Consider The Reaction Of An Alkyl Bromide And Hydroxide Ion.
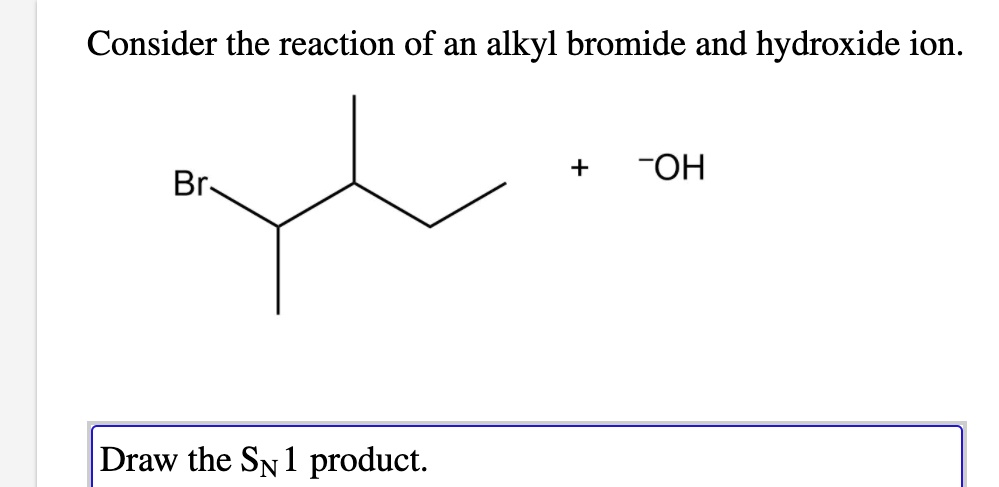
So, you've got this molecule, right? An alkyl bromide. Think of it like a tiny, little party animal. It's got this interesting feature – a bromine atom hanging off. Bromine is, well, a bit of a drama queen. It loves to hog all the attention, especially the electrons. This makes the carbon atom it's attached to a little bit needy, a little bit vulnerable. It's like the kid at school who's always losing their lunch money. Everyone knows it's an easy target.
Now, imagine this needy carbon atom, this alkyl bromide, bumping into a hydroxide ion. The hydroxide ion, on the other hand, is like a super-charged, no-nonsense cleaner. It's got this extra electron, this negative charge, and it's not afraid to use it. It's always looking for something to fix, something to… well, react with.
When these two meet, it's like a tiny chemical speed dating event. The hydroxide ion, being the proactive type, spots that needy carbon. It’s like, "Hey there, little guy! You look like you could use a friend. Or, more accurately, you look like you could use some my electrons!"
And so, the dance begins. The hydroxide ion, with its negative aura, gets all flirty with that slightly positive, electron-deprived carbon on the alkyl bromide. It's like a moth to a flame, or maybe a particularly enthusiastic Labrador to a dropped piece of cheese. There's just an undeniable pull.
Now, here's where it gets interesting, and where I think the textbooks maybe, just maybe, are a tad too serious. They talk about these things called "mechanisms." Like it's some intricate choreography. But honestly, when you see an alkyl bromide and a hydroxide ion get together, it feels a lot simpler than that. It feels like pure, unadulterated chemical chemistry.
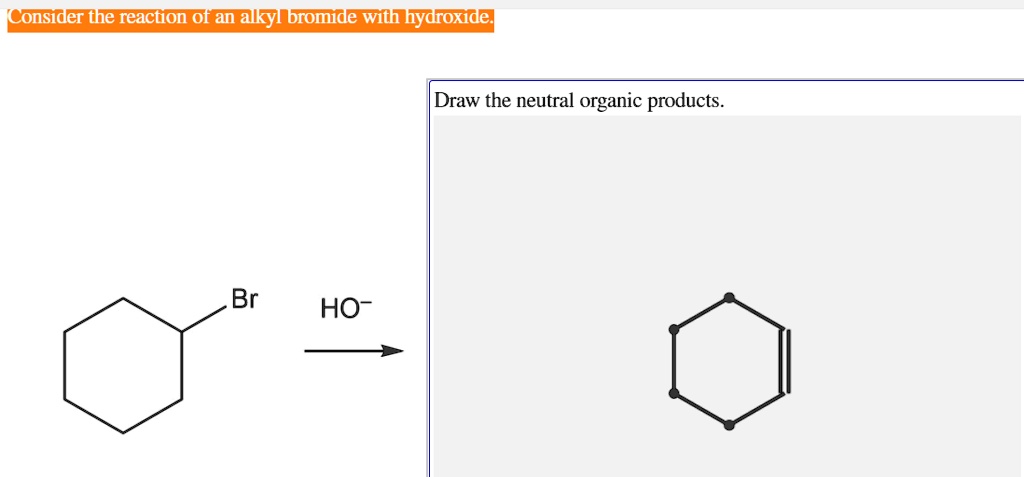
The hydroxide ion is just so… ready. It's got this abundance of negative charge. It’s like it’s just been waiting for an opportunity to share its goodness. And the alkyl bromide? It's just begging for it. It’s like the other side of the coin. One’s got too much, the other’s got not enough. Perfect match, right?
