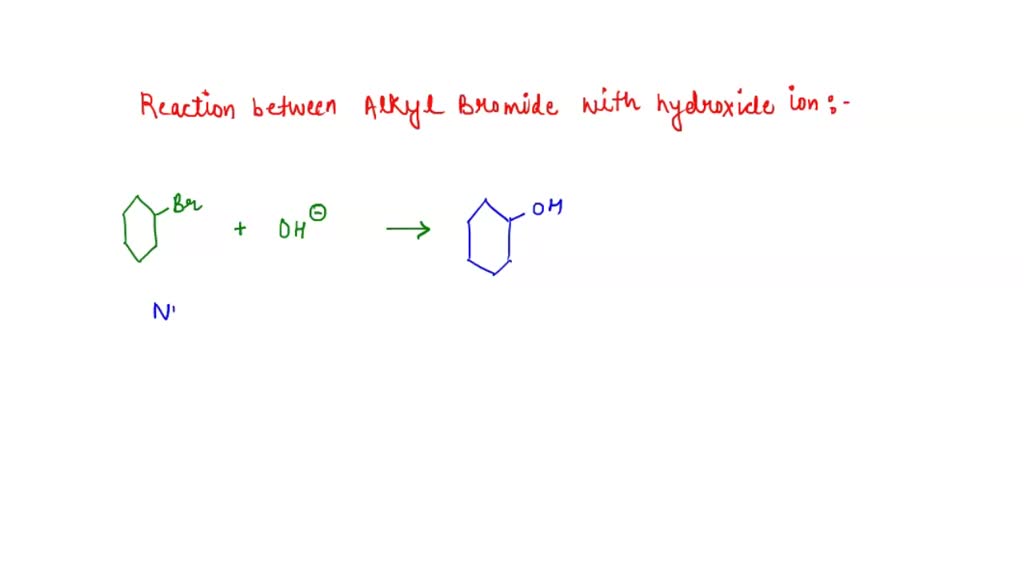
Consider The Reaction Of An Alkyl Bromide With Hydroxide
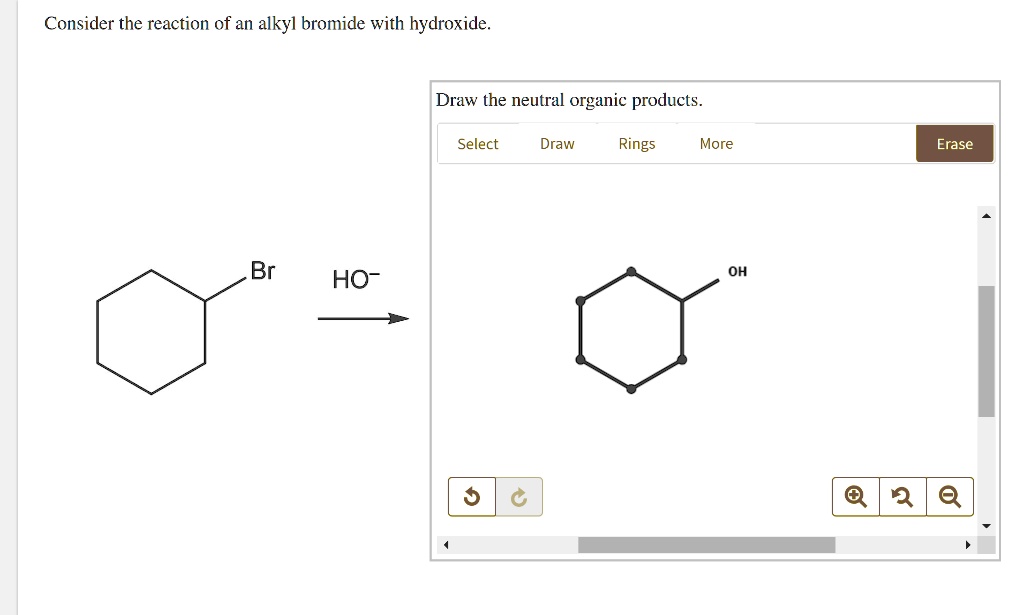
Hey there! Grab your mug, settle in. We’re gonna chat about something kinda cool, but also… well, it depends on what you're going for, you know? We're diving into what happens when an alkyl bromide decides to hang out with a hydroxide ion. Sounds fancy, right? But honestly, it's just a little chemical meet-cute, or sometimes, a total showdown!
