Designing A Galvanic Cell From Two Half-reactions
Hey there, science enthusiasts and curious cats! Ever looked at a battery and wondered, "How in the heck does this tiny thing power my life?" Well, buckle up, buttercups, because today we're diving into the super cool world of galvanic cells. And guess what? You don't need a lab coat or a PhD to get it. We're going to build one, conceptually at least, from the ground up. Think of it as assembling a LEGO set, but instead of plastic bricks, we're using chemical reactions!
So, what's a galvanic cell anyway? Basically, it's a fancy name for a battery. It's a device that converts chemical energy into electrical energy. You know, the magic that makes your phone hum or your flashlight shine. And the secret sauce? Two half-reactions working together like a dream team.
The Dream Team: Understanding Half-Reactions
Before we start playing mad scientist, let's chat about these "half-reactions." Imagine a reaction where electrons are being transferred. This transfer doesn't happen in one fell swoop. Nope. It's like a relay race. One side loses electrons, and the other side gains them. These individual legs of the race are our half-reactions. Simple, right?
We've got two main players in this electron drama:
- Oxidation: This is where a substance loses electrons. Think of it as "LEO" – Lose Electrons Oxidation. It's like someone saying, "Here, you can have these!" and giving away their precious electrons.
- Reduction: This is the flip side – where a substance gains electrons. Remember "GER" – Gain Electrons Reduction. These guys are the electron hoarders, happily accepting what's offered.
Together, these two processes – oxidation and reduction – make up what we call a redox reaction. And in a galvanic cell, we separate these two halves to force the electrons to travel through an external circuit. That's where the electricity comes from! It's like saying, "Okay, you guys, play nice, but you have to do it through this wire."
Picking Your Players: Choosing the Right Half-Reactions
Now, here’s the fun part: designing your own cell! It’s all about picking the right chemical reactions. You can’t just throw any old reactions together and expect sparks to fly (unless you're making fireworks, which is a whole different ballgame). You need a pair that are willing to play the electron transfer game.
