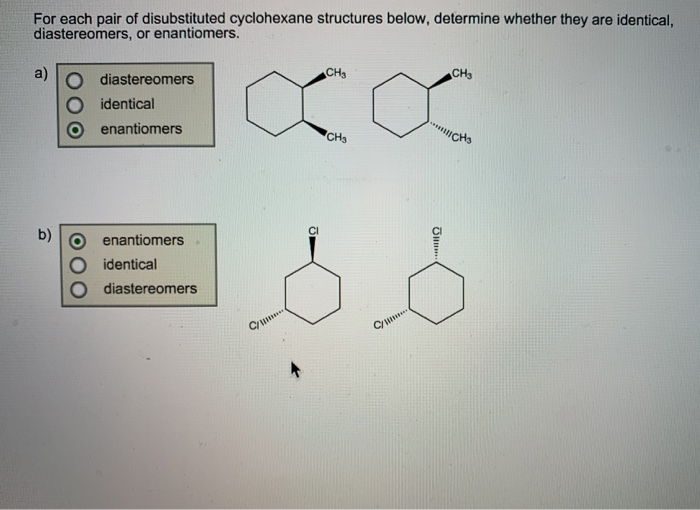
Consider The Disubstituted Cyclohexane Structures In Pair A.

Okay, let's talk about something that might sound a little… intimidating at first. We're diving into the world of disubstituted cyclohexane structures. Now, before your eyes glaze over and you start dreaming of simpler times, like when you just had to worry about whether your socks matched, hear me out!
Think of it like this: you've got this six-sided ring, a bit like a tiny, very organized merry-go-round. On this merry-go-round, we've attached two "things." These "things" are called substituents. They're like the little decorations you put on your Christmas tree. Some are sparkly, some are funny shapes, and they all add character.
Now, the pair A situation we're considering is where these two decorations are attached to specific spots on our little cyclohexane merry-go-round. It's not random! The universe has a plan, and in this case, the plan involves specific carbon atoms. We're talking about carbons that are neighbors, like the two friends who always sit next to each other on the bus.
So, you've got your ring, and you've got your two friends, the substituents. They're buddies, right there on adjacent seats. Easy peasy, you might think. But oh, the subtle complexities that unfold!
Here's where things get a little more interesting, and dare I say, a tad misunderstood. These substituents, even though they're on neighboring carbons, can have different orientations. Imagine our merry-go-round seats. One decoration can be sticking "up," and the other can be sticking "down." Or, maybe they're both sticking "up," or both sticking "down."
This is where the concept of stereoisomers waltzes onto the dance floor. These are molecules that have the same formula, the same atoms connected in the same order, but they're arranged differently in 3D space. It's like having two identical LEGO bricks, but one you build to face left and the other to face right. They're the same LEGOs, but they lead to different creations.
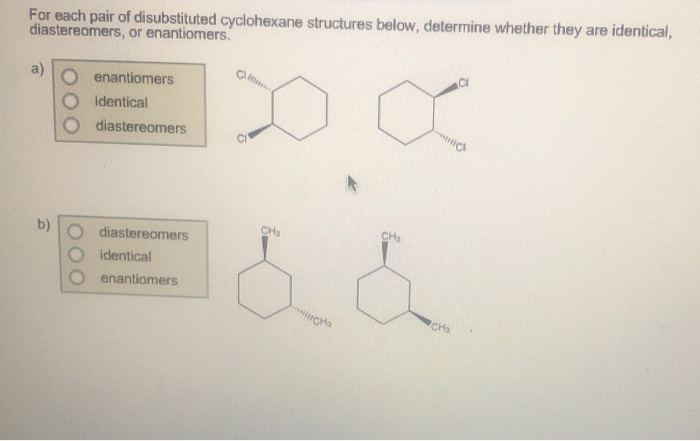
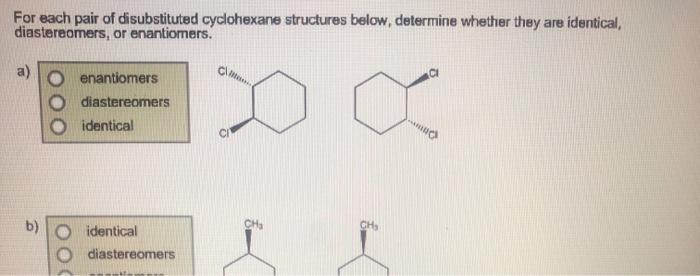
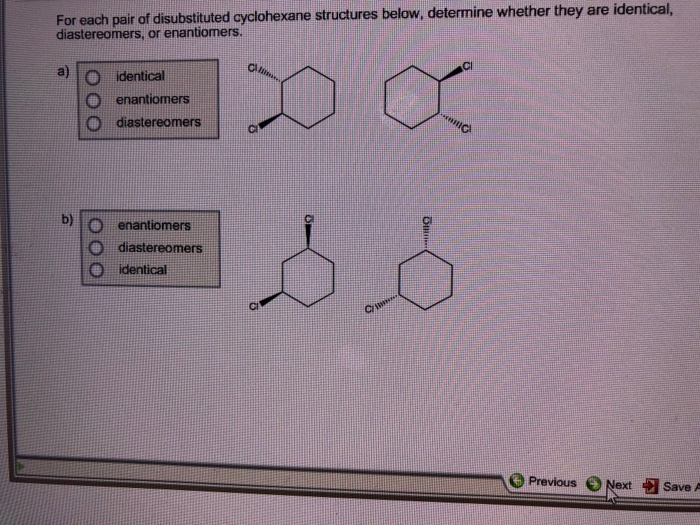
In our disubstituted cyclohexane scenario, these different 3D arrangements give us different structures. We have what are called cis and trans isomers. Now, these aren't like the song "Hey, Soul Sister" by Train. This is chemistry talk, and it’s surprisingly important.
