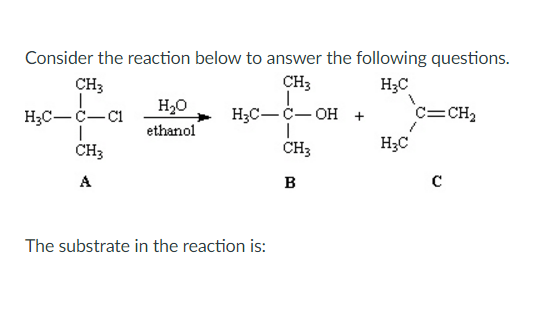
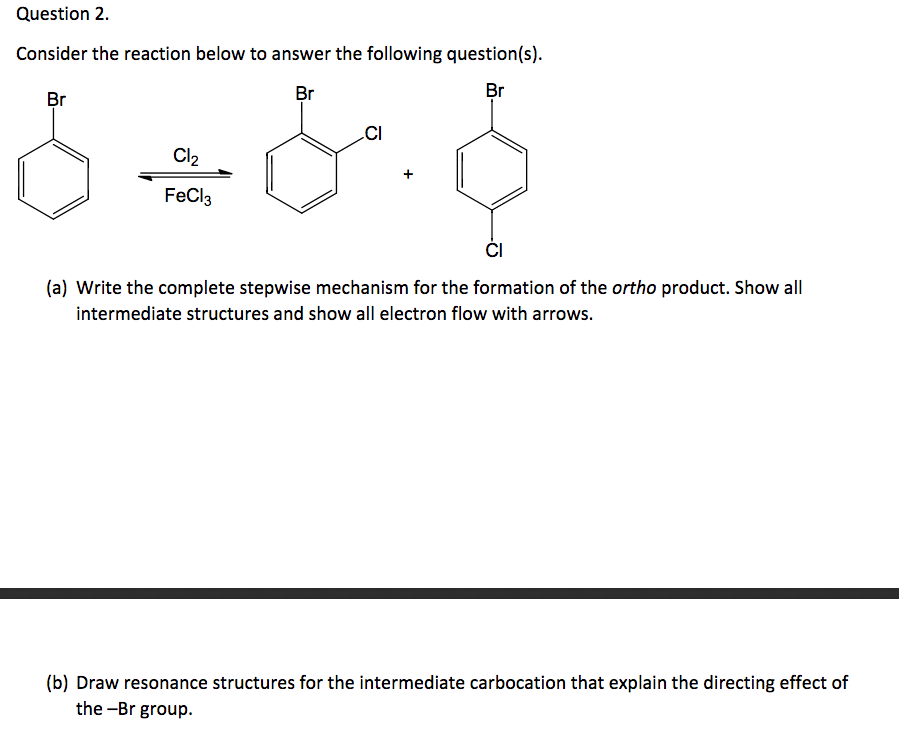
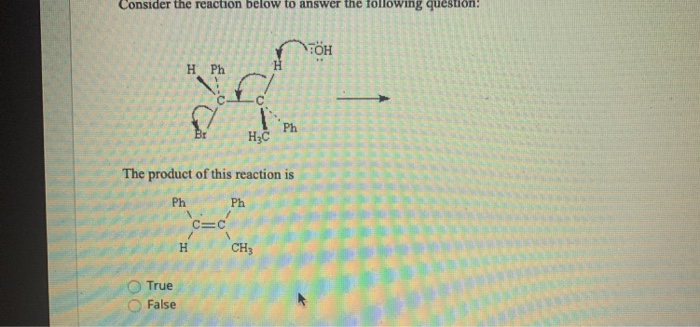
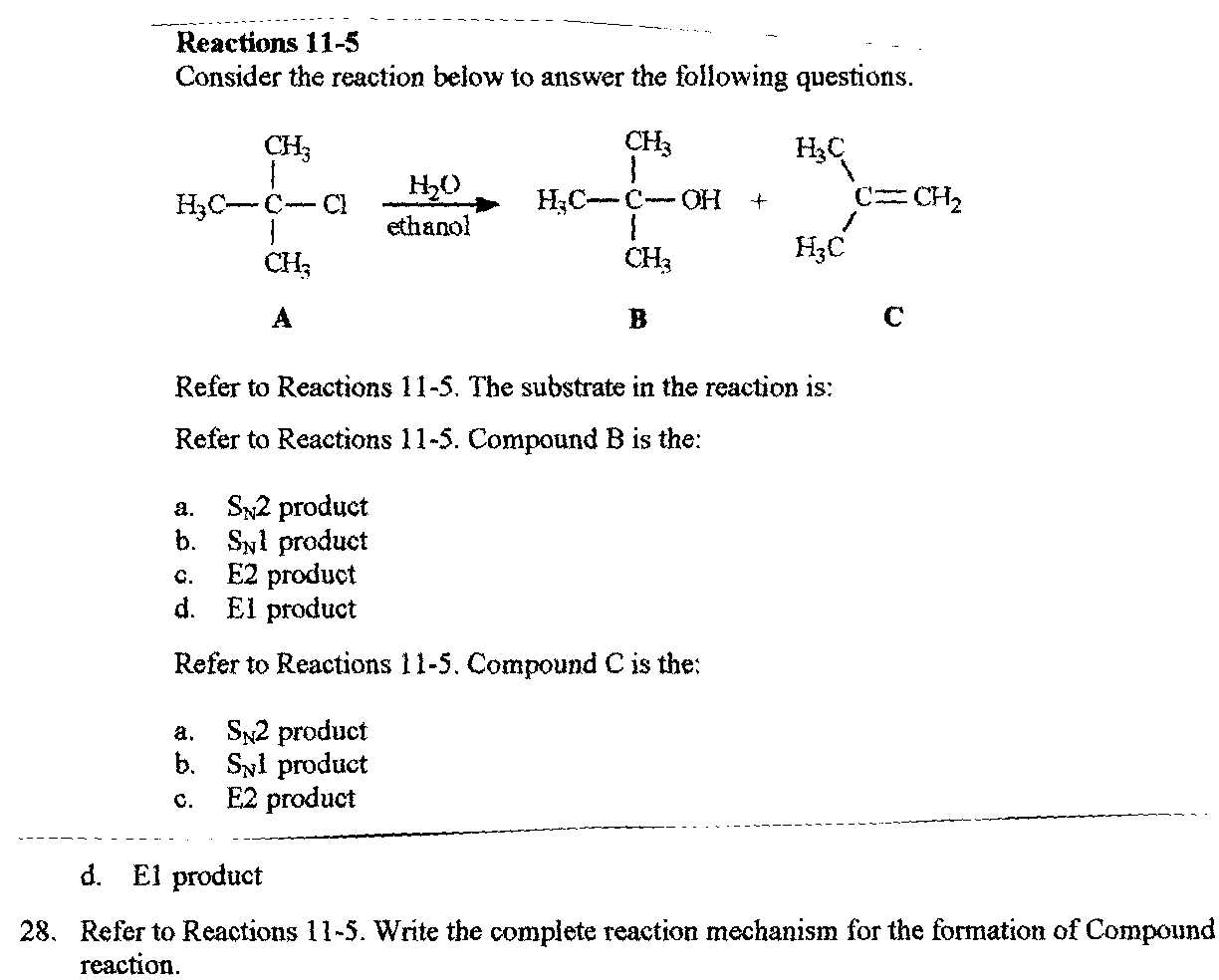
Consider The Reaction Below To Answer The Following Questions

Hey there, science adventurers! Today, we're diving headfirst into a super cool chemistry reaction. Don't worry, it's not going to be all complicated formulas and scary-looking equations. Think of me as your friendly guide, leading you through this little chemical playground. We’re going to tackle some questions about a reaction, and honestly, it’s going to be more like a fun puzzle than a pop quiz. So, grab your imaginary lab coat (mine's probably covered in coffee stains, let's be real) and let's get started!