How do we know which ones will play nice? We use something called the standard reduction potential. Don’t let the fancy name scare you. Think of it as a score for how much a substance wants to gain electrons. The higher the score (more positive value), the more it wants to be reduced. The lower the score (more negative value), the more it wants to be oxidized. It’s like a popularity contest for electrons!
The Golden Rule of Galvanic Cells
Here’s the golden rule, the secret handshake of galvanic cell design: You need to pair a substance that wants to be oxidized with a substance that wants to be reduced. More specifically, you want to pair a half-reaction with a lower reduction potential (the one that’s more likely to give up electrons) with a half-reaction with a higher reduction potential (the one that’s more likely to grab them).
Think of it this way: you have a generous donor (oxidation) and an eager receiver (reduction). Without both, your electron relay race grinds to a halt. It's like trying to play catch with only one person – it just doesn't work!
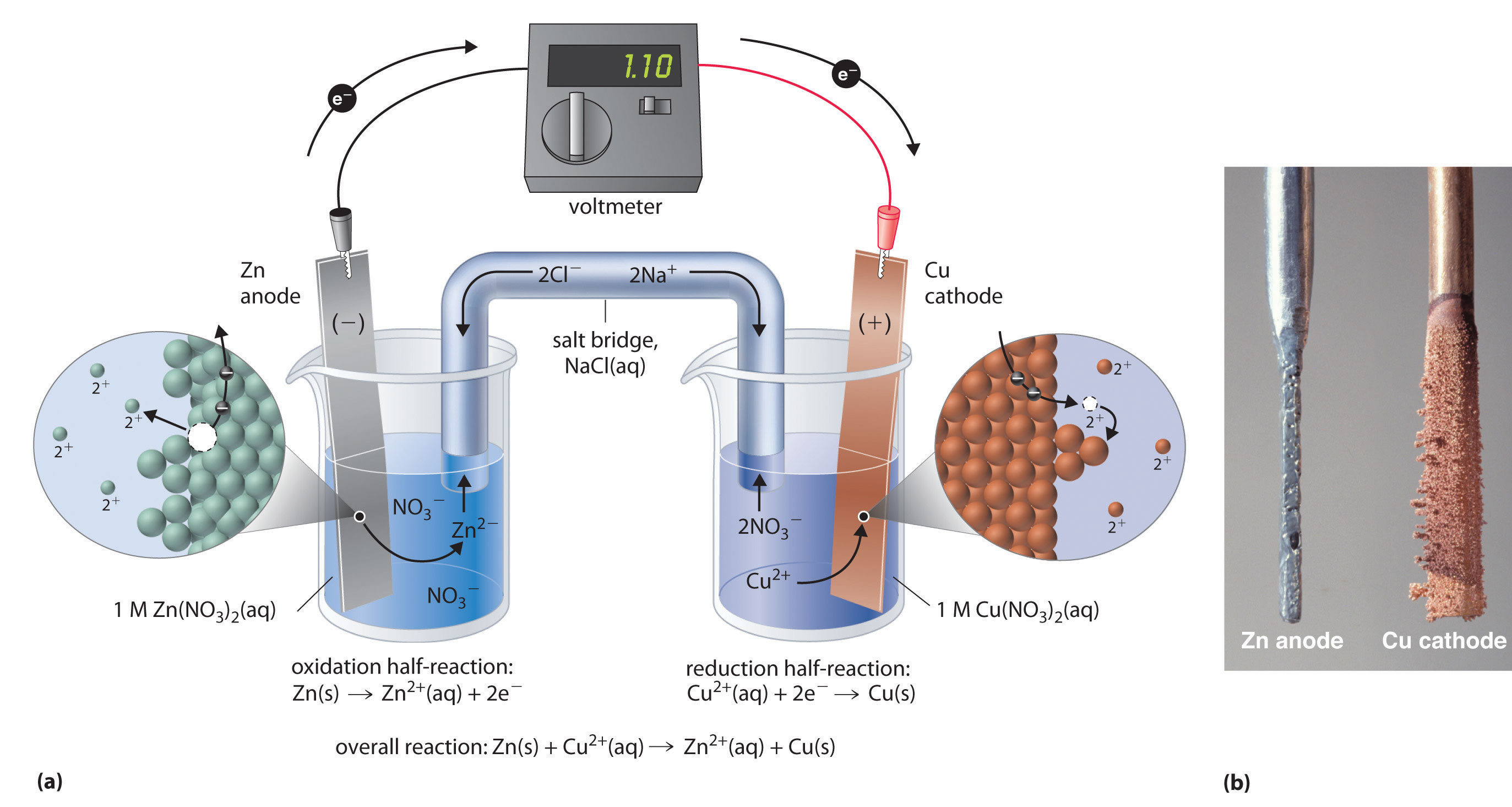
Let's Build a (Conceptual) Daniell Cell!
To make this concrete, let’s whip up a classic: the Daniell cell. It's like the vanilla ice cream of galvanic cells – reliable and universally understood.

We're going to use two common metals: Zinc (Zn) and Copper (Cu).
First, we need to consult our trusty reduction potential chart (or just remember these common ones). Here's what we find:
- Cu²⁺(aq) + 2e⁻ → Cu(s) E° = +0.34 V (This guy really wants electrons!)
- Zn²⁺(aq) + 2e⁻ → Zn(s) E° = -0.76 V (This guy is a bit more chill about giving them up.)
See the difference? Copper ions (Cu²⁺) have a much higher reduction potential (+0.34 V) than zinc ions (Zn²⁺) (-0.76 V). This tells us that copper ions are much more likely to grab electrons and become solid copper metal. And conversely, solid zinc metal is more likely to give up its electrons and become zinc ions.
So, for our galvanic cell, we’ll make zinc metal the anode (where oxidation happens) and copper ions the cathode (where reduction happens).
Setting Up the Stage
To make this work, we need two separate containers, or half-cells.
Half-Cell 1 (The Oxidation Station):
- We'll have a strip of solid zinc metal (Zn(s)).
- This strip will be dipped into a solution containing zinc ions (Zn²⁺(aq)), like zinc sulfate (ZnSO₄).
- Here, the zinc metal will undergo oxidation: Zn(s) → Zn²⁺(aq) + 2e⁻. Our zinc is happily shedding electrons and dissolving into the solution!
Half-Cell 2 (The Reduction Station):
- We'll have a strip of solid copper metal (Cu(s)).
- This strip will be dipped into a solution containing copper ions (Cu²⁺(aq)), like copper sulfate (CuSO₄).
