Could Two Atoms Of Oxygen Engage In Ionic Bonding

Hey there, science enthusiasts and curious minds! Ever found yourself staring at your water bottle and wondering, "What's really going on in there?" Or maybe you've seen the periodic table and thought, "So many elements, so little time to figure them all out!" Well, today we're diving into a particularly neat corner of chemistry that might sound a bit... electric. We're talking about atoms, those tiny building blocks of everything, and specifically, the intriguing question: Could two atoms of oxygen ever get together through ionic bonding?
It's a question that sparks a lot of interest, and honestly, it's got a bit of a "gotcha!" element to it, which makes it even more fun to explore. Think of atoms like tiny little characters, each with their own personalities and desires. Some are super eager to share, others are a bit more greedy, and some just want to find a stable, happy home. Ionic bonding is like one atom saying, "Hey, you have something I really need, and I'm willing to give you something you might want in return!" It's a bit of a give-and-take, a transaction of sorts.
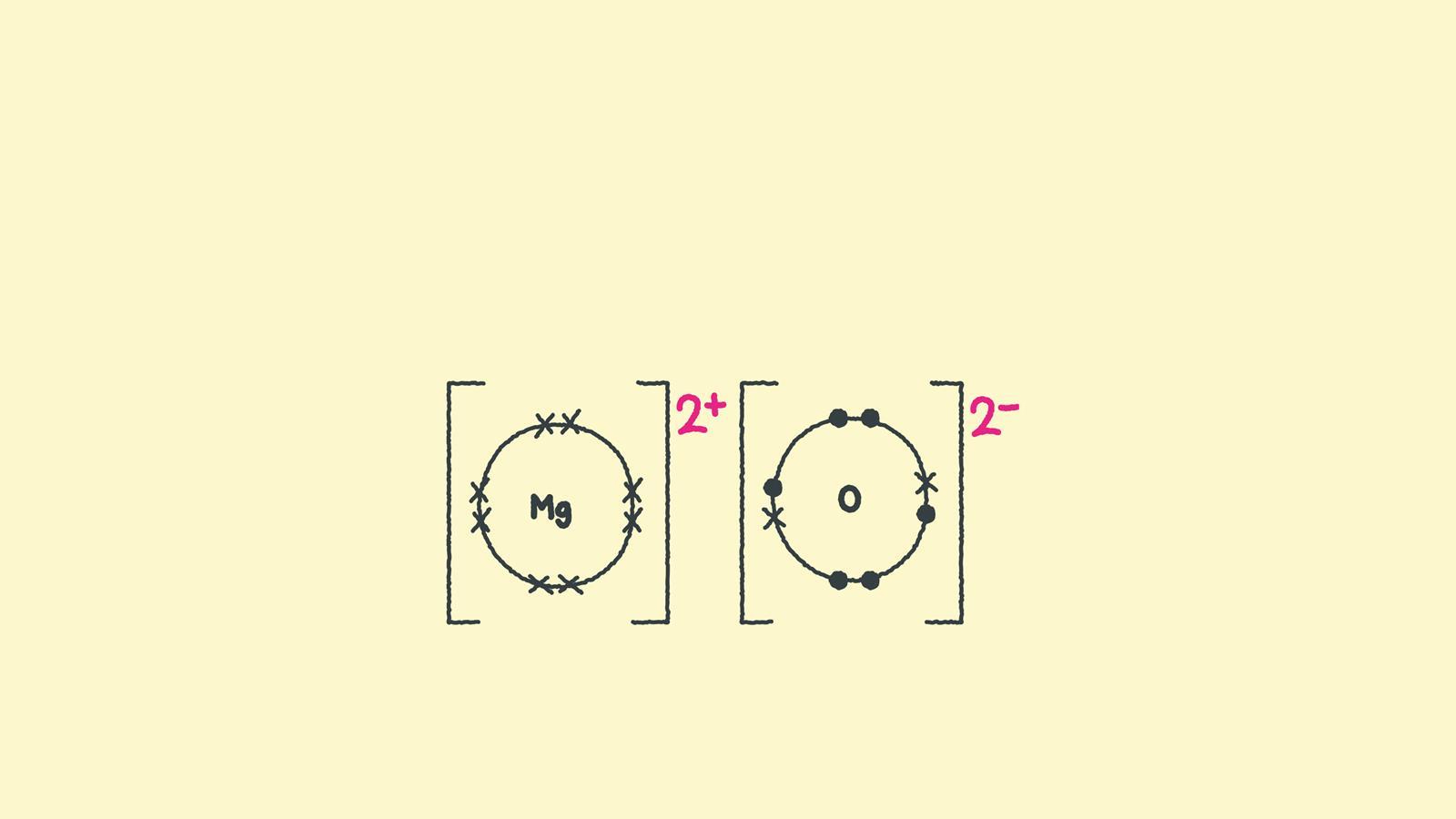
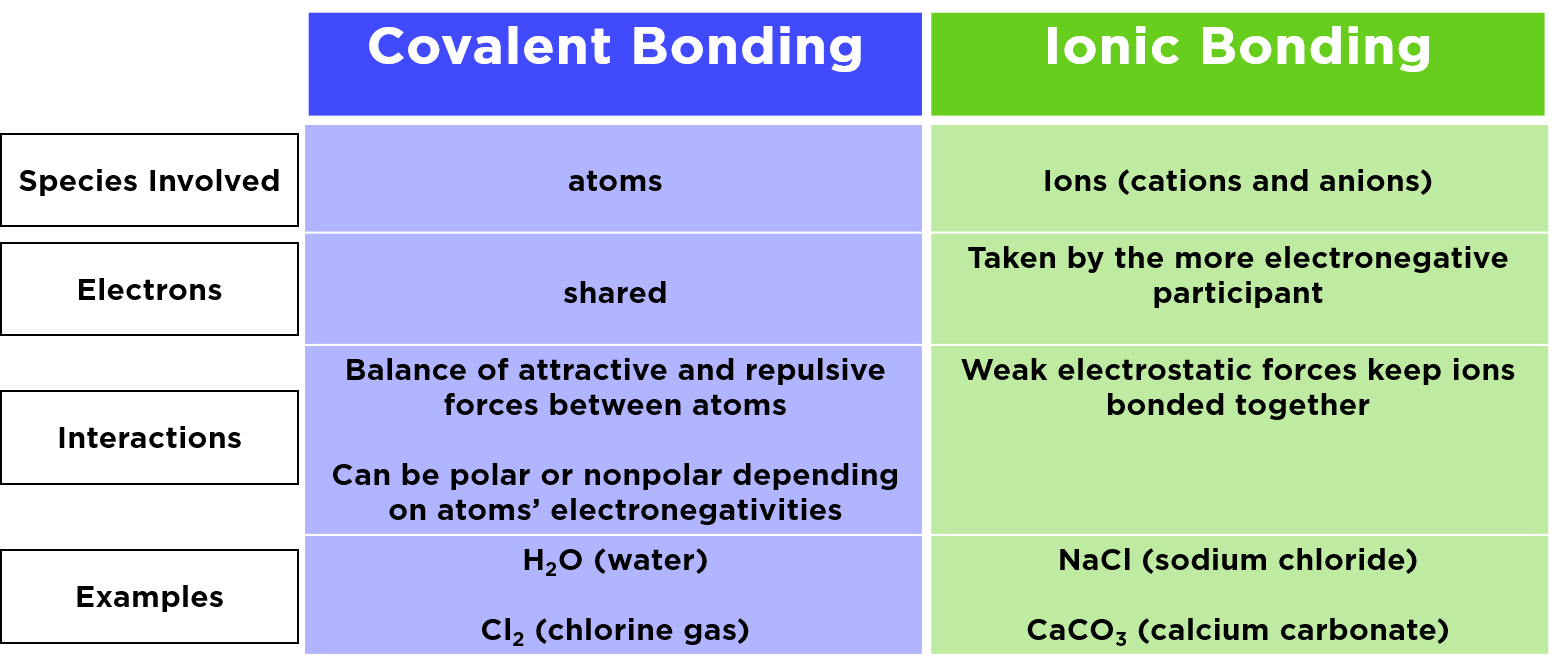
Specifically, ionic bonding happens when one atom is really, really good at giving away electrons (those tiny negative charges that zip around the atom's core), and another atom is really, really good at taking those electrons. When this electron exchange happens, both atoms become charged. The one that gives away electrons becomes positively charged, and the one that gains electrons becomes negatively charged. And what do opposite charges do? You guessed it – they stick together, like magnets! That's the essence of ionic bonding. It's what holds things like table salt (sodium chloride) together, where sodium gives an electron to chlorine.
