Construct The Expression For Kc For The Following Reaction

Ever wondered how chemists keep track of who's winning in a chemical reaction? It's not about cheering or waving little flags, but they have a super cool way to measure the "balance" of things. It’s all about this awesome thing called the equilibrium constant, or as the pros call it, Kc. Think of it like a scorekeeper for a chemical game. And guess what? You can actually write down the "scorecard" for any reversible reaction! Pretty neat, right?
Let's imagine a chemical reaction. It's like a little dance where some molecules break apart and rearrange to form new ones. But here's the twist: this dance can go both ways! The products can also decide to get back together and form the original reactants. This is what we call a reversible reaction. It’s like a seesaw, constantly adjusting its tilt.
Now, the fun part. We want to figure out how to write down the expression for this magical Kc. It’s like creating a secret code that tells us how much of each ingredient is hanging around when the reaction decides to chill out and reach its happy equilibrium. It’s not just random; there’s a specific way to do it, and once you know the trick, it’s super satisfying!
So, what’s the big deal? Why should you care about this Kc thing? Well, it’s like having a crystal ball for chemistry! This number, Kc, can tell us if a reaction is going to produce a ton of products, or if it's going to mostly stick around as the starting ingredients. It helps scientists predict and control chemical reactions, which is pretty important for everything from making medicines to creating new materials.
Imagine you’re baking. You have flour, eggs, and sugar. You mix them up and bake a cake. That’s your forward reaction. But what if, somehow, the cake could un-bake itself back into flour, eggs, and sugar? That’s the reversible part. Kc would tell you, after all the mixing and baking and maybe a little un-baking, how much cake batter (products) you’d likely have compared to your original ingredients (reactants).
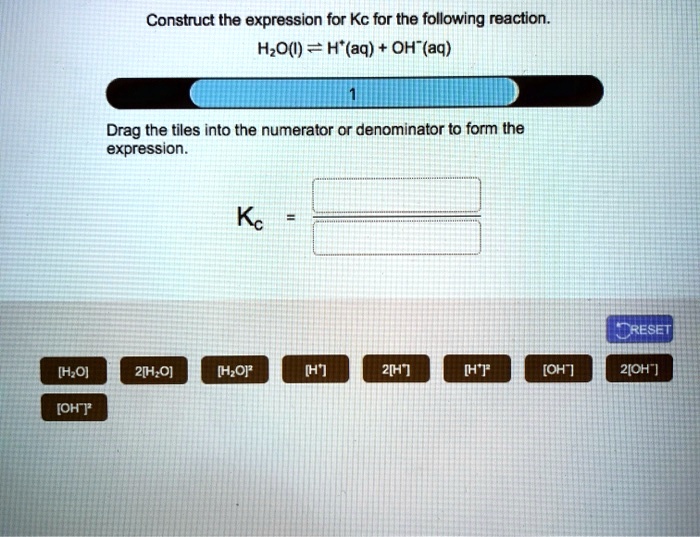
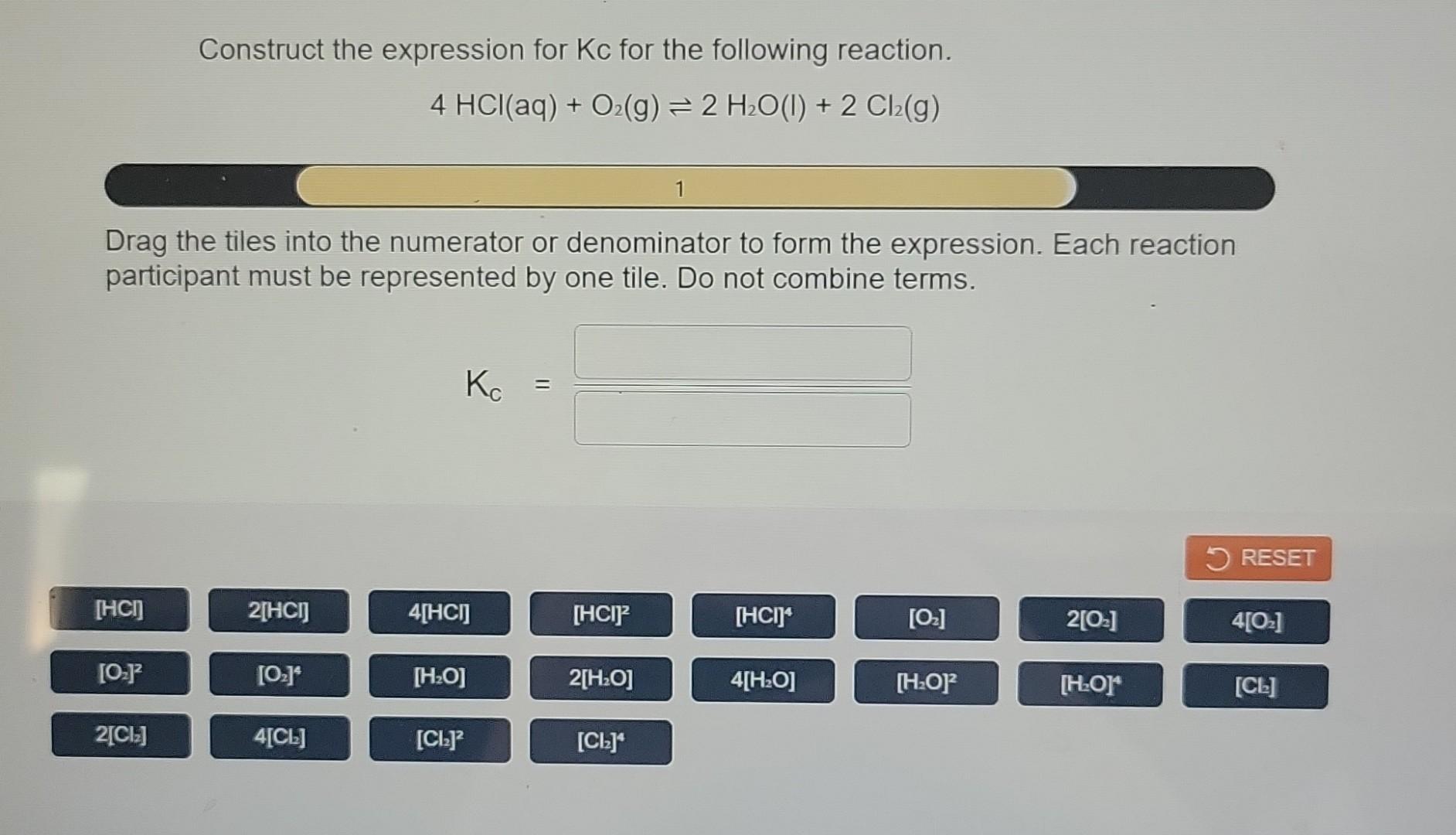
The expression for Kc is like a little recipe. For a general reversible reaction, let's say it looks like this: aA + bB ⇌ cC + dD. Now, don't let those letters and numbers scare you. The A and B are our starting ingredients, our reactants. And C and D are the new things we make, our products. The little letters a, b, c, and d are super important. They tell us how many of each molecule are involved in this chemical dance. Think of them as the dance steps!