- Here, the copper ions from the solution will grab the electrons: Cu²⁺(aq) + 2e⁻ → Cu(s). The copper ions are getting plated onto our copper strip, making it bigger and better!
Connecting the Dots: The Salt Bridge and the Wire
Now, the electrons are zipping away from the zinc and heading towards the copper. But we need a way for them to travel from one half-cell to the other. That's where the external circuit comes in – a simple wire connecting the zinc electrode to the copper electrode. Electrons flow through this wire, and voilà, we have electricity!
But wait, there's another crucial piece of the puzzle: the salt bridge. Imagine our two half-cells are like separate islands. If we just connect them with a wire, eventually, one side will get too positively charged (from losing electrons) and the other too negatively charged (from gaining them). This charge imbalance will grind the reaction to a halt. Annoying, right?
The salt bridge is our peacemaker. It's usually a U-shaped tube filled with a concentrated solution of an inert electrolyte, like potassium nitrate (KNO₃) or ammonium chloride (NH₄Cl). It's sealed with porous plugs so the solution doesn't just pour out, but ions can slowly move across.
Here's its superhero job: It allows ions to migrate between the two half-cells to maintain electrical neutrality. In the anode compartment (where oxidation happens), there's a buildup of positive Zn²⁺ ions, so negative ions (anions) from the salt bridge flow in to balance things out. In the cathode compartment (where reduction happens), there's a depletion of positive Cu²⁺ ions, so positive ions (cations) from the salt bridge flow in to keep the positive charge balanced.
It’s like a tiny traffic controller for ions, ensuring the electron flow can continue smoothly. Without it, our galvanic cell would be a one-hit wonder.
Putting It All Together: The Overall Reaction
So, we have our oxidation half-reaction at the anode:
Zn(s) → Zn²⁺(aq) + 2e⁻

And our reduction half-reaction at the cathode:
Cu²⁺(aq) + 2e⁻ → Cu(s)
To get the overall reaction for our galvanic cell, we add these two half-reactions together. Notice how the electrons (2e⁻) cancel out – they’re gained by one and lost by the other, so they don't appear in the final equation. This gives us:
Zn(s) + Cu²⁺(aq) → Zn²⁺(aq) + Cu(s)
This is the grand finale! When zinc metal meets copper ions (indirectly, through the magic of electricity and a salt bridge), zinc becomes zinc ions, and copper ions become solid copper. And in the process, electrons have flowed, creating an electrical current!
Calculating the Voltage (The "Oomph" Factor)
We can even predict how much "oomph" our battery will have – its voltage! We do this by subtracting the standard reduction potential of the anode (the one being oxidized) from the standard reduction potential of the cathode (the one being reduced).
E°cell = E°cathode - E°anode

For our Daniell cell:
E°cell = (+0.34 V) - (-0.76 V) = +1.10 V
So, our magnificent Daniell cell has a theoretical voltage of 1.10 volts. That's the electrical pressure pushing those electrons along!
Beyond the Daniell Cell: Endless Possibilities!
The amazing thing about this is that you can swap out zinc and copper for countless other metal combinations (and even other types of reactions). As long as you have a substance that’s willing to be oxidized and another that’s eager to be reduced, you can design a galvanic cell. Want a higher voltage? Pick half-reactions with a bigger difference in their reduction potentials. Want a different application? Explore reactions that produce specific products or require specific voltages.
It's like having a recipe book for electricity. You just pick your ingredients (half-reactions) and follow the steps (connecting them properly), and you can whip up your own power source.
So, the next time you pick up your phone or flick a light switch, take a moment to appreciate the ingenious dance of electrons happening inside. You've just learned the fundamental principles behind that magic. You’ve peeked behind the curtain and seen the chemistry that powers your world!
Designing a galvanic cell from two half-reactions isn't just about memorizing formulas; it’s about understanding a fundamental principle of how energy can be harnessed from chemical reactions. It’s about the beautiful, ordered flow of electrons, driven by the natural inclination of certain substances to gain what others are willing to give. And the best part? You can now look at any battery, from the tiny one in your watch to the massive ones in electric cars, and know you understand the core idea at play. So go forth, be curious, and remember that even the smallest chemical interactions can lead to the most electrifying results. Keep exploring, keep experimenting, and keep that scientific spark alive! You’ve got this!