So, imagine this: you’ve got this molecule, an alkyl bromide. Think of it like a little chain of carbons, and somewhere on that chain, there’s a bromine atom. Bromine's a bit of a drama queen in the chemical world, always looking for a bit of action. And then, BAM! Along comes the hydroxide ion. This little guy is basically water that's lost a hydrogen, and it's got a negative charge. Negativity, anyone? It's ready to pounce, so to speak.
Now, what happens when these two meet? It’s not always a simple hug, is it? Sometimes, it’s a full-blown chemical adventure. It all boils down to how these two characters decide to interact. And trust me, they’ve got a couple of different ways of doing things. It's like picking between a romantic comedy and an action flick, all depending on the co-stars and the setting.
The SN2 Smooch
Okay, so one of the main events here is what we call the SN2 reaction. Sounds like a secret agent code, doesn't it? S for substitution, N for nucleophilic, and the 2? That just means two molecules are involved in the slow step. Pretty neat.
In an SN2 scenario, the hydroxide ion, our little negative Nancy, is playing the role of the nucleophile. Nucleophile, by the way, just means it loves positive things. And guess what? The carbon atom bonded to the bromine is feeling a bit positively charged. Why? Because bromine is all electronegative, like a tiny black hole for electrons. It’s pulling all the electron-y goodness towards itself, leaving the carbon a little electron-starved and, therefore, a tad positive. Perfect bait for our hydroxide!
So, the hydroxide ion comes in, all suave and ready to go. It doesn't tiptoe; it goes in for a full-on attack! It’s a concerted reaction, meaning everything happens at once. The hydroxide starts to form a bond with that slightly positive carbon, and as it's doing that, the bond between the carbon and the bromine starts to break. It’s like a perfectly timed dance. One hand reaches out, the other lets go. Beautiful!
And the result? The bromine atom, now an ion all on its own (a bromide ion, obviously), gets kicked to the curb. Adios, bromine! Hello, alcohol! The hydroxide has successfully replaced the bromine. So, if you started with, say, bromomethane (that’s CH3Br), you end up with methanol (CH3OH). Boom! Instant alcohol. Easy peasy, lemon squeezy, right?
This SN2 thing is super efficient, especially if the alkyl group is nice and small. Think methyl or primary. Like, a tiny little carbon chain. Why? Because the hydroxide ion needs space to do its thing. If the carbon is all bunched up with other bulky groups, the hydroxide is gonna have a hard time getting to its target. It’s like trying to sneak a cookie into a crowded elevator – not happening smoothly!
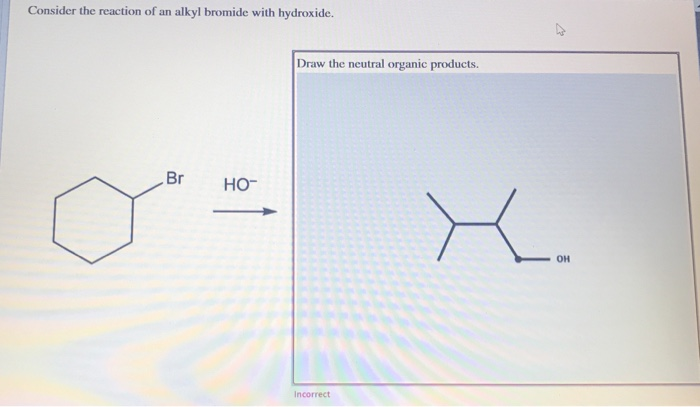
So, for SN2, we're talking about steric hindrance being a big deal. Less hindrance, more reaction! Primary and secondary alkyl bromides are usually the superstars here. Tertiary? Forget about it. It's like trying to park a bus in a compact car spot. No way!
The solvent also plays a nice little role. Polar aprotic solvents, like DMSO or acetone, are like the VIP lounges for SN2 reactions. They help stabilize the charged ions without getting in the way of the nucleophile's approach. It's all about creating the perfect environment for that smooth substitution.
