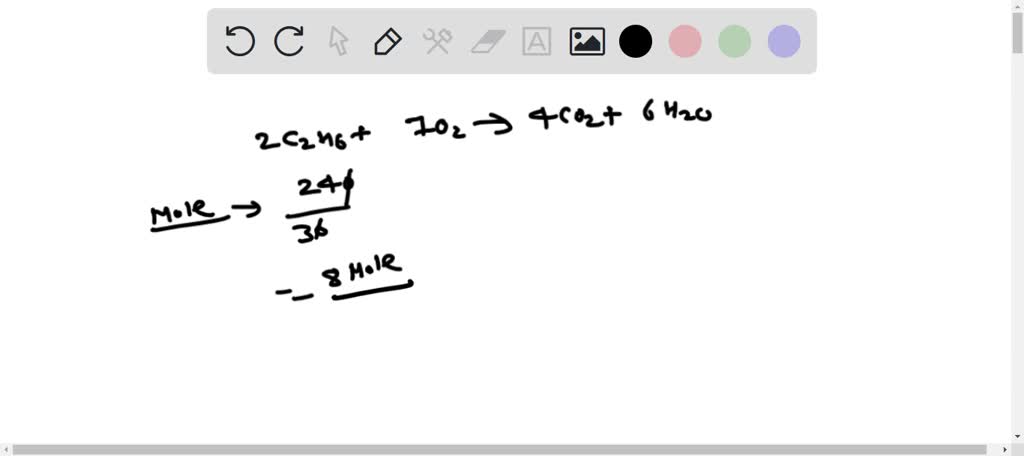
Ever wondered about the invisible stuff that fills our world? We're talking about gases, and in particular, carbon dioxide (CO2). It's a gas we hear about a lot, especially concerning our planet's health. But have you ever stopped to think about how much of it might be produced in a particular scenario? Today, we're going to dive into something a little more specific and surprisingly fascinating: how to determine the number of liters of CO2 formed at STP. Now, "STP" might sound like something out of a sci-fi movie, but it's actually a standard scientific term!
STP stands for Standard Temperature and Pressure. Think of it as a universal measuring stick for gases. When scientists talk about gases at STP, they're referring to a specific set of conditions: 0 degrees Celsius (which is 273.15 Kelvin) and a pressure of 1 atmosphere. Why is this important? Because gases change volume depending on temperature and pressure. By standardizing these conditions, we can make fair comparisons and predictions about gas behavior. So, when we're calculating the liters of CO2 formed, doing it at STP gives us a consistent and meaningful result. It’s like agreeing on a standard ruler before you start measuring anything!
The purpose of determining the volume of CO2 formed at STP is often rooted in chemical reactions. Many common reactions, like burning fuel or the fermentation of sugar, produce CO2. Understanding how much CO2 is generated helps us to quantify the progress of a reaction, assess its impact (especially in terms of environmental emissions), and even design more efficient processes. For instance, in industry, knowing the volume of CO2 produced can be crucial for managing waste streams or for capturing and reusing the gas. It's all about understanding and controlling chemical transformations.
This skill has wonderful applications in both education and daily life, even if you don't realize it. In chemistry classrooms, it's a fundamental concept for understanding stoichiometry – the quantitative relationships between reactants and products in chemical reactions. Students learn to predict how much of a gas will be made from a certain amount of starting material. In a more everyday sense, consider baking! When yeast ferments, it produces CO2, making bread rise. While we don't usually calculate liters of CO2 at STP for our sourdough, the underlying principle is the same: a chemical reaction creating gas. Understanding this can lead to a deeper appreciation of the processes happening around us.

So, how can you explore this concept further? One simple way is to look up common chemical reactions that produce CO2. For example, the reaction between baking soda and vinegar is a classic. You can find the balanced chemical equation and then, using molar mass and the ideal gas law (or a simplified version for STP), calculate the theoretical volume of CO2 produced from a specific amount of baking soda. Many online calculators and educational resources are available that can guide you through these steps. Even if you're not doing the math yourself, observing experiments that produce CO2, like the fizzing of an antacid tablet in water, can spark curiosity. Think about the next time you see something fizzing – you're witnessing the creation of CO2!
Ultimately, understanding how to determine the number of liters of CO2 formed at STP is about unlocking a deeper comprehension of the invisible world of gases and the chemical reactions that shape it. It's a useful skill that bridges the gap between abstract scientific principles and tangible, observable phenomena. So, next time you encounter a chemical reaction, take a moment to consider the potential for gas production – you might be surprised at what you discover!