The cis isomer is like when both your decorations are pointing in the same direction. Think of it as a coordinated dance move. Both dancers are doing the same step, facing the same way. They’re in sync.
The trans isomer, on the other hand, is when your decorations are pointing in opposite directions. It's like one dancer is doing a pirouette and the other is doing a moonwalk. They’re on the same stage, but their movements are a bit more… individualistic.
And this, my friends, is where my unpopular opinion might creep in. While chemists meticulously draw out these different forms, and they are, of course, scientifically significant, sometimes I feel like we overcomplicate the simple joy of having two things on a ring.
Think about it. We've got cis-1,2-disubstituted cyclohexane. That sounds like a mouthful, doesn't it? It's basically saying, "Hey, two things are on the neighboring seats, and they're both doing the same move." Simple enough!
Then we have trans-1,2-disubstituted cyclohexane. This is the one where the moves are opposite. Again, perfectly logical and important for understanding reactions and properties.
But let's not forget the real fun starts when we consider the chairs. Ah, the chairs! Cyclohexane rings aren't flat. They're all wiggly and wobbly, like a lazy teenager trying to avoid chores. They prefer to sit in a chair conformation.
Imagine our merry-go-round now has little chairs instead of just flat seats. Some of these chairs are pointing "up," and some are pointing "down." This is where the terms axial and equatorial come into play. It’s like having two types of seating: the "all the way up" seat and the "chilling on the side" seat.
In a chair conformation, the axial positions are like the high chairs, straight up and down. The equatorial positions are more like the comfy lounge chairs, sticking out to the sides.
Now, when our two substituents are on neighboring carbons in the cis arrangement, one will be axial and the other equatorial. Or, if they flip chairs (which they do, constantly!), they can both be equatorial or both axial in different chair forms. It's a bit like musical chairs, but with atoms!
For the trans isomer, it's a similar story, but the combination of axial and equatorial positions will differ. One might be axial and the other equatorial in one chair, and then flip to the other axial/equatorial combo when the ring wiggles.
And here's my lighthearted observation: sometimes, just looking at the diagrams, it feels like we're playing a very serious game of 3D Tetris. We're fitting these bits and bobs onto the cyclohexane ring, making sure they're not bumping into each other too much. Because, like any good party guest, these substituents don't like being too crowded.
When bulky substituents are in the axial position, they tend to get a bit grumpy. They're sticking straight up and down, right in the path of other things. This is called 1,3-diaxial strain. It's like having a giant inflatable dinosaur blocking the hallway – not ideal.
So, the cyclohexane ring, in its infinite wisdom, will try to place the bulkier substituents in the equatorial positions. This is where they have more elbow room, like finding a seat by the window at a crowded cafe. Ah, sweet relief!
This preference for the equatorial position is a fundamental principle that guides how these molecules exist and behave. It explains why one cis or trans isomer might be more stable than another.
Honestly, when I first encountered these disubstituted cyclohexane structures, I felt a bit overwhelmed. It was like being presented with a Rubik's Cube but being told I only had to solve two sides. Why so many variations? Why so many words for what seemed like simple attachments?
But then, I started to see the elegance in it. It's not just about attaching things; it's about how they relate to each other in space. It's about the subtle dance of molecular geometry, the constant jiggling of the ring, and the ultimate quest for stability.
My unpopular opinion, I suppose, is that while the detailed explanations are crucial for advanced chemistry, sometimes we can appreciate the core idea: we have a ring, and we have two decorations. The cis and trans tell us if they're on the same side or opposite sides. The axial and equatorial describe their orientation on the wiggly ring. And the whole point is that these arrangements matter.
They matter because they affect how molecules interact, how they react, and ultimately, what they can do. It's a reminder that even in the seemingly simple world of organic chemistry, there are layers upon layers of fascinating detail. And sometimes, just smiling at the intricate dance of these disubstituted cyclohexane structures is a perfectly valid way to learn.
So next time you see cis-1,2-disubstituted cyclohexane or trans-1,3-disubstituted cyclohexane, don't be intimidated. Think of it as a tiny molecular party, with guests on a wobbly dance floor, trying to find the best seats. It’s complex, yes, but also, in its own way, wonderfully playful and full of character.
And who knows? Maybe with a little humor and a lot less pressure, these molecular structures can become less like a daunting exam question and more like an interesting puzzle to unravel. We’re just adding some decorations to our ring, after all!