So, we've got this reaction kicking around. Let's call it our "Mystery Mix" for now. It’s like the secret sauce of the chemistry world, and we're here to figure out what makes it tick. It's a bit like trying to understand why your cat suddenly decides to zoom around the house at 3 AM – there's a reason, and we're going to uncover it!
Unpacking the Ingredients: What's in the Pot?
Alright, first things first. Every good recipe, whether it's for cookies or a chemical reaction, starts with the ingredients. In our Mystery Mix, we've got a few players. We’re going to be looking at things like reactants – these are the starting materials, the raw ingredients that are going to get all mixed up and transformed. Think of them as the flour, sugar, and eggs before they become a delicious cake.
Then, we have the products. These are the superstars that emerge after the reaction has done its thing. They're the brand-new goodies, the transformed versions of our initial ingredients. So, in our cake analogy, the products are the beautifully baked, maybe even frosted, cake itself! Voila!
Understanding which is which is like knowing your onions from your carrots when you’re chopping for a stew. It’s fundamental, but also surprisingly satisfying when you get it right. It’s the difference between a culinary masterpiece and… well, something you’d rather not talk about. But fear not, this reaction is pretty straightforward.
The Balancing Act: Making Things Even Stevens
Now, chemistry has this funny thing called the law of conservation of mass. It sounds super serious, but it basically means that in a chemical reaction, you can’t just magic atoms out of thin air, and you can’t make them disappear either. They just get rearranged, like LEGO bricks being rebuilt into a new spaceship. Mind. Blown.
This is where balancing the equation comes in. Imagine you’re trying to build a LEGO castle. You start with a certain number of red bricks and a certain number of blue bricks. When you build your castle, you’re going to use all those bricks, just in a different configuration. The total number of red bricks and blue bricks stays the same, right? That’s what balancing an equation is all about. We make sure the number of atoms of each element is the same on both sides of the reaction arrow.
It’s like a cosmic game of Tetris, where we need to ensure every block has a perfect counterpart. Sometimes it’s super easy, and the equation practically balances itself. Other times, it’s a bit more like trying to get those stubborn puzzle pieces to fit. You might have to add little numbers, called coefficients, in front of the chemical formulas. These coefficients tell us how many molecules or formula units of each substance are involved. They’re the secret sauce to making everything equal.
So, when we look at our Mystery Mix, one of the first things we'll be asking is: "Is this thing balanced?" It's the chemical equivalent of checking if you have enough ingredients before you start baking. No one wants to run out of baking soda halfway through a cookie recipe, and chemists certainly don't want to find they're missing a crucial atom!
The Direction of Travel: Which Way Is It Going?
Chemical reactions, like a good story, have a direction. They go from the reactants to the products. That's why we draw a nice little arrow in the middle of the equation, pointing from left to right. It's like the river of chemistry, flowing from its source (reactants) to its destination (products). Smooth sailing!
Sometimes, reactions are like a one-way street. Once those reactants decide to become products, there’s no turning back. They’ve made their decision, and that’s that. Irreversible reactions are like a very committed relationship – once it’s happened, it’s happened. Think of burning wood; you can't un-burn it back into a log, can you? (Unless you have a time machine, which, as far as I know, isn't standard lab equipment.)
Other times, though, reactions are more like a lively dance with a bit of back-and-forth. These are called reversible reactions. The reactants can turn into products, and then the products can decide to turn back into reactants. It's like a conversation where both sides have something to say. These reactions reach something called equilibrium, which is a fancy term for a state where the forward reaction (reactants to products) and the reverse reaction (products to reactants) are happening at the same rate. It doesn’t mean the reaction stops; it just means things are perfectly balanced in terms of how fast they're going in each direction. It’s like a seesaw that’s perfectly level because the weight on both sides is exactly the same.
