Consider The Molecule: Specify The Hybridization Of Each Carbon Atom.

Let's talk about molecules. You know, those tiny building blocks of everything. They’re pretty important, right?
And within these tiny worlds, we find carbon atoms. They’re like the superheroes of chemistry. Always busy, always bonding.
Now, these carbon atoms have a secret life. A life of hybridization. Sounds fancy, doesn’t it?
It's like they have different "personalities" depending on who they're hanging out with. And that affects how they behave.
Our unpopular opinion? We should all be a little more aware of this. It’s not just for super-smart science folks.
Think of it like dating. Sometimes you’re all about one person, super focused. Other times, you’re spread a little thinner.
Carbon atoms do something similar with their electrons. They can get all cozy and mixed up. It’s called hybridization.
And for each carbon atom, we can tell you its specific hybridization. It’s like giving each one a little ID tag.
So, when we look at a molecule, we can point and say, "Aha! That carbon atom is sp3 hybridized!"
It’s like being a detective for molecules. Peeking into their atomic arrangements. Discovering their inner workings.
And this specific detail, the hybridization of each carbon atom, is actually quite telling. It dictates the molecule's shape.
It determines how strong its bonds are. And how it will react with other molecules. It’s all connected.
Let’s start with the most common one. The “chilled out” carbon. This one is sp3 hybridized.
Imagine a carbon atom with four single bonds. It’s like it’s got four best friends, and it’s giving each of them its full attention.
This carbon has a happy, tetrahedral shape. Think of a perfectly balanced tripod. Everything is spread out evenly.
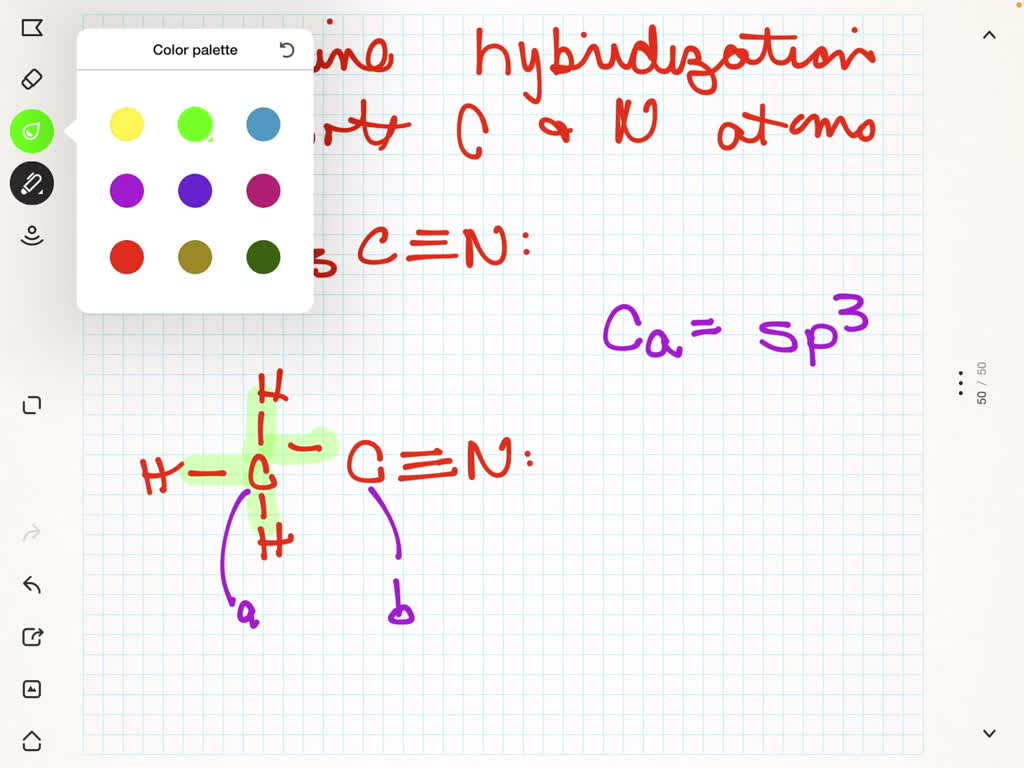
Examples are everywhere! Think of methane. That’s one carbon, four hydrogens. All single bonds.
Or the carbon atoms in ethane. Two carbons, six hydrogens. Still all single bonds for those carbons.
Even in larger molecules like glucose, many carbons will be in this sp3 state. They’re the backbone, the steady workers.
Then we have the "a little more intense" carbon. This one is sp2 hybridized.
This carbon is involved in a double bond. So, it’s sharing more of its electron love. It’s a bit more popular.
Instead of four separate friends, it’s got three main connections. One of those connections is a double bond.
This leads to a trigonal planar shape. Think of a perfectly flat pizza. All the toppings are in one layer.
A classic example is the carbon atoms in ethene. That’s where the double bond lives.
And in molecules like benzene, all the carbon atoms are sp2 hybridized. That’s what gives benzene its special stability.
Think of the carbon atoms in the rigid rings of many organic compounds. They are often sp2.
Finally, we have the "super focused, almost intense" carbon. This one is sp hybridized.
This carbon atom is involved in a triple bond. Or two double bonds! Talk about commitment.