To write the Kc expression, we put the products on top, like a delicious topping, and the reactants on the bottom, like the crust. Each chemical is represented by its symbol, and we raise it to the power of its "dance step" number. So, for our imaginary reaction aA + bB ⇌ cC + dD, the Kc expression looks like this:
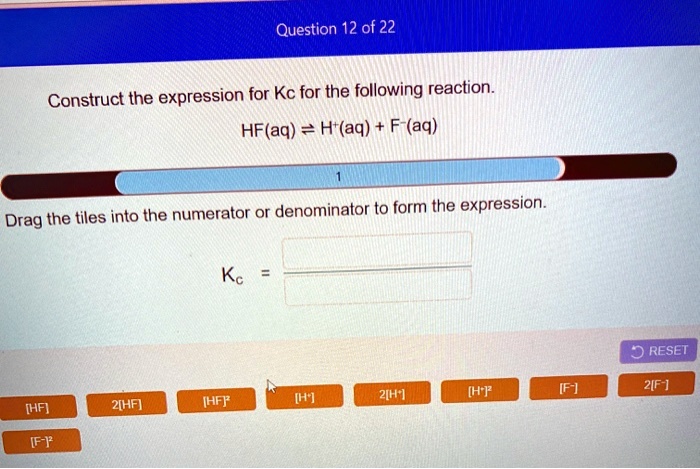
Kc = ([C]c[D]d) / ([A]a[B]b)
See? The products, C and D, are on the top, each multiplied together, and raised to their respective coefficients (c and d). The reactants, A and B, are on the bottom, also multiplied and raised to their coefficients (a and b). The square brackets, like [C], are just a shorthand for "the concentration of C." It's all about how much of each thing is present when the reaction has decided to take a break and let everything settle.
It's like saying, "The score (Kc) is found by taking the amount of stuff we made (products), times each other, adjusted for how many steps they took, and then dividing that by the amount of starting stuff we had, also adjusted for their steps." Simple, right?
This is what makes it so fascinating! You can look at any reversible chemical equation, and with a little understanding of these simple rules, you can write down the expression for its Kc. It’s like having a universal key to understanding chemical balance. No matter how complex the reaction might seem, the way to express its equilibrium is elegantly consistent. It’s a fundamental concept that opens up a whole world of understanding in chemistry.
Think about the aha! moment when you finally grasp it. It’s like unlocking a puzzle. You see a complicated equation, and suddenly, you can see its inherent order and how to quantify its tendency to favor products or reactants. This ability to construct the Kc expression is a foundational skill that allows chemists to delve deeper into reaction mechanisms and predict outcomes with confidence. It's a testament to the beautiful, underlying logic of the chemical world.
So, next time you see a reversible reaction, don't just see a jumble of letters and arrows. See an invitation to construct its Kc expression, a little window into the dynamic dance of molecules at equilibrium. It’s a skill that’s both useful and incredibly satisfying to master. Give it a try; you might just find yourself charmed by the elegant simplicity of it all!
