Determine The Hybridization At Each Of The 3 Labeled Atoms

Ever glanced at a molecule and felt a tiny pang of curiosity? You know, those intricate little structures that make up… well, everything? From the air we breathe to the coffee that fuels our mornings, they're the unsung heroes of our existence. Today, we're diving into a fun, approachable exploration of a concept called hybridization. Think of it as the molecular equivalent of choosing the right outfit for the occasion – it's all about how atoms get their geometry and how they interact. Don't worry, we're not hitting you with a dense textbook chapter. We're talking chill vibes, easy explanations, and maybe a sprinkle of science-y magic.
We've all got those friends who just fit in anywhere, right? They can rock a fancy dinner party and then hit up a casual BBQ without missing a beat. Atoms are a bit like that, and hybridization is their secret sauce for fitting together perfectly. It's essentially how atomic orbitals, those fuzzy regions where electrons hang out, decide to mix and mingle to form new, more stable shapes. This mixing allows atoms to form bonds with their neighbors in just the right way, creating the diverse and beautiful world we see around us.
Imagine you have a bunch of LEGO bricks – some are round, some are square, some are long. Hybridization is like deciding to smash some of those bricks together and mold them into entirely new shapes that are perfect for building whatever structure you have in mind. These new, molded bricks (hybrid orbitals) are more effective at bonding and give molecules their characteristic shapes. Pretty neat, huh?
Let's Get Down to the Nitty-Gritty (But Keep it Breezy!)
Our mission today is to peek at a specific molecular scenario – one with three distinct, labeled atoms – and figure out the hybridization of each. Think of it as a molecular scavenger hunt! We'll be looking at how many "things" are attached to each atom and how many "lone pairs" of electrons it's got hanging out. These two factors are our trusty compass for navigating the world of hybridization.
In the simplest terms, hybridization is determined by the steric number. Don't let the fancy name scare you! The steric number is simply the sum of the number of atoms bonded to a central atom plus the number of lone pairs of electrons on that central atom. So, if an atom is holding hands with two other atoms and has two lonely electrons chilling nearby, its steric number is 2 + 2 = 4. Easy peasy.
Here are the main players in our hybridization game:
- sp³ Hybridization: This happens when an atom has a steric number of 4. It’s like having four hands ready to high-five its neighbors! Think of tetrahedral shapes, like a perfect pyramid with a base.
- sp² Hybridization: This is for atoms with a steric number of 3. Three hands ready to go! This usually leads to trigonal planar geometry – imagine a flat, triangular pizza.
- sp Hybridization: When an atom has a steric number of 2, it's got two hands to offer. This often results in linear geometry, like a perfectly straight string.
You might be wondering, "Why bother with this?" Well, these different hybridizations dictate the shape of the molecule. And the shape of a molecule is super important! It affects everything from how a drug interacts with your body to how a scent molecule fits into your nose receptors. It’s like how the right shoe fits your foot perfectly – form and function, working in harmony.
Our Molecular Muse: Unpacking the Labels
Let's set the scene. Imagine a molecule, and we've got three atoms in focus, conveniently labeled. We'll call them Atom 1, Atom 2, and Atom 3. Our job is to go through each one, count its attachments and lone pairs, and declare its hybridization with confidence. Think of it as giving each atom its own unique molecular superpower.
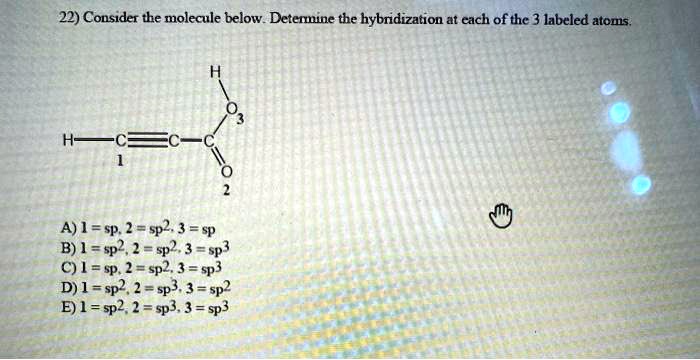
Atom 1: The Solid Citizen
Let's start with Atom 1. We take a close look, and we see it's bonded to, let's say, four other atoms. No lonely electrons are visible hanging out on Atom 1. So, its steric number is 4 (4 bonded atoms + 0 lone pairs). When we see a steric number of 4, what does that signal? That's right, it's our friend sp³ hybridization.
Atoms with sp³ hybridization are the workhorses of the molecular world. They tend to form single bonds and create molecules with a nice, spread-out, tetrahedral geometry. Think of methane (CH₄), the simplest organic molecule. The carbon atom in methane is sp³ hybridized, and the molecule is shaped like a little diamond. It's a classic, reliable structure, much like a sturdy oak table – dependable and ready for anything.
This sp³ hybridization is responsible for the strength and stability of countless organic compounds, the very building blocks of life. It’s the foundation upon which complex biological molecules are built. So, next time you see a molecule with a tetrahedral arrangement, you can confidently say, "Ah, sp³ at play!"
