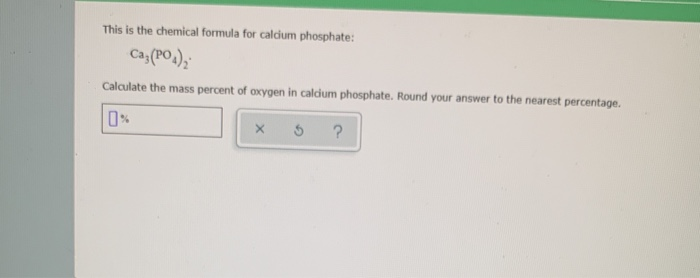
Determine The Mass Percent Of Oxygen In Calcium Phosphite
Okay, so we need to figure out the mass percent of oxygen in a compound called calcium phosphite. Sounds fancy, right? Like something you’d find in a mad scientist’s lab or maybe a really old cookbook. But trust me, it’s less about bubbling beakers and more about… well, numbers.
Now, before you run for the hills or start humming the Jeopardy theme song, let’s break this down. Think of it like trying to guess how much of a pizza is just pepperoni. You know, the good stuff. In our case, the "good stuff" is oxygen, and the "pizza" is calcium phosphite.
First things first, we need to know what this calcium phosphite is made of. It’s like having the ingredients list for your pizza. We’ve got calcium, which is a metal. Think of it as the dough. Then we have phosphite. This is a bit more complicated. It’s a bit like a fancy topping, a blend of phosphorus and oxygen.
So, the full recipe for calcium phosphite is a bit like this: one part calcium (Ca) hanging out with some phosphite bits. The phosphite bit itself is made of phosphorus (P) and a couple of oxygen atoms (O). Put it all together, and our calcium phosphite has the formula Ca₃(PO₃)₂. See? Not so scary. It’s just a chemical handshake.
Now, for the math part. This is where it gets a little… math-y. We need to find the atomic masses of each element. These are basically the "weights" of the atoms. You can find these on a handy little chart called the periodic table. It’s like a cheat sheet for the elemental world.

Let’s grab our numbers:
Calcium (Ca): About 40.08 grams per mole. Let’s just call it 40 for simplicity.
Phosphorus (P): About 30.97 grams per mole. We’ll round this to 31.
50. Calculate the mass percentage of calcium, phosphorus and oxygen in ca..Oxygen (O): About 16.00 grams per mole. This one’s a nice round 16.
Now, back to our formula: Ca₃(PO₃)₂. This little notation tells us how many of each atom we have in one "unit" of calcium phosphite.
- We have three calcium atoms (Ca₃). So, 3 * 40 = 120.
- Then we have the parentheses, which means everything inside gets multiplied by the little 2 outside. Inside, we have one phosphorus atom (P) and three oxygen atoms (O₃). So, for one set of parentheses, it’s (31 + (3 * 16)). That’s 31 + 48 = 79.
- Since we have two of these parenthesized groups (PO₃)₂, we multiply that 79 by 2. So, 79 * 2 = 158.
Okay, so the total mass of one unit of calcium phosphite is the sum of all these parts: 120 (from calcium) + 158 (from the phosphite bits) = 278. This is our total "pizza" weight.

Now, we’re only interested in the oxygen part. How much of that total weight comes from the oxygen? Well, remember how we calculated the phosphite part? Inside each (PO₃)₂, we had three oxygen atoms. So, 3 * 16 = 48. And since we have two of those groups, it's 48 * 2 = 96. That’s the "pepperoni" weight!
To find the mass percent of oxygen, we do a simple division. We take the "pepperoni" weight (the total mass of oxygen) and divide it by the "pizza" weight (the total mass of calcium phosphite). Then we multiply by 100 to get a percentage.
(Mass of Oxygen / Total Mass of Calcium Phosphite) * 100
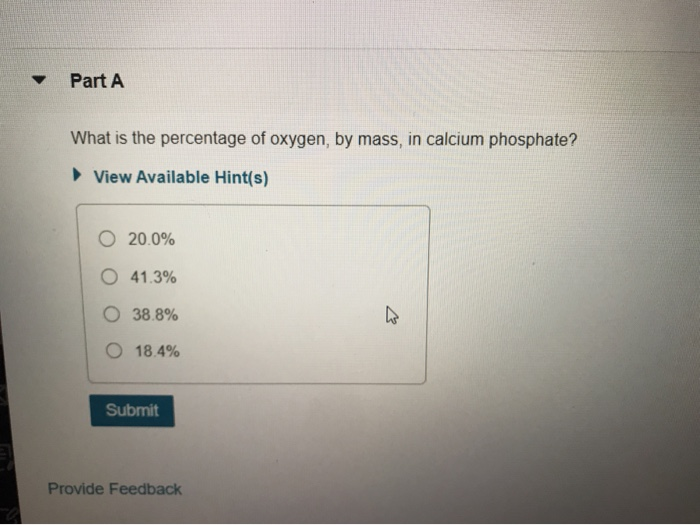
Solved This is the chemical formula for calcium phosphate: | Chegg.com(96 / 278) * 100
And when you crunch those numbers, you get approximately 34.53%. So, about 34.53% of calcium phosphite is made up of good old oxygen.
There you have it! We’ve demystified calcium phosphite and its oxygen content. Was it thrilling? Maybe not in the way a roller coaster is thrilling. But there’s a certain satisfaction in understanding these tiny, invisible building blocks of the universe. It’s like solving a very small, very specific puzzle. And who doesn’t love a good puzzle? Even if it involves slightly too much math for a Tuesday afternoon. My unpopular opinion? Math can be surprisingly fun when it’s about pizza toppings. Or, you know, chemical compounds. Whatever floats your boat, or in this case, whatever makes your calcium phosphite complete.