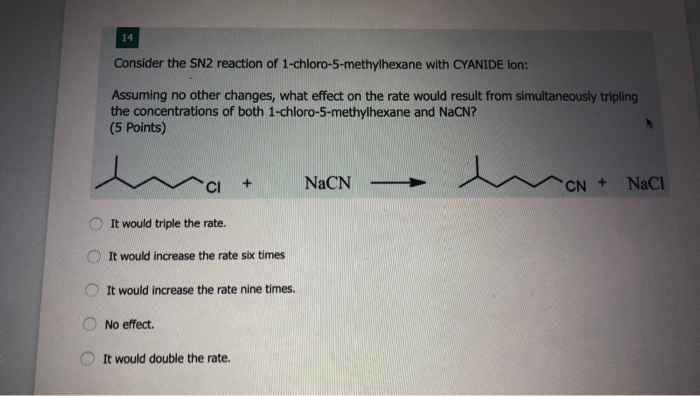
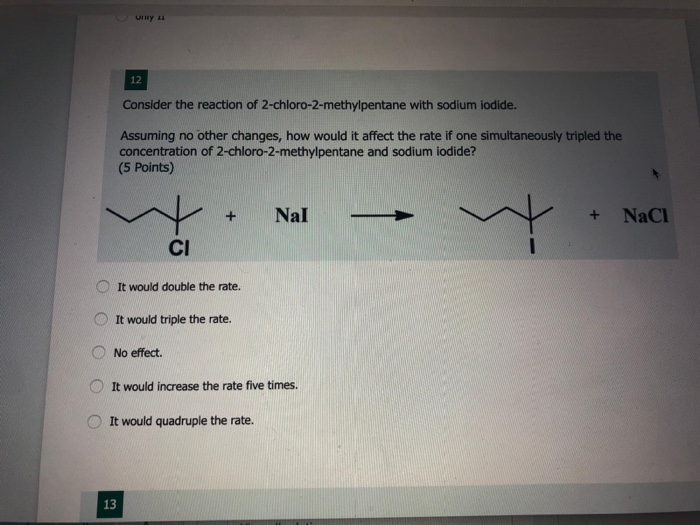
Consider The Reaction Of 2-chloro-2-methylpentane With Sodium Iodide

So, you've got 2-chloro-2-methylpentane hanging around. Fancy name, right? It sounds like something a mad scientist would whip up in their lab. Or maybe it's just another one of those oddly named ingredients in a particularly pungent cleaning product. Either way, it's got a story to tell.
And then, wham! In waltzes sodium iodide. Talk about an entrance! It’s like the cool kid showing up to a party. Sodium iodide probably has its own theme music. It’s got that certain sparkle, that certain je ne sais quoi.
Now, what happens when these two meet? It's a bit like a blind date, really. You’ve got one molecule minding its own business, and then another one comes along. Will they hit it off? Will it be awkward? Will there be sparks?
Let’s just say, this particular chemical rendezvous is more of a friendly swap. Think of it as a cosmic game of tag. 2-chloro-2-methylpentane is holding onto its chlorine atom really tightly. It’s like, “Nope, this is mine!”
But sodium iodide is feeling adventurous. It sees that chlorine and thinks, “You know what? I’m feeling a bit…iodine-y today. I think I want that.” It's a bold move, but hey, that's the spirit of science!
So, sodium iodide basically taps 2-chloro-2-methylpentane on the shoulder. It’s a polite tap, of course. No pushing or shoving allowed in the molecular world, unless you’re dealing with truly chaotic substances. And these two? They’re not that dramatic.
The chlorine atom, bless its heart, is kind of a quitter. It's like, “Oh, you want me? Fine. Here you go.” It lets go. It’s probably tired of being attached anyway. A change of scenery is good for everyone, right?
And in its place? Enter iodine! It slides right in. It’s a perfect fit. It’s like a missing puzzle piece. Or like finding the last slice of pizza. Pure satisfaction.
So, our once 2-chloro-2-methylpentane is now, ta-da!, 2-iodo-2-methylpentane. See? A simple switcheroo. It’s a whole new molecule with a whole new vibe.