Atom 2: The Artistic Assembler
Now, let's move to Atom 2. We observe that Atom 2 is connected to three other atoms. And, importantly, there's one lone pair of electrons chilling on Atom 2. So, the steric number for Atom 2 is 3 (3 bonded atoms + 1 lone pair) = 4.
Hold on a sec! A steric number of 4 usually means sp³? Well, not always. When lone pairs get involved, things can get a little more interesting. In this specific case, with three bonded atoms and one lone pair, Atom 2 is sporting sp³ hybridization. It's still using those four hybrid orbitals, but one of them is occupied by the lone pair, influencing the molecule's overall shape.

This arrangement often leads to a trigonal pyramidal shape, which is like a pyramid with a triangular base, but with one of the corners "pushed in" by the lone pair. A perfect example is ammonia (NH₃). The nitrogen atom in ammonia has three hydrogen atoms bonded to it and one lone pair. This is why ammonia is not flat and triangular, but has that distinct pyramidal shape. The lone pair acts like an invisible hand, subtly steering the molecule's form.
The presence of lone pairs is super important. They contribute to the molecule's reactivity and its ability to form intermolecular attractions, like hydrogen bonds. Think of it as the subtle nuances in a person's personality that make them unique and capable of deep connections. Lone pairs add that extra dimension!
Atom 3: The Elegant Emitter
Finally, let's shine a spotlight on Atom 3. We see that Atom 3 is bonded to two other atoms. And, crucially, there are two lone pairs of electrons nestled around Atom 3. So, the steric number for Atom 3 is 2 (2 bonded atoms + 2 lone pairs) = 4.
Again, we have a steric number of 4, but with a different combination of bonded atoms and lone pairs. In this scenario, with two bonded atoms and two lone pairs, Atom 3 is also utilizing sp³ hybridization. The four sp³ hybrid orbitals are occupied by the two bonding pairs and the two lone pairs.
What kind of shape does this create? This combination typically leads to a bent or angular geometry. The classic example is water (H₂O). The oxygen atom in water is bonded to two hydrogen atoms and has two lone pairs of electrons. This is why water isn't linear, like O=H-H, but rather bent, with the hydrogen atoms forming an angle. It's this bent shape that gives water its unique properties, like its ability to act as a universal solvent.
The bent shape of water is crucial for life as we know it. It allows water molecules to form hydrogen bonds with each other, which is essential for processes like the transport of nutrients in our bodies and the regulation of Earth's temperature. So, even with "just" two bonds and two lone pairs, the resulting shape can have profound implications!
A Quick Recap and Some Fun Facts
So, in our little molecular exploration today, all three of our labeled atoms ended up being sp³ hybridized. This is a common scenario, especially in organic chemistry, where carbon atoms often form four single bonds or have a combination of single bonds and lone pairs that add up to a steric number of 4.
It's important to remember that hybridization isn't just about counting. It's about understanding how atoms adjust their electron configurations to achieve the most stable and efficient bonding. It's like a molecular dance, with atoms rearranging themselves to find their perfect partners and positions.
Fun Fact Alert! Did you know that the concept of hybridization was first proposed by Linus Pauling in 1931? He was trying to explain the bonding in molecules like methane, and it revolutionized our understanding of chemical bonding. He later went on to win two Nobel Prizes – one in Chemistry and one for Peace! Talk about a versatile mind.
Another little tidbit: While sp³, sp², and sp are the most common, there are also higher levels of hybridization like sp³d and sp³d² for elements in the third period and beyond, which involve the use of d orbitals. These allow for even more complex molecular geometries, like octahedral shapes. But for our easy-going introduction, sp³, sp², and sp are your essential toolkit.
Think of it this way: sp³ is like the reliable SUV – it’s versatile, can handle most terrains, and is great for everyday tasks. sp² is like the sleek sports car – it's built for speed and agility, often found in flat, dynamic structures. And sp is like the nimble motorcycle – it’s all about linear efficiency and directness.
Connecting to the Everyday Vibe
Why does all this molecular geometry matter beyond the lab? Well, consider the world of flavors and fragrances. The specific three-dimensional shape of a molecule determines how it interacts with the smell receptors in your nose or the taste buds on your tongue. That's why a strawberry smells like a strawberry and not, say, a pine tree. The subtle differences in how atoms are arranged, dictated by their hybridization, create the distinct sensory experiences we enjoy.
Even something as simple as a plastic bottle has its structure determined by the hybridization of the atoms within it. The strength and flexibility of that plastic are a direct result of the molecular architecture. It’s a constant reminder that even the most seemingly mundane objects are intricate marvels of molecular design.
So, the next time you’re enjoying a cup of coffee, taking a deep breath of fresh air, or even just looking at the world around you, take a moment to appreciate the invisible world of molecules. And when you encounter a molecule with labeled atoms, you’ll have a little trick up your sleeve to decipher their hybridization. It’s a small piece of knowledge, but it unlocks a deeper appreciation for the complexity and beauty that surrounds us, all thanks to the clever ways atoms decide to get together.
It’s a journey of discovery, one atom at a time, making the familiar feel a little more magical. And that, in essence, is what makes life, and science, so endlessly fascinating.
