Cyclohexane And 1-hexene Are Constitutional Isomers
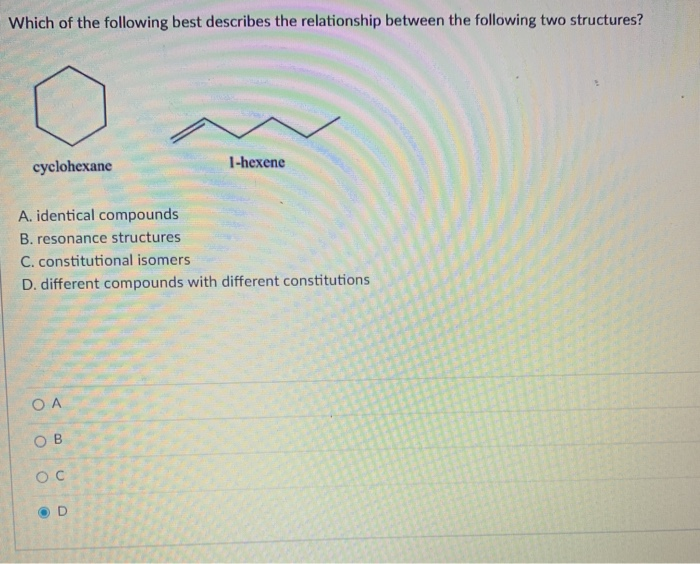
Hey there, science fans! Ever thought about molecules having doppelgangers? Well, buckle up, because we're diving into the totally cool world of cyclohexane and 1-hexene. They might sound a bit fancy, but trust me, this is where the magic happens in chemistry, and it's more fun than you might think!
So, picture this: you've got two molecules. On one hand, we have cyclohexane. Imagine a ring. A perfect little circle made of six carbon atoms, all holding hands with hydrogen atoms. It's like a tiny, closed bicycle frame, but made of atoms. It’s neat, it’s tidy, and it’s complete. Think of it as a closed loop of fun, where everything is connected and happy.
Now, let's swing over to our other star player: 1-hexene. This one is a bit more of a free spirit. Instead of a neat circle, it’s a straight chain. Six carbon atoms in a row, like a little train. But here's the kicker, the thing that makes it super interesting: right at the beginning of this chain, there’s a special little party happening. It’s a double bond between two of the carbon atoms. This double bond is like a handshake, but with a little extra vigor, holding hands a bit tighter. It’s a place where things can get exciting, where reactions love to happen. It’s the energetic teenager of the hydrocarbon family!
So, what’s the big deal, right? They both have six carbon atoms. They both have a bunch of hydrogen atoms too. If you were to count them up, they actually have the exact same number of atoms, and the exact same types of atoms. Mind. Blown. Yet, they look and act so different! This is where the term constitutional isomers comes into play. It's a fancy way of saying they are like twins, but they grew up with very different personalities. They have the same ingredients, but they're put together in a different order, leading to totally different outcomes.
It's like having the same set of Lego bricks. You could build a cool, sturdy castle (that’s kind of like cyclohexane – stable, self-contained), or you could build a speedy race car with some parts sticking out and ready to zoom (that's your 1-hexene – a bit more reactive and ready for action).

What makes this so entertaining is the sheer cleverness of nature (and chemistry!). It shows us that the arrangement of atoms is just as important as the atoms themselves. A tiny tweak in how things are connected can lead to a whole new molecule with entirely new properties. It’s like how rearranging letters can turn "listen" into "silent." Same letters, different meaning. Same atoms, different molecule!
Cyclohexane, with its closed ring, is pretty stable. It’s like the dependable friend who’s always there, not easily swayed. It’s great for things like solvents, where you need something that won't easily react and mess things up. It’s the quiet achiever.

On the other hand, that double bond in 1-hexene is a hotbed of activity. It's like the life of the party, always ready to mingle and make new connections. This double bond makes 1-hexene much more reactive. It’s eager to join in chemical reactions, to break that double bond and form new attachments. This makes it super useful as a building block for making other, more complex chemicals. Think of it as the versatile ingredient that can be used to create all sorts of delicious dishes.
It's the subtle difference in structure that unlocks a universe of possibilities!
Hexene Isomers
This concept of constitutional isomers is everywhere in chemistry. It’s what makes life so diverse! Different arrangements of the same basic building blocks (like carbon and hydrogen) lead to everything from the plastics in your phone to the fuels that power your car, and even the complex molecules that make up your own body. It's a fundamental principle that fuels innovation and understanding.
So, the next time you hear about cyclohexane and 1-hexene, don't just think of them as boring chemical names. Think of them as a perfectly formed ring and a lively chain, two sides of the same atomic coin. They are a fantastic example of how just a little bit of rearrangement can lead to a whole new world of properties and uses. It’s a simple idea, but it’s incredibly powerful and, dare I say, even a little bit magical. It's a reminder that even in the smallest things, there’s always something fascinating to discover if you just look a little closer!
Isn't it amazing how much difference a single bond can make? It’s this kind of elegant simplicity and boundless creativity that makes chemistry so captivating. So, keep your eyes peeled for more molecular marvels. You never know what wonders are hiding in plain sight, just waiting for you to notice them!