It’s like it’s decided to share its attention with only two main directions. Very economical.
This results in a linear shape. Think of a perfectly straight train track. No detours allowed.
The carbon atoms in ethyne, also known as acetylene, are perfect examples. They’ve got a triple bond.
And in molecules with multiple double bonds in a row, like allenes, you’ll find sp hybridized carbons.
So, when we’re looking at a molecule, we can go through it, carbon by carbon. And assign its hybridization.
It’s like a fun little game of chemical bingo. Each carbon gets its unique label. sp3, sp2, or sp.
And this isn’t just some abstract concept. It has real-world implications. It affects everything.
The flexibility of your muscles. The way your DNA is structured. Even the taste of your food.
It’s all thanks to these little carbons and their varied hybridizations. Their different ways of bonding.
So, next time you’re looking at a chemical structure, don’t just see a jumble of lines and letters.
Try to see the individual carbon atoms. And then, my friends, try to specify their hybridization.
You can be the cool one at the party who casually mentions, "Oh, this molecule? Yes, that carbon there is definitely sp2."

It's a small detail, yes. But it's a detail that unlocks a whole universe of understanding.
It’s like knowing the secret handshake of the molecular world. A little bit of insider knowledge.
And once you start looking, you’ll see it everywhere. In the air you breathe. In the water you drink.
The carbon atoms are doing their thing, in their specific hybridizations. Shaping our reality.
So, let’s appreciate the humble carbon atom. And its incredible versatility. Its ability to change its electron game.
It’s not just about the bonds; it’s about how those bonds are formed. And that’s where hybridization comes in.
Each carbon atom has its story to tell. And its hybridization is a key chapter.
Whether it’s the tetrahedral sp3, the planar sp2, or the linear sp. Each tells a tale of electron arrangement.
It's a reminder that even in the smallest things, there’s complexity and beauty. And a lot of very clever arrangement.
So, the next time you encounter a molecule, remember this. You can indeed specify the hybridization of each carbon atom.
It’s a fun challenge. A way to connect with the molecular world on a deeper level.
And who knows, you might just impress yourself. And maybe a few others.

It's a little bit of chemistry knowledge that's both useful and, dare we say, a tad bit glamorous.
After all, understanding molecular structure is like understanding the architecture of life itself. And it all starts with those carbons.
So, consider the molecule. And then, bravely, specify the hybridization of each carbon atom. You can do it!
It's not rocket science. It's just really, really small science.
And each carbon atom, with its particular hybridization, is a tiny, crucial piece of the puzzle.
It’s the unseen detail that makes all the difference. The difference between a flexible chain and a rigid ring.
So, let’s embrace this understanding. Let’s celebrate the different "personalities" of carbon.
The sp3 carbons, happy and spread out. The sp2 carbons, in their flat, organized groups. And the sp carbons, sticking to their straight and narrow path.
It’s a molecular ballet. And hybridization is the choreography.
So, go forth, and analyze those carbon atoms. Their hybridizations await your keen observation.
It’s a simple step, but it leads to a profound appreciation for the world around us. A world built atom by atom.
And for each of those atoms, especially carbon, its hybridization is a key identifier. A fundamental characteristic.
So, let’s not shy away from it. Let’s invite it in. Let’s make knowing the hybridization of each carbon atom our little, fun secret.