And the sodium atom? Well, it got a new buddy too. It hooked up with the departed chlorine. They’re like, “Hey, we’re both single now. Let’s hang out.” And they form sodium chloride. You might know it as table salt. Fancy, huh?
This whole process? It's called a nucleophilic substitution. Big words, I know. But think of it as a molecular dance. The iodide ion is the energetic dancer, and the chlorine atom is the one who gets replaced.
And what kind of dance is this? It’s often a SN2 dance. It's a bit of a group effort. Everyone is involved at the same time. It’s a well-choreographed routine. No waiting around.
But wait, there's a twist! Our starting molecule, 2-chloro-2-methylpentane, is a bit of a special case. It's a tertiary carbocation waiting to happen. This means it's a little more inclined to go its own way.
Sometimes, the chlorine just up and leaves on its own. It's like it has FOMO (Fear Of Missing Out). It sees the party happening and just dips. This leaves behind a positively charged carbon atom.
This positively charged carbon atom? It's called a carbocation. It's feeling a bit lonely and desperate. It’s like a single person at a wedding, looking for a dance partner.
And who’s there to answer the call? Our friendly neighborhood iodide ion! It’s the perfect rebound. It swoops in and fills that positive void. It’s a match made in chemical heaven.
This second dance? It's more of a solo act, followed by a duet. It's a SN1 dance. First, the chlorine leaves. Then, the iodide jumps in. It’s a two-step process.
So, depending on the exact conditions, you could have a bit of both. A little bit of SN2 flair and a dash of SN1 drama. It's like a molecular mixtape.
The real beauty here, though, is the halide exchange. That’s the fancy term for swapping one halogen for another. Think of it as trading baseball cards, but way more important for science.
Iodide is a much better leaving group than chloride. It’s like it’s more eager to pack its bags and go. It’s more mobile, more willing to be part of a new adventure.
And sodium iodide is a fantastic source of these eager iodide ions. It’s like a vending machine for good leaving groups. Just pop in a bit of solvent, and out comes your desired ion.
So, what's the takeaway from this little chemical escapade? It’s that sometimes, a simple swap can lead to a whole new compound. It’s like a makeover, but with atoms.
And it’s all thanks to the power of nucleophilic substitution. A concept that sounds intimidating but is really just about molecules finding new partners.
It’s kind of like dating, isn’t it? You’ve got your current situation, and then someone new comes along, and things change. Sometimes for the better, sometimes… well, sometimes you just end up with a bunch of salt. And honestly, who doesn’t need more salt?
This reaction is a classic example of how different molecules interact. It’s a fundamental concept in organic chemistry. It helps us understand how to build and transform other compounds.
It's also a testament to the fact that even seemingly complex chemical names can lead to surprisingly straightforward reactions. 2-chloro-2-methylpentane and sodium iodide. They might sound like aliens, but their interaction is quite down-to-earth.
So, the next time you hear about 2-chloro-2-methylpentane and sodium iodide, don’t be intimidated. Just picture a friendly molecular swap. A little bit of rearrangement, a little bit of new beginnings.

And who knows? Maybe this reaction is what dreams are made of for some chemists. The perfect substitution, the ideal product. It's their version of finding a unicorn.
But for us mere mortals? It’s just a fun little reminder that chemistry is happening all around us, even if it’s just a subtle exchange of atoms. It’s the silent symphony of the molecular world.
And honestly, isn't that a little bit magical? A world where atoms can trade places like they’re swapping stickers? I think it’s pretty cool. It's an unpopular opinion, perhaps, but I stand by it.
So, next time you’re looking at a chemical reaction, remember this one. Remember the 2-chloro-2-methylpentane, the sodium iodide, and the simple, elegant swap that takes place. It's a small change with big implications.
And who knows, maybe you'll start seeing chemical reactions everywhere. In your coffee, in the air, in that slightly strange smell from the science lab. It’s all part of the grand, interconnected chemical tapestry.
It’s a testament to the fact that even the most complicated-sounding substances can engage in simple, relatable interactions. They just want to find their perfect match, or in this case, their perfect swap.
So, let’s raise a glass – or a beaker – to 2-chloro-2-methylpentane and sodium iodide. May their future reactions be just as entertaining and enlightening. And may we all find our own perfect substitutions in life.
