Counting Sigma And Pi Bonds In A Small Molecule
Hey there, fellow science explorers! Ever look at a molecule and wonder what makes it tick? You know, those tiny building blocks of everything around us? Well, today we’re going to peek under the hood of some small molecules and get a little curious about something called sigma bonds and pi bonds. Don’t let the fancy names scare you; it’s actually pretty neat and not nearly as complicated as it sounds. Think of it like figuring out the secret handshake of atoms!
So, what exactly are these bonds? In chemistry, bonds are like the invisible glue that holds atoms together to form molecules. They’re the reason water is H₂O and not just a jumbled mess of hydrogen and oxygen atoms floating around. And within these bonds, there are different types. We're going to focus on the two most common players: sigma ($\sigma$) and pi ($\pi$) bonds.
Imagine atoms as little kids holding hands. A sigma bond is like the most basic, solid hug. It's formed when two atoms directly share electrons, kind of like a firm handshake, right in the middle, along the imaginary line connecting their centers. This is the strongest type of covalent bond, the reliable workhorse of molecular structure. Every single bond between two atoms in a molecule, no matter if it's a single, double, or triple bond, must have at least one sigma bond. It’s the foundation, the bedrock, the essential handshake that keeps the atoms from drifting apart.
Think of it like this: when you're building with LEGOs, the basic brick-to-brick connection is your sigma bond. It’s what holds everything together fundamentally. Without it, you wouldn’t have any structure at all. It’s all about direct, head-on overlap of atomic orbitals. Super straightforward, super important.
Now, where do pi bonds come in? Well, sometimes, atoms get a little more… enthusiastic with their bonding. If two atoms already have that essential sigma bond handshake, and they have leftover electrons they want to share, they can form a pi bond. Unlike the direct, head-on sigma bond, a pi bond is formed by the side-by-side overlap of atomic orbitals. Imagine two kids, after their firm handshake, deciding to high-five or give each other a friendly nudge from the side. That’s kind of what a pi bond is like.
These pi bonds aren't as strong as sigma bonds, but they add extra stability and, interestingly, they’re responsible for a lot of the cool chemistry we see, like reactions and the way molecules interact. They're like the fancy flourishes on a signature, adding extra flair and complexity. You’ll only find pi bonds as part of double bonds and triple bonds. A double bond is made of one sigma bond and one pi bond. A triple bond is even more impressive: one sigma bond and two pi bonds!

So, if a single bond is just a handshake, a double bond is a handshake and a high-five, and a triple bond is a handshake, a high-five, and a fist bump! Getting the picture?
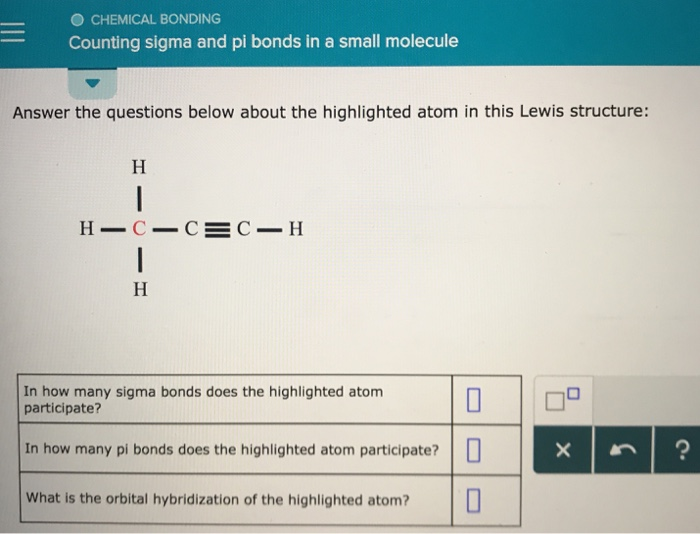
Okay, ready to put on our detective hats and count these bonds in some small, familiar molecules? Let’s start with something super simple: methane. You know, CH₄. It’s the main ingredient in natural gas. If we draw it out, we see a central carbon atom bonded to four hydrogen atoms.