Now, let's bring in our star of the show: oxygen. Oxygen, bless its little atomic heart, is a pretty reactive element. It's in the group of elements called "halogens" on the periodic table, and these guys are known for being electron-grabbers. Oxygen atoms are generally quite keen to gain electrons. In fact, they're pretty much always looking for a way to get two more electrons to feel complete and stable. They’re like that friend who’s always looking for a snack – they have a craving!
So, if oxygen is so good at taking electrons, how could it possibly engage in ionic bonding with another oxygen atom? Remember how ionic bonding involves one atom giving and one atom taking? This is where the plot thickens, and it gets really interesting.
In a typical scenario, you'd need an atom that's a fantastic electron donor. Think of something like sodium (Na) or potassium (K) – these elements are practically begging to get rid of an electron or two. They’re the generous givers of the atomic world. But what happens when you put two oxygen atoms together? They’re both electron-grabbers! It’s like having two people at a buffet who both want the last piece of cake.
So, can one oxygen atom say, "Okay, I'll give you an electron," and the other say, "Great, and I'll give you one"? Well, not in the traditional, straightforward sense of ionic bonding. Both oxygen atoms have a strong pull on electrons. They both want to gain electrons to reach that stable electron configuration. This is why oxygen usually forms covalent bonds with itself.

Covalent bonding is a different dance. Instead of a full-on transfer of electrons, it's more like a cooperative sharing. Two oxygen atoms will come together and share their electrons. Each atom contributes some of its electrons to a shared pool, and by sharing, both atoms feel like they have enough electrons to be stable. This is how the oxygen we breathe (O2) is formed – two oxygen atoms happily sharing electrons in a double covalent bond. It’s like they’re holding hands and sharing a toy, rather than one person handing over their toy and the other person keeping it.
But wait, you might be thinking, "Are there any loopholes? Any weird exceptions?" And that, my friends, is where the real fun of chemistry lies! While it's highly unlikely for two oxygen atoms to form a typical ionic bond in isolation, there are some very specific and often extreme conditions where we can see something resembling ionic character, or where oxygen exists in an ionic state within a larger compound.
Consider situations involving extremely electronegative elements. Now, "electronegativity" is just a fancy science word for how strongly an atom pulls on electrons. Fluorine is the undisputed champion of electronegativity. If you were to put oxygen next to fluorine, fluorine would pretty much snatch electrons away from oxygen without a second thought. This can lead to species like the difluoride cation (O F2+), or even more complex structures where oxygen might temporarily carry a positive charge. But again, this isn't two oxygen atoms bonding ionically with each other; it's oxygen interacting with a much stronger electron-grabber.
There are also very peculiar situations in highly oxidizing environments or in certain solid-state compounds where you might find oxygen in unusual oxidation states. For instance, in compounds like cesium oxide (Cs2O), oxygen exists as the oxide ion (O2-), which is a highly negatively charged species. Here, the cesium (Cs) atoms are donating their electrons to oxygen. But in this case, we have cesium, a very electropositive metal, doing the donating, not another oxygen atom. It’s like having a very generous person (cesium) giving their belongings to someone who really needs them (oxygen).
So, to circle back to our original question: Can two atoms of oxygen engage in ionic bonding? The short, sweet, and most scientifically accurate answer is: not really, not in the conventional sense. Their personalities, their inherent desire to gain electrons, make them much more suited for a cooperative, sharing relationship (covalent bonding) with each other.

It’s a bit like asking if two cats would form a perfect hunting partnership where one cat gives all the prey to the other. They’d probably end up sharing, or maybe even fighting over it! They’re both built for the hunt, and they both want to eat!
But the beauty of science is that there are always shades of gray, always edge cases, and always new discoveries waiting to be made. While pure ionic bonding between two oxygen atoms is a bit of a chemical impossibility due to their similar electron-grabbing tendencies, understanding why it doesn't happen beautifully illustrates the fundamental principles of how atoms interact. It highlights the power of electronegativity and the elegance of covalent sharing.
And honestly, isn't it kind of cool that the very air we breathe, the life-giving oxygen, is formed through this amazing act of sharing between two oxygen atoms? It’s a testament to how nature finds stability and creates the world around us, not always through dramatic give-aways, but often through gentle cooperation. So next time you take a deep breath, give a little nod to those oxygen atoms and their fantastic covalent bond!
