Crenation Is Likely To Occur In Blood Cells Immersed In

Hey there, curious minds! Ever wonder what happens when you mix things up in the world of tiny, microscopic stuff? Today, we're diving into something super neat that happens to our amazing blood cells. It's called crenation, and honestly, it sounds a bit like a spooky spell, doesn't it? But don't worry, it's all about science, and it's pretty darn cool when you think about it.
So, what's the big deal with crenation? Well, it's all about what happens when our little blood cells, those tiny powerhouses that keep us going, find themselves in a bit of a pickle. Specifically, it's likely to occur in blood cells immersed in... are you ready for it?... a hypertonic solution. Ooh, fancy words! But what does that even mean? Let's break it down, nice and easy.
Imagine a Tiny Balloon!
Think of a blood cell like a tiny, delicate balloon. It's got this thin outer layer, a membrane, that holds all its important insides together. Now, inside this balloon (our blood cell), there's a certain concentration of stuff – salts, sugars, proteins, you name it. This is its normal, comfy environment.
But what happens if you suddenly plop this little balloon into a solution that has way more of that "stuff" – like a super-concentrated sugary syrup, or a really salty bath?
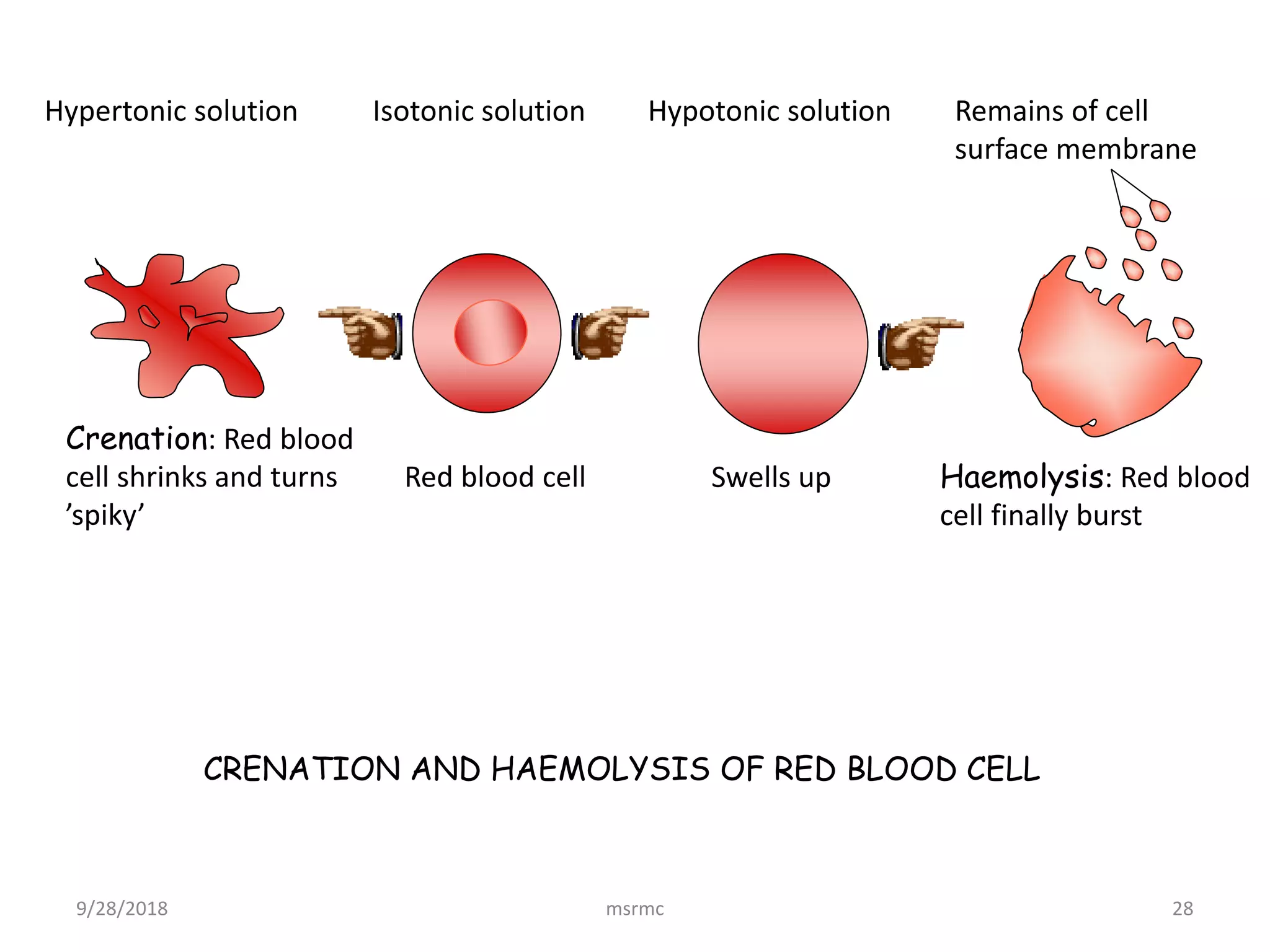
This is where our hypertonic friend comes in. A hypertonic solution is basically a solution with a higher concentration of solutes (those dissolved "stuff" particles) than what's inside our blood cell. So, our blood cell is sitting in a solution that's more... shall we say, crowded with dissolved particles than its own internal environment.