Now, remember our rule: every bond has at least one sigma bond. So, between the carbon and each of the four hydrogen atoms, there’s a sigma bond. That gives us a total of four sigma bonds in methane. Since all the bonds are single bonds, there are no extra electron-sharing parties happening, so there are zero pi bonds. Easy peasy!
Let’s level up a bit. How about ethene (also known as ethylene)? This is C₂H₄. This molecule is a bit more exciting because it has a double bond between the two carbon atoms. So, let’s break it down. We have two carbon atoms. They are bonded to each other with a double bond. And each carbon atom is also bonded to two hydrogen atoms. So, we have:
First, the bond between the two carbon atoms. A double bond, remember, is one sigma bond and one pi bond. So, that’s 1 sigma bond and 1 pi bond right there.
Then, we have the bonds between each carbon and its two hydrogen atoms. These are single bonds, so they are each sigma bonds. There are four of these (two on each carbon). So, that’s another 4 sigma bonds.
Add it all up! For sigma bonds, we have 1 (between carbons) + 4 (between carbon and hydrogen) = 5 sigma bonds. And for pi bonds, we have just the 1 pi bond from the carbon-carbon double bond. Pretty cool how that double bond introduces a whole new type of interaction!
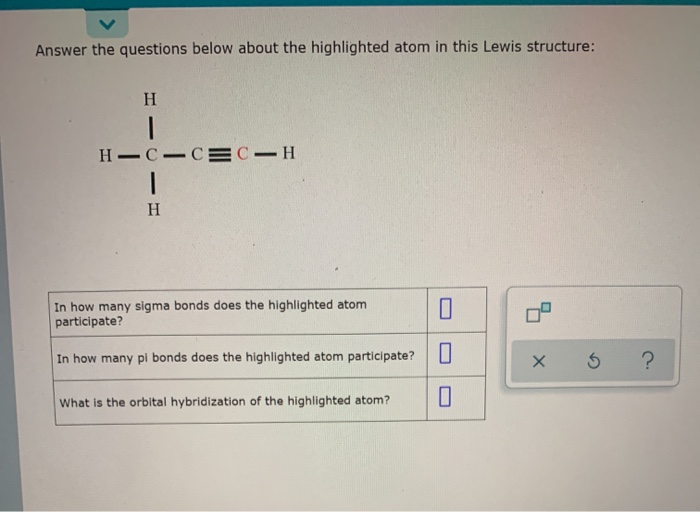
Let’s try one more: ethyne (also known as acetylene). This is C₂H₂. This one is really exciting because it has a triple bond between the two carbon atoms! This is where things get a bit more crowded in the bonding department.
So, again, we have two carbon atoms. They are linked by a triple bond. And each carbon atom is also bonded to just one hydrogen atom.
The triple bond between the two carbon atoms is our star here. A triple bond, as we learned, is made of 1 sigma bond and 2 pi bonds. Wowza!
Then, we have the bonds between each carbon atom and its single hydrogen atom. These are simple single bonds, so they are each sigma bonds. There are two of these (one on each carbon). So, that’s another 2 sigma bonds.
Let’s tally them up: For sigma bonds, we have 1 (from the triple bond) + 2 (between carbon and hydrogen) = 3 sigma bonds. And for pi bonds, we have 2 pi bonds from that impressive triple bond! It’s like a triple high-five with a handshake!
Why is this even interesting, you ask? Well, understanding these bonds helps chemists predict how molecules will behave. For instance, molecules with pi bonds are often more reactive. They’re more likely to participate in chemical reactions, like when you burn fuel or when your body digests food. The pi bonds are the more flexible, more accessible electron clouds that other molecules can “grab onto.”
Think of it like a dance. Sigma bonds are the steady, reliable steps that keep the dancers together. Pi bonds are the more dynamic, flashy moves that make the dance interesting and allow for interactions with other dancers. Without the sigma backbone, there's no dance. But without the pi flourishes, the dance might be a bit… boring and less likely to involve anyone else.
So, the next time you see a chemical formula, you can now imagine the hidden world of sigma and pi bonds holding it all together. It’s a fantastic way to appreciate the intricate beauty and functionality of even the smallest bits of the universe. It’s like having a secret code to understanding how the world is put together, one bond at a time!
