Consider The Following Reaction At Equilibrium
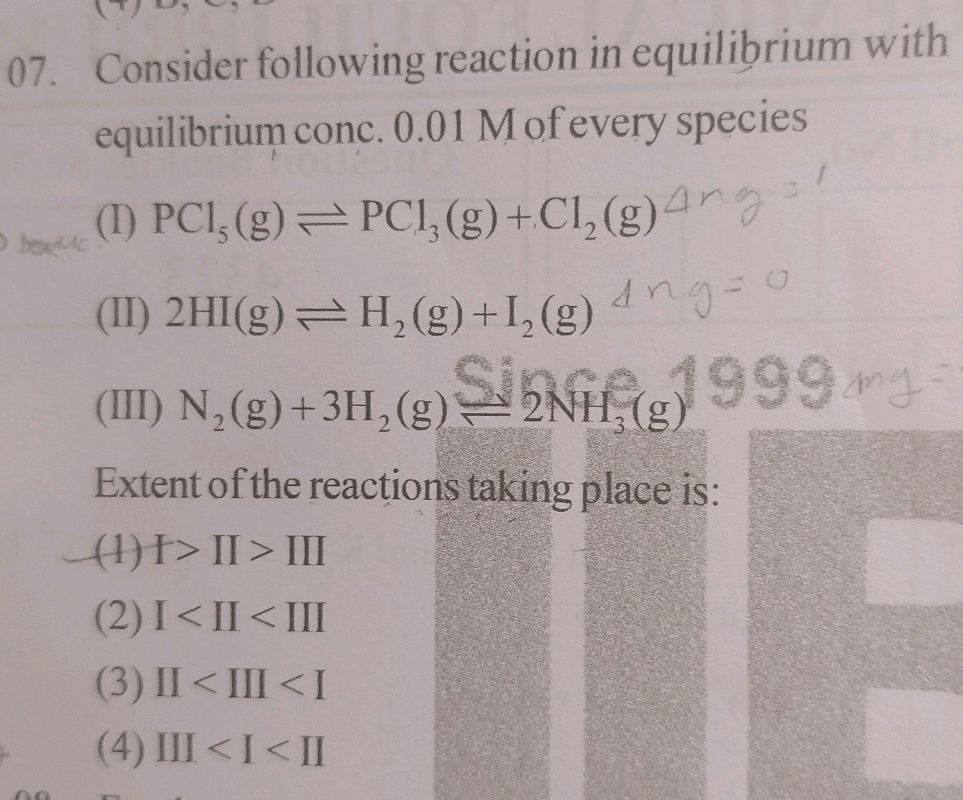
Hey there, science explorers! Ever found yourself staring at something and just thinking, "Whoa, how does that even work?" Well, today we're diving into a little something that might sound a tad academic at first, but trust me, it's got some seriously cool vibes. We're going to be pondering this: Consider the following reaction at equilibrium. Yeah, I know, thrilling, right? But stick with me, because this "equilibrium" thing is basically the universe's way of saying, "Everything's chill, for now."
So, what in the world are we talking about when we say "reaction at equilibrium"? Imagine you’ve got a busy kitchen. People are making sandwiches (that’s one reaction) and then other people are eating those sandwiches (that’s a reverse reaction). If the sandwich-makers are working just as fast as the sandwich-eaters, the number of sandwiches on the counter stays the same. They’re still making and eating, but the overall pile of sandwiches isn't growing or shrinking. That, my friends, is pretty much equilibrium. It’s not that nothing is happening; it's that the forward and reverse processes are perfectly balanced.
Think about it like a seesaw. When it’s perfectly balanced, no one’s going up or down, but both kids are still pushing and pulling, right? The forces are equal and opposite. In chemistry, it’s the same deal. We’ve got molecules zipping around, breaking bonds, and forming new ones. But at equilibrium, the rate at which reactants are turning into products is exactly the same as the rate at which products are turning back into reactants. It’s a dynamic dance, a constant hustle, but the net result? No visible change.
Why is this even a big deal?
Okay, so why should you care about a bunch of molecules having a perfectly balanced party? Well, understanding equilibrium is like having the cheat codes to a whole lot of natural and industrial processes. It helps us understand why some reactions go all the way to completion (like burning wood – once it’s ash, it’s pretty much done) and why others just… hang out. It's the secret sauce behind how our bodies function, how medicines work, and even how your favorite soda stays fizzy.
Let’s take a super simple example. Imagine you have a bunch of red marbles on one side of a table and blue marbles on the other. If you start moving red marbles to the blue side and blue marbles to the red side, at the same speed, you’ll eventually reach a point where the number of red marbles on each side is roughly the same, and the number of blue marbles is also roughly the same. You’re still moving marbles, but the overall distribution isn’t changing. That’s equilibrium in action!
