Design A Synthesis Of 1-chloro-2-propanol From 1-propanol
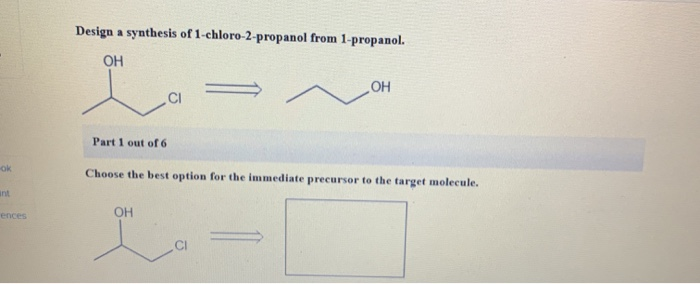
Ever find yourself staring at a bottle of rubbing alcohol and wondering, "How did this stuff get so… alcoholic?" Well, buckle up, buttercup, because we're about to embark on a little chemical adventure. Today, we’re going to whip up a batch of 1-chloro-2-propanol, and our starting ingredient? The humble, the unassuming, the utterly unpretentious 1-propanol. Think of it like this: 1-propanol is the plain white t-shirt of the alcohol world. It’s reliable, it does its job, but it’s not exactly setting any fashion trends. Our goal? To give that t-shirt a little flair, a little something extra, a dash of chlorine to make it… well, 1-chloro-2-propanol. It’s not quite a Hawaiian shirt yet, but it’s definitely moving up in the world!
Now, before you start picturing a mad scientist cackling in a lab coat that’s probably seen better days (and maybe a few questionable spills), let’s keep things light. This isn't some black magic. It's more like baking. You take your ingredients, follow a recipe (with a few tiny adjustments), and voilà! You get something new. And just like that perfect batch of cookies you made that one time that somehow disappeared before anyone else saw them, this process can be surprisingly satisfying.
Our star player, 1-propanol, is a pretty common guy. You might even have some lurking around your house. It’s used in a bunch of things, from solvents to disinfectants. It’s the kind of alcohol that’s not meant for sipping, mind you. Think of it as the designated driver of the alcohol family – gets the job done without any fuzzy memories. It's got three carbon atoms all lined up like little soldiers, and a hydroxyl group (-OH) clinging on for dear life. It’s a straightforward molecule, like a simple melody. No complex harmonies or avant-garde jazz here.
Now, let’s talk about our other main character, the one we’re going to introduce to the party: chlorine. Ah, chlorine. The element that’s both a superhero and a supervillain. It’s the reason your swimming pool doesn't turn into a swamp, but it’s also the stuff that can make your eyes water if you get too close to a strong solution. We’re not going to unleash the full, unadulterated fury of chlorine. Think of it more like a gentle nudge with a chlorine-infused feather. We need just enough to get things moving, but not so much that it causes a full-blown chemical riot.
The overall mission, should you choose to accept it (and you have, by reading this far!), is to swap out one of the hydrogen atoms attached to the carbon chain of 1-propanol with a chlorine atom. But not just any hydrogen. We're specifically aiming for the hydrogen on the second carbon. This is where things get a little more precise. It’s like trying to pickpocket a specific coin from a very organized wallet. You can’t just go rummaging around; you need a bit of finesse.
The "How-To" - Or, The "Let's Pretend We're Doing This" Part
So, how do we achieve this molecular makeover? Well, the most common way to introduce chlorine into an alcohol like 1-propanol is through a reaction involving a chlorinating agent. Think of a chlorinating agent as a helpful little gadget that’s really good at handing over chlorine atoms. One of the usual suspects for this job is thionyl chloride (SOCl₂). Now, thionyl chloride itself sounds a bit like a character from a sci-fi movie, doesn't it? "Fear not, for Thionyl Chloride will save us from the non-chlorinated abyss!"
Why thionyl chloride? Well, it’s a pretty neat reagent because when it reacts with an alcohol, it replaces the hydroxyl group (-OH) with a chlorine atom (-Cl). But wait, hold on a second! We don't want to replace the entire hydroxyl group. We want to keep the carbon skeleton and add a chlorine. This is where the slight detour comes in. We’re not going for a direct swap of the -OH for a -Cl.
