Determine The Number Of Ch2cl2 Molecules In 25.0 G Ch2cl2

So, you've got a little bit of dichloromethane, huh? Maybe you found it lurking in the back of a dusty science kit. Or perhaps you're just trying to impress your cat with your newfound chemical prowess. Whatever the reason, you're staring at a container with 25.0 grams of this stuff, labeled CH₂Cl₂.
And then the question pops into your head, the one that keeps you up at night, right after "why do socks disappear in the dryer?" It's a doozy. You're wondering, "How many of these tiny CH₂Cl₂ molecules are actually in there?" It sounds like a riddle, doesn't it?
Let's be honest, nobody actually wakes up in a cold sweat thinking about the molecular count of dichloromethane. That's for the serious science folks. The people who wear lab coats for fun and probably use words like "stoichiometry" in their sleep.
But here we are, contemplating the vastness of the microscopic. It’s like trying to count all the grains of sand on a beach, but way, way, way smaller. And slightly more likely to make you sneeze if you inhale too deeply.
The Grand Molecular Mystery
You see, when we talk about grams, we're dealing with stuff we can actually, you know, hold. We can weigh it. We can even put it in a tiny bottle and pretend we're a mad scientist.
But molecules? They're on a whole other level of small. We can't see them. We can't even really feel them individually. They're like the invisible ninjas of the chemical world, zipping around, minding their own business.
So, how do we bridge this enormous gap between something tangible like grams and something utterly ethereal like individual molecules? It's a bit like trying to measure the number of thoughts in your head by weighing your skull. Not quite right.
A Little Help From Our Friends
Thankfully, science has bestowed upon us a magical number. A true hero of the mole universe. It's called Avogadro's Number. And it’s a lot bigger than you might think.
Avogadro's Number is approximately 6.022 x 10²³. That's a 6 followed by 23 zeros. It's a number so ridiculously huge, it makes the national debt look like pocket change.
This number represents how many "things" are in one "mole" of a substance. Think of a mole as a super-duper, industrial-sized dozen. A dozen is 12. A mole is… well, it's 6.022 x 10²³.
And what are these "things"? In our case, they are our beloved CH₂Cl₂ molecules. So, one mole of dichloromethane contains 6.022 x 10²³ molecules of dichloromethane. Mind-blowing, right?
Now, we have 25.0 grams of CH₂Cl₂. But we need to know how many moles that is before we can unleash the full power of Avogadro's Number. This is where another handy concept comes in: the molar mass.
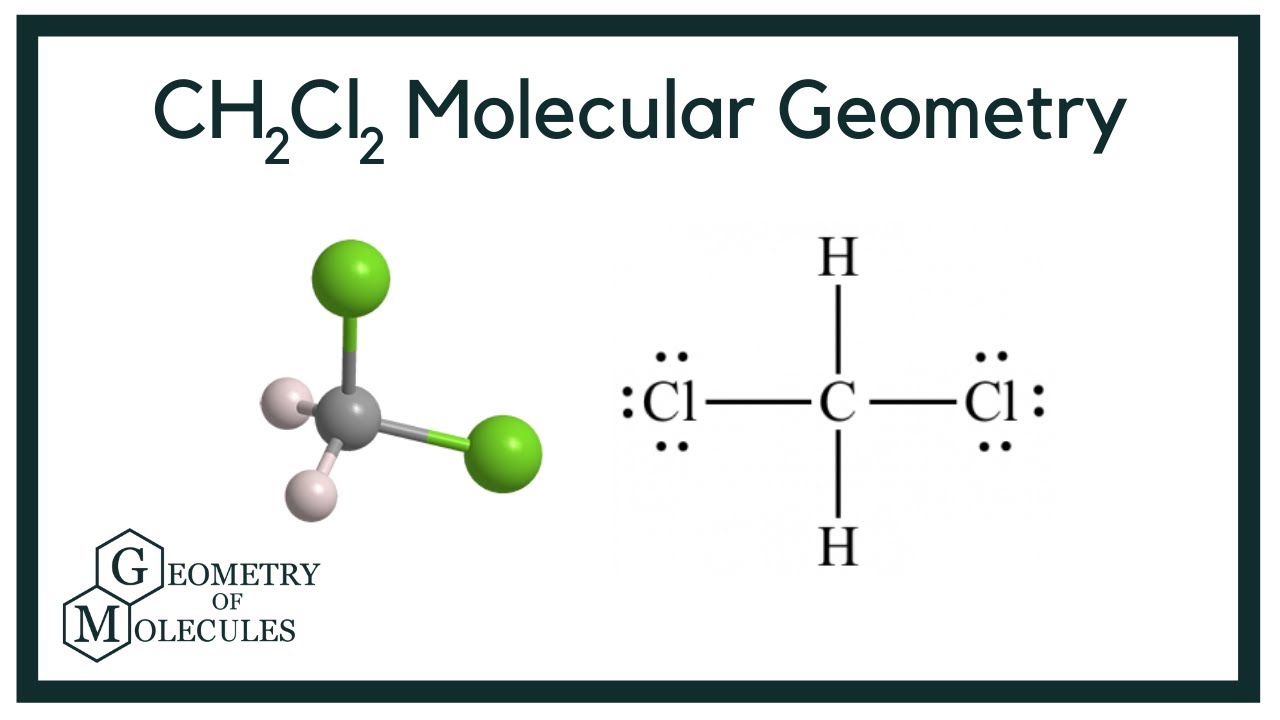
The molar mass is essentially the weight of one mole of a substance. For CH₂Cl₂, we need to add up the atomic weights of each atom in the molecule. We've got one carbon (C), two hydrogens (H), and two chlorines (Cl).
Carbon clocks in at about 12.01 grams per mole. Hydrogen is a lightweight at around 1.01 grams per mole. Chlorine is a bit heftier, at about 35.45 grams per mole.