When we’re analyzing our Mystery Mix, figuring out if it's a one-way street or a two-way dance is pretty important. It tells us a lot about what's going on and what we can expect to see at the end of it all. It's the plot twist in our chemical story!
State of Mind (or Matter!): Solid, Liquid, or Gas?
Things in chemistry, just like in life, can exist in different forms. We've got solids (nice and rigid, like a grumpy old man), liquids (flowy and adaptable, like a chill barista), and gases (everywhere and nowhere, like your Wi-Fi signal). These are the states of matter, and they're often indicated in chemical equations using little letters in parentheses.
You might see an (s) for solid, an (l) for liquid, a (g) for gas, and sometimes even an (aq) for aqueous, which just means something is dissolved in water. Think of it as the outfit each chemical is wearing for the reaction. Is it in its sturdy boots (solid), its flowing scarf (liquid), or its invisible cape (gas)?
Why does this matter? Well, a solid behaving like a solid is very different from a gas bouncing around. The state of matter can really affect how a reaction happens, how fast it happens, and what kind of products you end up with. It’s like trying to cook a steak versus trying to boil water – the starting state makes a big difference to the end result!
So, when we’re looking at our Mystery Mix, keeping an eye on these states of matter is like checking the weather report before you plan a picnic. It helps us understand the environment in which the reaction is taking place. Are things bubbling and steaming (gas involved)? Are things dissolving and mixing (aqueous)? It all adds another layer to our understanding.
The Energy Game: Heat It Up or Cool It Down?
Every reaction needs a little nudge, or sometimes, it gives us a little nudge back! This is about energy. Some reactions need to absorb energy to happen. They’re like us needing a good cup of coffee to get going in the morning. These are called endothermic reactions. They soak up heat from their surroundings, making the environment feel colder. Think of those instant cold packs – they get chilly because they’re absorbing heat!
On the flip side, some reactions release energy. They’re like a fireworks show, giving off light and heat! These are called exothermic reactions. They produce heat, making the surroundings warmer. Burning fuel is a classic example. Woo, heat!
Sometimes, this energy change is written directly into the chemical equation. You might see "heat" or "energy" appearing on one side or the other. It's like the reaction is telling us if it's feeling a bit chilly and needs some warmth, or if it’s feeling super energetic and wants to share!
When we analyze our Mystery Mix, understanding its energy game is crucial. Is it going to warm up our little flask, or is it going to chill us out? This can tell us a lot about its stability and how it might behave in different situations. It's the emotional temperature of our reaction!
Putting It All Together: The Big Picture
So, we've chatted about reactants, products, balancing, direction, states of matter, and energy. Now, when you see a chemical reaction, you've got a toolkit to start asking the right questions. You can look at the starting ingredients and predict the final goodies. You can make sure everything is accounted for, like a meticulous accountant of atoms.
You can tell if it's a journey with a clear destination or a scenic route with detours. You can understand the physical form of everything involved, from a stubborn solid to an elusive gas. And you can even get a sense of whether the reaction is feeling energetic or a bit sluggish.
It’s really not as intimidating as it sounds, is it? It’s all about observation and a little bit of logical deduction. Think of it as a detective story, but instead of a stolen jewel, you’re solving the mystery of how atoms rearrange themselves.
Your Turn to Shine!
Now, when you're presented with our little Mystery Mix, you're ready to tackle those questions. You've got the inside scoop! You're not just looking at symbols; you're understanding a process, a transformation, a mini-adventure happening right before your eyes (or at least, on paper!).
Every time you encounter a new reaction, remember this chat. You've got this! Chemistry is all about understanding how the world around us works, on a tiny, atomic scale. And that’s pretty darn amazing if you ask me. So go forth, be curious, and remember that even the most complex things can be broken down into understandable, and dare I say, fun pieces. You’re not just learning chemistry; you’re unlocking the secrets of the universe, one reaction at a time. And that, my friends, is something to smile about!