The Great Escape (or In-Take?)
Now, here's the truly fascinating part. Nature, in its infinite wisdom, loves balance. Things tend to move from where there's a lot of them to where there's less of them. This is a fundamental principle called osmosis. It's like when you open a bottle of perfume in one corner of a room, and pretty soon, the whole room smells nice. The perfume molecules are moving!
In the case of our blood cell and the hypertonic solution, water is the main mover. Remember that membrane around our blood cell? It's semi-permeable. That means it lets some things through, but not others. Water is pretty good at squeezing through these membranes.
Because the hypertonic solution has a higher concentration of solutes outside the cell, it means there's a lower concentration of water outside the cell compared to inside. So, what does our watery friend, water, do? It wants to even things out! It will start to move out of the blood cell and into the surrounding hypertonic solution.

The Shrinking Show!
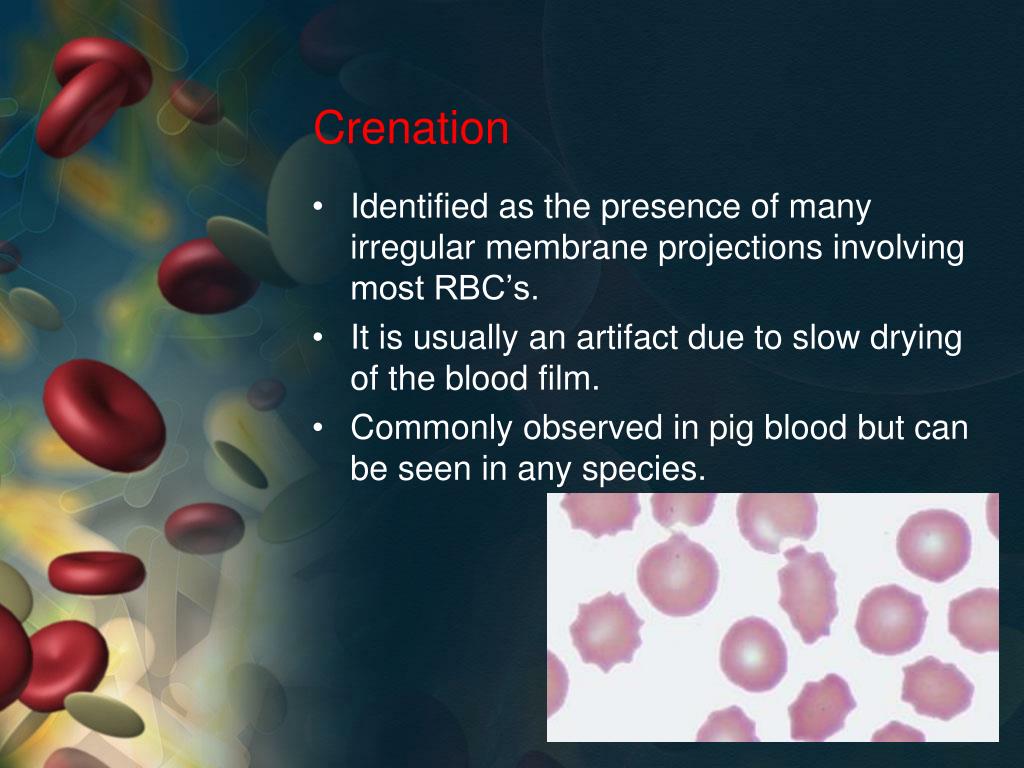
And what happens when water starts leaving our little balloon-like blood cell? It starts to shrink. Imagine deflating a balloon – it gets smaller, right? But it's not just a smooth deflation. As the water pulls away, the cell membrane starts to pull inwards, creating little spiky, irregular edges.
This, my friends, is crenation! The cell literally looks like it's shriveling up, developing those little nooks and crannies, almost like a tiny, dried-up raisin. It’s not a pretty sight for the cell, but it’s a beautiful illustration of how cells respond to their environment. Think of a grape left out in the sun – it turns into a raisin, right? Similar concept, just on a microscopic level and driven by water movement.
Why is This Even Interesting?
You might be thinking, "Okay, so cells shrink. Big deal." But it's actually a really important concept in a few areas:
Medical Applications & Diagnostics
In the medical world, understanding how blood cells behave in different solutions is crucial. When doctors or lab technicians collect blood samples, they need to handle them carefully. If blood is stored in a solution that's too concentrated (hypertonic), those red blood cells will crenate. This can mess with tests that rely on the normal size and shape of blood cells, like those that count them or analyze their properties.
Imagine a doctor trying to diagnose a problem based on how many healthy-looking cells there are, but half of them have shriveled up due to improper handling. It would be like trying to count apples in a basket, but a third of them have been squished into raisins – not ideal!
Cell Biology Research
For scientists studying cells, controlling the environment is key. If they want to observe cells in a stable state, they need to make sure the surrounding solution is isotonic – meaning it has the same concentration of solutes as the cell itself. This way, water doesn't move in or out, and the cell stays happy and plump. By intentionally creating hypertonic conditions, researchers can study how cells respond to stress or dehydration, giving us insights into cell survival and damage.
Understanding Body Processes
Even within our own bodies, subtle changes in the concentration of fluids can occur. While our bodies are usually very good at keeping things balanced, understanding osmotic principles like crenation helps us grasp how cells would react if that balance were to shift. It’s a reminder of the delicate equilibrium that keeps our biological systems functioning smoothly.
A Tiny Battle of Wills
So, next time you hear the word "crenation," don't picture something gruesome. Picture a tiny, microscopic battle of wills between water and dissolved particles. It’s the cell’s membrane, designed to protect its internal world, being pushed and pulled by the invisible forces of osmosis. It’s a testament to the amazing adaptability of life at its smallest scale.
It's a quiet drama playing out in every drop of blood that’s not handled just right, a gentle reminder that even the most fundamental processes of life are governed by elegant, natural laws. Pretty cool, right? Just remember, if you ever see blood cells looking a bit shriveled, it's a sign they've been chilling in a solution that's a little too intense for their liking!