So, the molar mass of CH₂Cl₂ is roughly:
- 1 * 12.01 (for Carbon)
- + 2 * 1.01 (for the two Hydrogens)
- + 2 * 35.45 (for the two Chlorines)
Do the math, and you get a molar mass of approximately 84.93 grams per mole. This means that 84.93 grams of CH₂Cl₂ contains one mole of CH₂Cl₂ molecules.
We've got 25.0 grams. This is significantly less than 84.93 grams. So, we're definitely dealing with less than one mole. That's a relief, in a way. We don't have to wrap our heads around a trillion trillion molecules just yet.
The Big Calculation (Don't Panic!)
Here's the part where we connect the dots. If 84.93 grams equals 1 mole, then 25.0 grams must equal some fraction of a mole. We can figure that out by dividing our sample weight by the molar mass.
So, moles of CH₂Cl₂ = 25.0 g / 84.93 g/mol.

When you crunch those numbers, you get about 0.294 moles. See? Less than one whole mole. We're making progress. We're not drowning in abstract units of measurement.
Now, we bring in our superhero, Avogadro's Number. We know we have 0.294 moles, and each mole has 6.022 x 10²³ molecules.
To find the total number of molecules, we multiply the number of moles by Avogadro's Number.
Number of CH₂Cl₂ molecules = 0.294 moles * (6.022 x 10²³ molecules/mole).
And the grand total? Drumroll, please…

Around 1.77 x 10²³ molecules of CH₂Cl₂. That's 177 followed by 21 zeros. It’s a number so big it could probably buy its own small country.
So, the next time you’re fiddling with that 25.0 grams of dichloromethane, you can casually inform yourself (or your bewildered pet) that you’re holding over 177 sextillion tiny CH₂Cl₂ molecules.
It’s a fun little fact to have in your back pocket, isn't it? It makes those mundane lab tasks feel a little more… cosmic. And it’s a good reminder that even the tiniest things can exist in truly astonishing quantities.
And who knows? Maybe understanding the sheer number of molecules in a small sample will inspire you. Maybe you’ll go on to discover a new element, or invent a self-folding laundry machine. Or maybe you’ll just appreciate the silent, invisible dance of the atoms a little more.
Either way, you’ve conquered the riddle of the CH₂Cl₂ molecules. And that, my friend, is pretty darn cool. Even if it wasn't a question you really needed answered, it’s nice to know the answer, isn't it? It’s like finding a lost button. You didn't realize you were missing it, but now that it's found, everything feels a little more complete. Even the molecular count of dichloromethane.
Just remember to keep it away from open flames and anything that might spontaneously combust from sheer awe at your scientific knowledge. Because even though these molecules are small, their collective power is, well, astronomical.