The SN1 Sidestep (or Maybe a Stumble?)
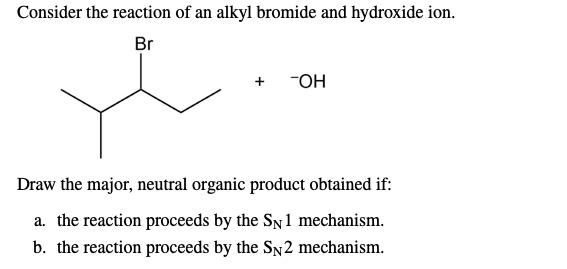
But wait, there’s more! What if the alkyl bromide is a bit more… complicated? Or what if the conditions are a little different? Then we might be looking at the SN1 reaction. S for substitution, N for nucleophilic, and the 1? That means only one molecule is involved in the slow, rate-determining step. Big difference!
In SN1, it’s a two-step process, which is, frankly, a lot less dramatic than SN2. It’s more of a slow burn. First, the alkyl bromide is all by itself, chilling. And then, it spontaneously decides to break the bond with the bromine. The bromine ion wanders off, leaving behind a carbocation. This carbocation is a carbon atom with a positive charge. It's a bit unstable, like a teenager with too much energy and no direction.
This carbocation formation is the slow step. It’s the part that takes the longest because it’s just the bromide breaking away. It needs a little push, a little encouragement, or just the right conditions to happen. And once it’s formed, that carbocation is like a beacon for anything negative.
Enter our friendly neighborhood hydroxide ion again! Now, it doesn’t have to fight for space. The carbocation is just sitting there, looking all exposed and positive. The hydroxide can waltz in from either side. Because the carbocation is planar (flat, like a pancake!), the nucleophile can attack from the top or the bottom. What does that mean? Racemization, my friends! If the starting material was chiral (meaning it’s got that "handedness," like your left and right hands), you’ll end up with a mixture of both possible versions, called enantiomers. It's like making a molecule that’s both left-handed and right-handed at the same time. Weird, but cool!
So, SN1 is all about creating that carbocation. And what makes a carbocation stable? More alkyl groups attached to the positive carbon! Tertiary carbocations are way more stable than secondary, which are way more stable than primary. So, tertiary alkyl bromides are the absolute VIPs for SN1. They just love forming those carbocations. It’s their jam!
And the solvent? For SN1, we like polar protic solvents. Think water or alcohols. These guys are great because they can solvate (surround and stabilize) both the bromide ion that leaves and the carbocation that forms. They’re like the supportive crowd at the carbocation’s coming-out party.
The big takeaway here is that SN1 reactions prefer tertiary alkyl halides because the tertiary carbocation is the most stable. Primary and secondary alkyl halides? They're usually too unstable to form a carbocation, so they'll probably go for the SN2 route instead, if they can.
The Elimination Encore: E1 and E2
But hold on, it’s not just about substitution! Sometimes, the hydroxide ion, or a similar strong base, has a different idea. Instead of replacing the bromine, it might decide to steal a proton (a hydrogen ion) from a neighboring carbon. This leads to an elimination reaction. We’re getting rid of bits and pieces, and usually, that means forming a double bond!
There are two main elimination pathways: E1 and E2. They sound a bit like dance moves, don't they? E for elimination, and the 1 or 2 tells you about the mechanism, just like in substitution.
E2 is the more direct one, and it’s a lot like SN2. It's a concerted reaction. The base (hydroxide, in our case) attacks a proton on a beta carbon (that’s the carbon next to the one with the bromine). As the base is grabbing that proton, the electrons from the C-H bond move to form a pi bond between the alpha and beta carbons, and at the same time, the C-Br bond breaks, and the bromide leaves. It’s a whole symphony of bond-making and bond-breaking happening simultaneously!
For E2, we need a strong base. Hydroxide is a good one. And the alkyl halide? Well, just like SN2, steric hindrance can be a factor, but it's more about the availability of those beta-hydrogens. Also, E2 prefers more substituted alkyl halides for product stability (Zaitsev's rule, anyone? Bigger, more substituted alkenes are generally favored). It’s a bit of a race between SN2 and E2 when you have a strong base and a secondary or tertiary alkyl halide. Which one wins? It depends on the specific conditions, like temperature. Higher temperatures often favor elimination!