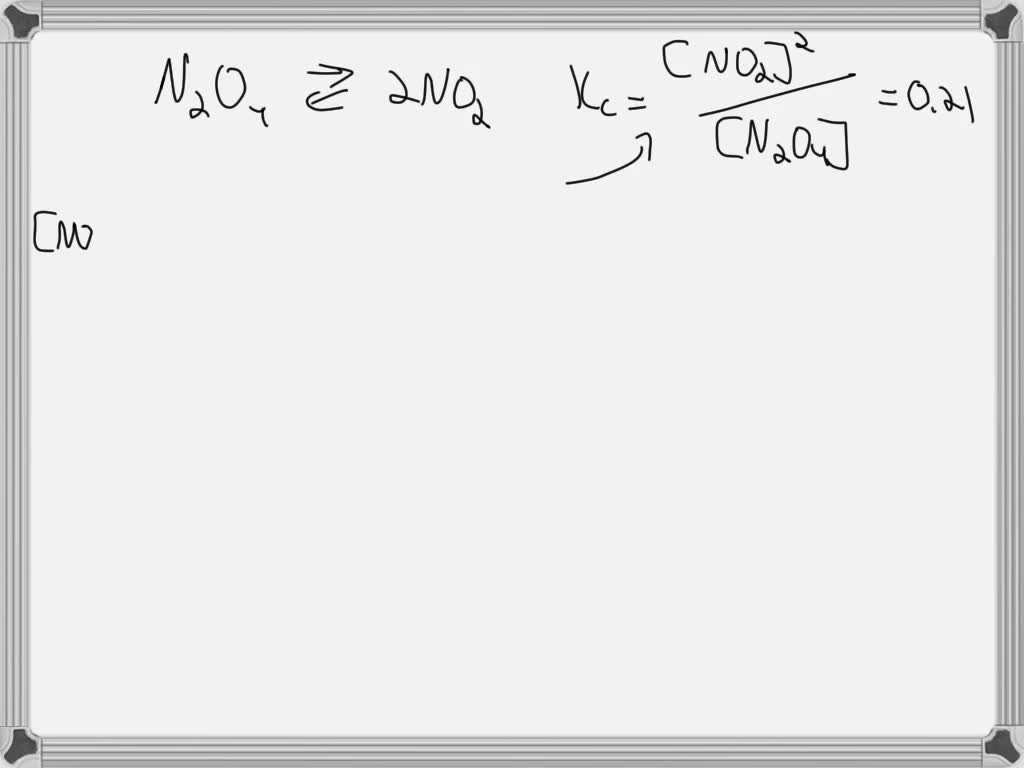
In chemistry, it’s not just about marbles. It's about atoms and molecules, the tiny building blocks of everything. When a reaction reaches equilibrium, it means the system has found its happy place. It’s not going to spontaneously shift one way or the other unless something external nudges it. And that "nudging" is where things get really interesting.
The Nudge Factor: Le Chatelier's Principle
So, what if we want to change things? What if we want more products? Do we just have to accept the equilibrium and shrug? Nope! Nature has a cool rule for this, and it's called Le Chatelier's Principle. Basically, if you mess with a system at equilibrium – say, by adding more of a reactant, removing a product, or changing the temperature or pressure – the system will try its best to counteract that change and get back to a new equilibrium. It’s like trying to balance a wobbly table; if you add weight to one side, you’ll instinctively adjust on the other to make it stable again.
Let’s say our sandwich shop is at equilibrium (sandwiches made = sandwiches eaten). If suddenly a huge catering order comes in (you add more reactants, i.e., sandwich ingredients), what happens? The sandwich-makers will speed up to meet the demand. The system will shift to make more sandwiches. Eventually, if the catering order is fulfilled, the shop will settle into a new equilibrium, maybe with a slightly higher rate of sandwich production overall.

Or, what if someone steals a bunch of already-made sandwiches (you remove a product)? To compensate, the remaining sandwiches will get eaten faster, and the sandwich-makers might even slow down a bit, all to get back to a balanced state. The system is constantly trying to find its footing.
This principle is super important for industries. Want to make more ammonia for fertilizer? You can play with the temperature, pressure, and concentrations of the ingredients to push the reaction towards making more ammonia. It’s like adjusting the settings on a thermostat to get your desired room temperature.

The Magic of Reversibility
The fact that these reactions are reversible is key. Not all reactions are. Some are like dropping a glass – once it’s shattered, you can’t just un-shatter it. But many chemical reactions are like water freezing into ice and then melting back into water. They can go both ways.
Think about when you boil water. You’re turning liquid water into steam (a forward reaction). But if you put a lid on the pot, some of that steam will condense back into liquid water (the reverse reaction). At a certain temperature and pressure, these two processes can happen at the same rate, and you’ve got equilibrium. The water level in the pot might not change, but there's a constant exchange between liquid and gas.
This dynamic nature of equilibrium is what makes it so fascinating. It’s not stagnation; it’s a state of perfect, ongoing balance. It’s the universe’s way of maintaining order in the midst of constant activity. So, next time you hear someone mention a "reaction at equilibrium," don't just picture something boring and still. Picture a bustling, perfectly coordinated dance floor where everyone’s moving, but the overall mood and energy remain constant. Pretty cool, huh?