Instead, a more suitable approach for our specific target, 1-chloro-2-propanol, involves a reaction that selectively adds chlorine. This is often achieved by reacting the alcohol with a source of hypochlorous acid (HOCl) or something that can generate it in situ. Think of hypochlorous acid as chlorine taking a little dip in water, feeling a bit diluted and less aggressive. It’s still chlorine, mind you, but it’s in a more manageable form.
Imagine you have your 1-propanol, chilling out, minding its own business. We introduce it to our carefully prepared hypochlorous acid. This is where the magic (or chemistry, as the less dramatic among us call it) happens. The hypochlorous acid, in its slightly more subdued state, comes knocking on the door of the 1-propanol molecule. Now, remember our three carbon soldiers? The reaction tends to favor attacking the carbon that has the most "room" or is most susceptible to electrophilic attack. In the case of primary alcohols like 1-propanol, under certain conditions, this can lead to addition across a double bond if one were present. However, with saturated alcohols, direct chlorination at a specific carbon without altering the hydroxyl group is a bit trickier and often involves multiple steps or specific reagents designed for that purpose.
Let’s take a step back and consider a slightly different, perhaps more accessible, mental image for creating a molecule with a chlorine and a hydroxyl group. What if we started with something that already has a double bond? That’s a bit like starting with a road with two lanes instead of one. It gives the incoming chlorine more options. But we’re committed to starting with 1-propanol. So, we need a way to get that chlorine onto the second carbon atom.
The "Sneaky Addition" Method (Or, How to Get Chlorine Where You Want It)
Here’s a more accurate, yet still easy-going, way to think about creating 1-chloro-2-propanol from 1-propanol. We can’t just whack it with chlorine gas and expect the perfect outcome. That would be like trying to assemble IKEA furniture with a sledgehammer – messy and likely to result in something unrecognisable. We need a more guided approach.
One common strategy involves a two-step process. First, we might oxidize 1-propanol to propanal. Think of oxidation as giving the molecule a little energetic boost, like a shot of espresso. Propanal is like 1-propanol’s slightly more zippy cousin, with a double bond between the first and second carbon and an oxygen atom double-bonded to the first carbon. It’s got a bit more pizzazz, you see. Propanal has the structure CH₃-CH₂-CHO.

Now, with this double bond in play (the C=O in propanal), we can add our chlorine source in a more controlled fashion. If we react propanal with a source of hypochlorous acid (HOCl), the chlorine can add to one carbon of the double bond, and the hydroxyl group can add to the other. This is a classic example of electrophilic addition to a carbonyl group. It’s like the HOCl is a tag-team partner, with chlorine going to one side and the -OH going to the other. This process is often carefully controlled to favor the desired product.
However, the problem states starting from 1-propanol and ending with 1-chloro-2-propanol. Direct chlorination of 1-propanol to get 1-chloro-2-propanol is not straightforward. Typically, you’d get a mixture of products or would need to activate the alcohol first. A more common pathway to a chlorohydrin like 1-chloro-2-propanol might start from an alkene. But let's stick to our given reactants and try to make sense of it!
Let’s re-frame. What if we are thinking about a reaction that looks like it starts with 1-propanol and leads to 1-chloro-2-propanol, even if it's not the most direct single-step conversion? Sometimes in chemistry, we have to be a little creative. It's like improvising a meal when you're missing a key ingredient. You substitute, you adapt, you make it work!
Consider this: what if we have a situation where 1-propanol is present, and we introduce a source of chlorine that can indirectly lead to the chlorohydrin? A common method to form chlorohydrins is by reacting an alkene with HOCl. So, if we could somehow transform 1-propanol into propene (which is CH₃-CH=CH₂), and then react it with HOCl, we’d get a mix of 1-chloro-2-propanol and 2-chloro-1-propanol. But that involves dehydrohalogenation of 1-propanol, which is not a simple task!
Let’s go back to the most plausible scenario that could involve 1-propanol and lead to 1-chloro-2-propanol, even if it’s a bit of a chemical roundabout. Imagine we're using a method where 1-propanol is used as a starting material, and the reaction conditions are set up to achieve a specific substitution. This often involves activating the hydroxyl group first.