Now, E1 is the slower, two-step process. It's the elimination equivalent of SN1. First, the alkyl bromide breaks down to form a carbocation, just like in SN1. Then, a base (which can be the solvent, like water or alcohol, or a separate base) comes along and steals a proton from a neighboring carbon to form the double bond. So, E1 often happens alongside SN1 because they share that initial carbocation intermediate.
If you have a tertiary alkyl bromide, for instance, and you put it in water (a polar protic solvent), you're likely to get a mix of SN1 and E1 products. The carbocation is happy to form, and then the water can either act as a nucleophile to substitute (SN1) or as a base to abstract a proton and eliminate (E1).
So, What's the Verdict?
It’s not a one-size-fits-all situation, is it? The reaction of an alkyl bromide with hydroxide is like a choose-your-own-adventure story for chemists.
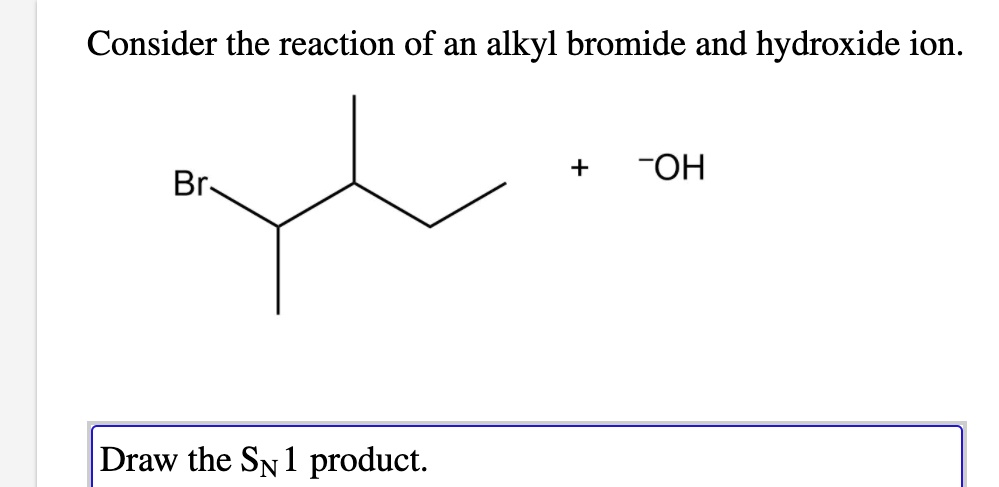
If you've got a primary alkyl bromide and a strong nucleophile like hydroxide, you're almost guaranteed an SN2 reaction. It’s clean, it’s fast, and you get your alcohol. Easy.
If you have a tertiary alkyl bromide, especially in a polar protic solvent, you're probably looking at a mix of SN1 and E1. The carbocation is just too stable to resist forming. You’ll get substitution, but you might also get an alkene.
For secondary alkyl bromides, it gets a bit more interesting. They can undergo both SN2 and E2 with a strong base like hydroxide. If you're trying to get an alcohol, you might favor conditions that push towards SN2. If you want an alkene, you might crank up the heat to encourage E2.
And what about the base strength and nucleophilicity? Hydroxide is both a strong base and a pretty good nucleophile. This is why it can drive both SN2 and E2. If you had a weaker, less nucleophilic base, it might favor elimination more. If you had a stronger nucleophile that wasn't such a strong base, it might favor substitution even more.
It’s all about the dance. The structure of the alkyl bromide, the strength and nature of the attacking species (is it more basic or more nucleophilic?), the solvent, and even the temperature – they all play a part in deciding which pathway wins. It’s like a chemical popularity contest!
So next time you see an alkyl bromide and a hydroxide ion chilling together, remember they're not just passively observing each other. They're potentially embarking on a whole chemical journey. It could be a swift substitution, a slow carbocation formation followed by attack, or even a dramatic elimination to form a double bond. It’s a fascinating little world, and understanding these reactions is key to building all sorts of cool molecules. Pretty wild, huh? Now, who needs a refill?
