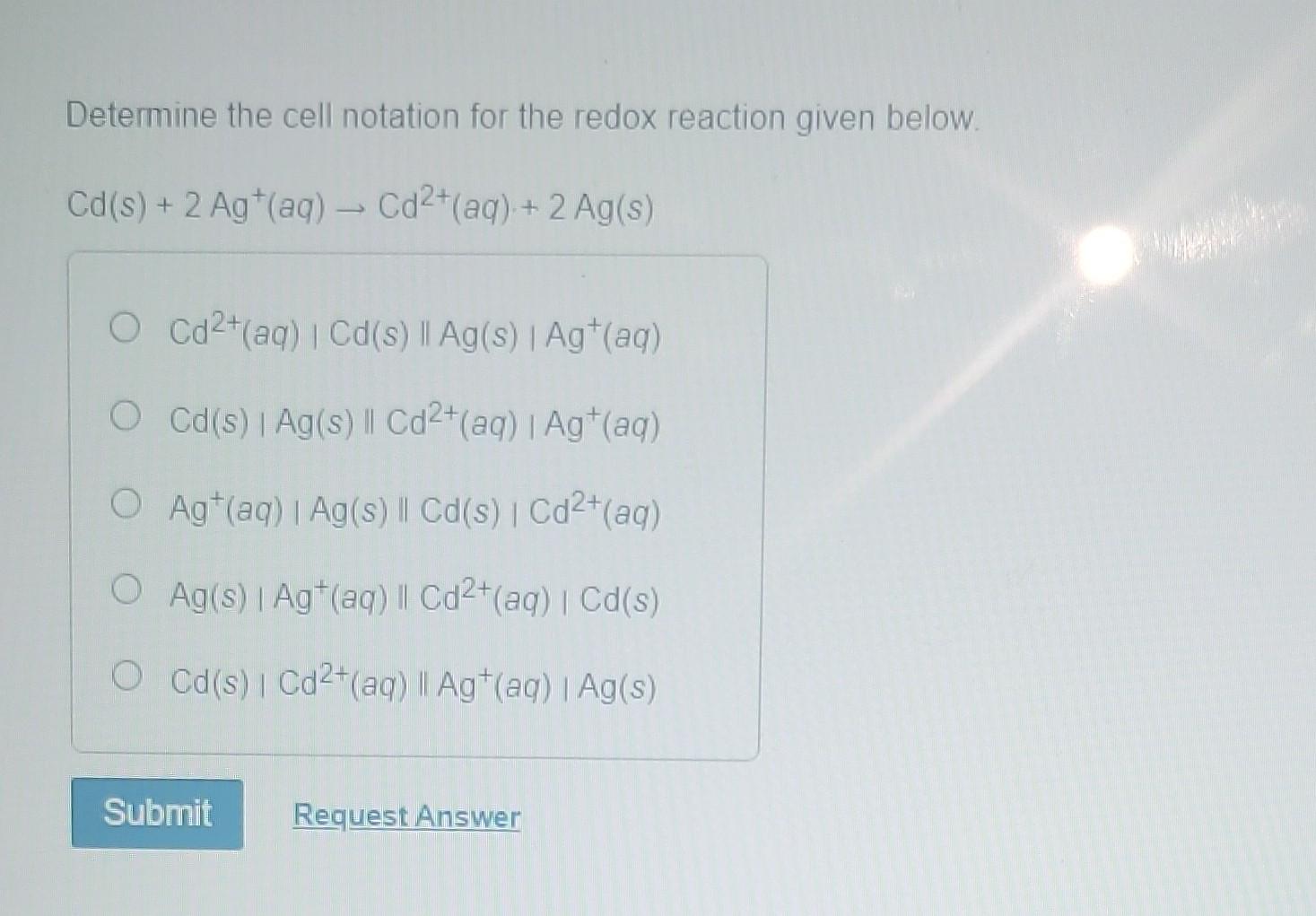
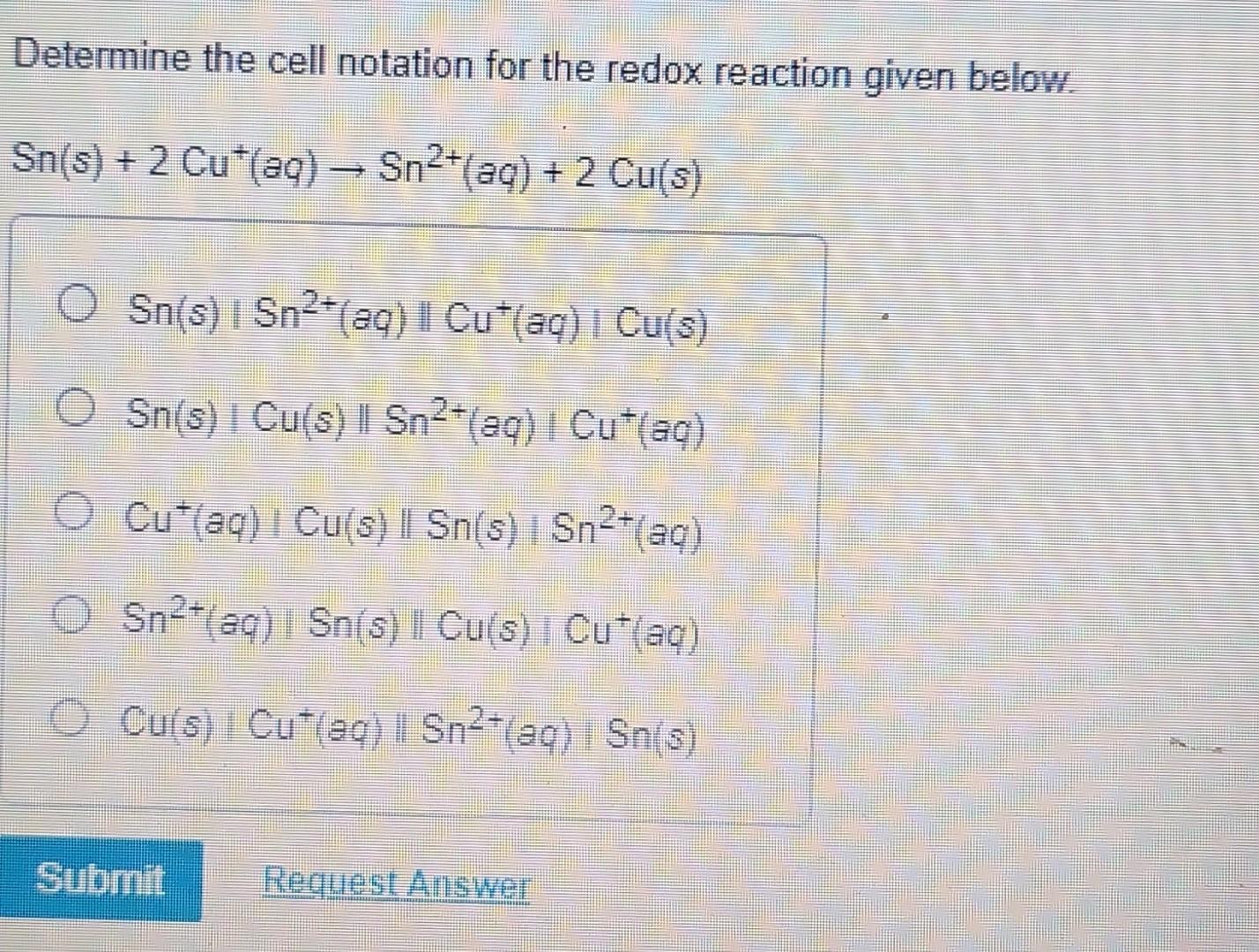
Determine The Cell Notation For The Redox Reaction Given Below
Ever wondered how batteries work, or how we get energy from chemical reactions? It all boils down to something super cool called redox reactions! These aren't just boring textbook concepts; they're the engine behind so many everyday technologies and natural processes. Think about how your smartphone keeps going, or how your body converts food into energy – yep, redox reactions are involved!
And the really neat part? We can represent these energetic dances of electrons using a special shorthand called cell notation. It’s like a secret code that lets chemists quickly and clearly describe a whole redox reaction without writing it all out. Pretty handy, right? It’s a way to visualize the fundamental players and the action happening at each side of the reaction. It’s a simplified yet powerful representation that speaks volumes to those in the know.
So, why is learning to decipher this cell notation a fun and useful skill? Well, it’s like learning to read a map for the microscopic world. Once you understand the symbols, you can instantly grasp where electrons are being lost (oxidation) and where they're being gained (reduction). This knowledge isn't just for scientists in lab coats; it’s fundamental to understanding electrochemistry, which powers everything from tiny implanted medical devices to massive industrial processes. Being able to read cell notation is like having a superpower for understanding how chemical energy is converted into electrical energy and vice-versa.