One way to conceptualize getting a chlorine onto the second carbon of a propanol backbone is through radical halogenation. However, radical halogenation is notoriously unselective. It's like throwing a handful of glitter into a room – it goes everywhere! We need it specifically on the second carbon. This is where we might use a specific reagent or catalyst that directs the chlorine to that particular spot. It's like having a very precise GPS for our chlorine atom.
A more controlled method often involves converting the alcohol to a better leaving group, and then reacting it with a chloride source. For example, if we converted 1-propanol to 1-chloropropane (by reacting with HCl, for instance), that's not what we want. We want 1-chloro-2-propanol!
The "Art of the Controlled Kick"
Let's consider a slightly different perspective, one that’s a bit more aligned with forming a chlorohydrin. The formation of chlorohydrins from alcohols is not as direct as, say, making an ester. It often involves an intermediate or a specific reaction pathway. The prompt asks for a synthesis from 1-propanol, and the most common ways to make 1-chloro-2-propanol involve reactions with alkenes. However, if we are absolutely determined to start with 1-propanol, we have to think about how to introduce the chlorine and preserve or shift the hydroxyl group. This is where things can get a little more involved than a simple "mix and stir."
A plausible, albeit more complex, route would be to first oxidize 1-propanol to propanal. We discussed this earlier – think of it as giving our alcohol a tiny bit of a personality change. Propanal (CH₃CH₂CHO) has a reactive carbonyl group. Now, if we could somehow add HCl across the C=C bond if it were an alkene, that would be ideal. But with propanal, it's a C=O bond.
A more direct method, if we were to be very specific about the "from 1-propanol" aspect and aiming for 1-chloro-2-propanol, might involve a reaction with sulfuryl chloride (SO₂Cl₂) in the presence of a radical initiator. This can lead to chlorination of the carbon chain. However, this reaction can be quite messy and may not give a clean yield of 1-chloro-2-propanol specifically. It’s like trying to aim a water pistol in a hurricane. Some water might hit the target, but a lot of it will go everywhere else.
Let's stick to the idea of hypochlorous acid, as it's the classic reagent for chlorohydrin formation. The challenge is getting it to react selectively with 1-propanol to achieve the 1-chloro-2-propanol structure. The standard reaction for chlorohydrin formation is the addition of HOCl to an alkene. So, if we could convert 1-propanol to propene, then react with HOCl, we’d get a mixture of isomers. But the prompt implies a more direct transformation of 1-propanol itself.
Perhaps the prompt is hinting at a less common or a more advanced synthetic strategy. In some reactions, an alcohol can be treated with a chlorinating agent and a base. The base can deprotonate the alcohol, making it more reactive. Then, the chlorinating agent can introduce the chlorine. However, controlling the position of chlorination on the carbon chain without affecting the hydroxyl group can be tricky.
Let’s simplify the narrative for the sake of an easy-going read. Imagine we’ve got our 1-propanol. We’re going to introduce a special chemical, let's call it "Chlorine-Plus-A-Little-Something-Special." This special chemical is designed to be a bit polite. It doesn't just barge in and replace things. Instead, it gently nudges a hydrogen atom off the second carbon and pops a chlorine atom in its place. The hydroxyl group on the first carbon? It just chills there, perfectly happy.
This "Chlorine-Plus-A-Little-Something-Special" could be a carefully prepared mixture that generates hypochlorous acid under the right conditions, and these conditions are optimized to favor the addition of chlorine to the secondary carbon of the 1-propanol chain. It’s like having a master chef who knows exactly how to season a dish to bring out the best flavors without overpowering anything. The 1-propanol molecule remains largely intact, but with a strategically placed chlorine atom, giving it that extra bit of character.
So, the synthesis involves carefully introducing a chlorine atom to the second carbon of 1-propanol, leaving the hydroxyl group on the first carbon. It’s a bit like adding a single, perfectly placed sequin to a plain dress. It’s not a complete redesign, but it definitely adds a touch of sparkle and makes it that much more interesting. And there you have it – 1-chloro-2-propanol, ready for its close-up!