So, the hydroxide ion swoops in. It’s not subtle about it. It’s like, "Here I come!" And that bromine atom on the alkyl bromide? Well, bromine is a bit of a flighty character. It’s like, "Uh oh, someone's getting a little too close for comfort!" It sees this approaching negativity, this incoming wave of chemical affection, and it decides it's time to make a strategic exit.
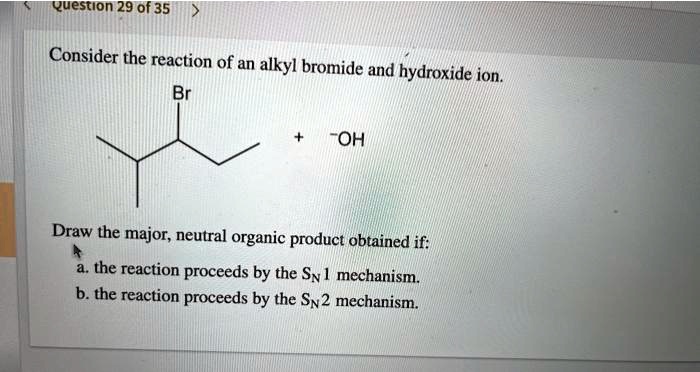
It’s like the bromine atom is the annoying ex who's still lurking around. The hydroxide ion arrives, and suddenly, the bromine is like, "You know what? I've had enough of this party. I'm out!" And it just leaves. Poof! Gone. It gets carried away, usually by the electron flow, becoming its own free-floating entity, a lonely bromide ion.
And what's left behind? The carbon that was just so desperately seeking electrons? It's now got the hydroxide ion all cozied up to it. It's like the shy kid who finally got asked to the dance by the most popular person. They're now bonded, a new molecule formed. It's an alcohol! Isn't that just lovely?
It’s like this:
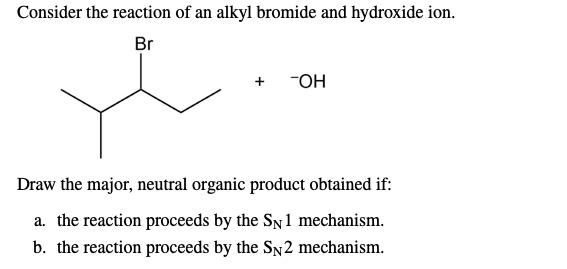
Alkyl Bromide (the needy one) + Hydroxide Ion (the generous one) = Alcohol (the happy couple) + Bromide Ion (the ex who left)
And honestly, that’s the beauty of it. It’s not always about complex steps and fancy names. Sometimes, it’s just about who’s got what, and who’s willing to share. The hydroxide ion is always ready to share its negative charge, and the alkyl bromide is always ready to accept it, especially when that bromine person is making a quick getaway.
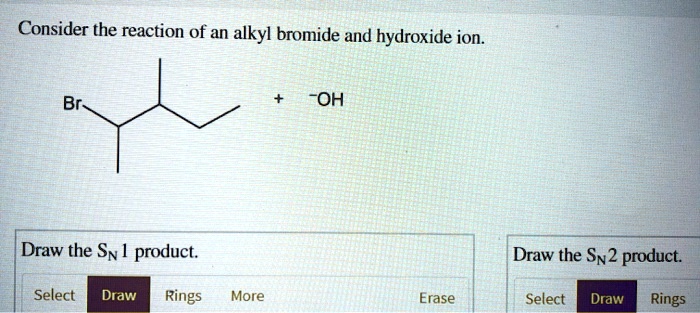
I know, I know. The chemists will say, "But there are pathways! There are intermediates!" And sure, they might be technically right. But if you watch it happen, if you picture it in your mind, it feels more like a simple exchange, a chemical handshake with a happy ending. The hydroxide ion doesn't plan to steal the carbon; it's just so attracted to its electron deficiency.
And that bromine? It's not forced out. It's more like it sees the writing on the wall. It’s like, "Okay, this is getting serious. I’m a lone wolf molecule, and this pairing is too intense for me." So, it dips. It’s a graceful, albeit slightly dramatic, departure.
So, the next time you hear about an alkyl bromide and a hydroxide ion, don't get bogged down in the jargon. Just picture that needy carbon, that generous hydroxide, and that slightly dramatic bromine. It’s a simple story of attraction, departure, and a new beginning. And sometimes, the simplest explanations are the most entertaining, and dare I say, the most true.