The purpose of cell notation is to provide a standardized and concise way to represent a galvanic cell (also known as a voltaic cell), which is a device that generates electrical energy from a spontaneous redox reaction. It’s the heart of batteries and fuel cells. By looking at the cell notation, you can immediately identify the anode (where oxidation occurs) and the cathode (where reduction occurs), the electrode materials, and the ions involved in the solution. It’s a visual blueprint of the electrochemical setup.
The benefits of understanding cell notation are numerous. Firstly, it makes it much easier to communicate complex electrochemical information efficiently. Instead of lengthy descriptions, a short string of symbols tells the story. Secondly, it’s crucial for predicting the cell potential, which is the driving force of the reaction. This allows us to determine if a reaction will occur spontaneously and how much energy it can produce. This is vital for designing and optimizing electrochemical devices. Imagine trying to build a better battery without knowing which reactions are happening where – it would be like building a house without a blueprint!

Moreover, understanding cell notation helps in troubleshooting electrochemical systems. If something isn't working as expected, being able to break down the reaction into its anodic and cathodic components using cell notation can be the key to diagnosing the problem. It’s a fundamental building block for anyone delving into electrochemistry, materials science, or even biology, where redox reactions are ubiquitous. So, let’s dive into how we can actually determine the cell notation for a given redox reaction. It’s less about memorization and more about understanding the flow of electrons and the roles of different species.
When faced with a redox reaction, the first crucial step is to identify which species are being oxidized and which are being reduced. Remember the handy mnemonic: OIL RIG – Oxidation Is Loss (of electrons), and Reduction Is Gain (of electrons). You'll need to look at the oxidation states of the atoms involved before and after the reaction. A change in oxidation state signifies that a redox process has occurred.
For instance, consider a reaction where zinc metal (Zn) reacts with copper ions (Cu2+) to form zinc ions (Zn2+) and copper metal (Cu). In this scenario, zinc starts with an oxidation state of 0 and ends up as Zn2+ (oxidation state +2). This means zinc has lost two electrons and has been oxidized. Conversely, copper ions start as Cu2+ (oxidation state +2) and end up as copper metal (Cu, oxidation state 0). Copper ions have gained two electrons and have been reduced.

Once you've identified the oxidation and reduction half-reactions, you can start constructing the cell notation. The general format for cell notation is:
Anode | Anions || Cathode | Cations
However, a more common and detailed format is:

Oxidized Species (in anode compartment) | Reduced Species (in anode compartment) || Oxidized Species (in cathode compartment) | Reduced Species (in cathode compartment)
Let's refine this further. The anode compartment, where oxidation happens, is written first. It’s represented by the species being oxidized followed by the species being reduced. A single vertical line (|) signifies a phase boundary, like between a solid electrode and a solution. For example, if a solid metal is oxidized into its ions in solution, it would be written as Metal | Metal Ion.
The cathode compartment, where reduction happens, is written second. It’s represented similarly, with the species being oxidized (which is gained in this case) followed by the species being reduced. A double vertical line (||) signifies a salt bridge or a porous barrier that allows ion flow to complete the circuit while keeping the two half-cells separate. So, if ions in solution are reduced to a solid metal, it would be written as Ion | Metal.
Applying this to our zinc and copper example: Zinc is oxidized: Zn → Zn2+ + 2e-. This happens at the anode. Copper ions are reduced: Cu2+ + 2e- → Cu. This happens at the cathode. So, the cell notation would be: Zn | Zn2+ || Cu2+ | Cu
Here’s a breakdown of what each part signifies:
- Zn: This represents the solid zinc electrode, which is being oxidized.
- |: This is the phase boundary between the solid zinc and the zinc ions in solution.
- Zn2+: This represents the zinc ions in the anode compartment solution.
- ||: This is the salt bridge, connecting the two half-cells.
- Cu2+: This represents the copper ions in the cathode compartment solution.
- |: This is the phase boundary between the copper ions in solution and the solid copper electrode.
- Cu: This represents the solid copper electrode, where copper ions are reduced and deposit.
It's important to note that if the species involved in a half-reaction are all in the same phase (e.g., ions in solution), we might include a Pt (platinum) electrode, which acts as an inert conductor. For example, if we had the reduction of Fe3+ to Fe2+, and Fe3+ and Fe2+ were in solution, the cathode notation might look like: Pt | Fe3+, Fe2+ (often written with concentrations or activities). The comma indicates species in the same phase.
The beauty of cell notation lies in its ability to encapsulate the essence of a redox reaction in a highly structured and understandable format. It’s a powerful tool for anyone wanting to unravel the mysteries of electrochemical processes and appreciate the fundamental electron transfers that drive so much of our modern world.
