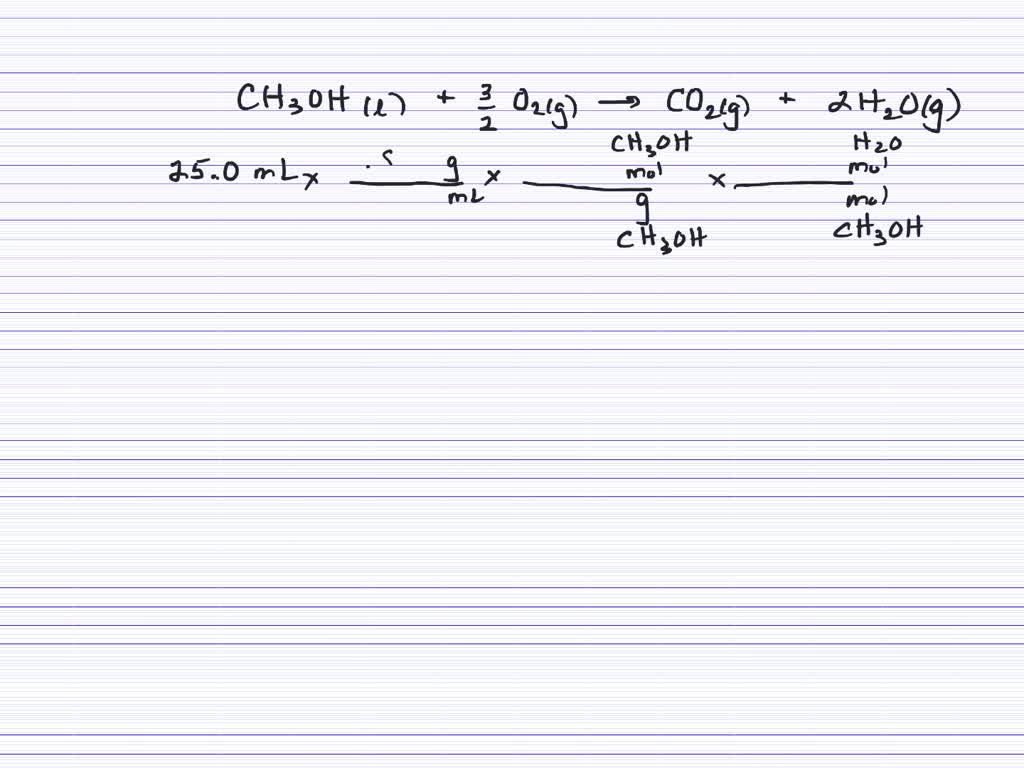
Consider The Combustion Reaction Between 25.0 Ml Of Liquid Methanol
Ever wondered what happens when you strike a match or when a car engine roars to life? It's all thanks to a fascinating chemical process called a combustion reaction. While the thought of chemical reactions might sound a bit intimidating, delving into combustion can be surprisingly fun and incredibly relevant to our everyday lives. Think of it as unlocking the secrets behind the heat and energy that power so much of our modern world, and even ancient discoveries!
The primary purpose of understanding combustion is to grasp how we harness energy. From cooking our food to lighting our homes and fueling our transportation, combustion is a cornerstone of human civilization. Its benefits are immense: it provides us with heat for warmth and cooking, light for visibility, and mechanical power for work. Learning about it helps us appreciate the science behind these conveniences and, on a larger scale, understand the fundamentals of energy production, efficiency, and even environmental impact.
In the realm of education, combustion is a staple in chemistry and physics classes. Students often conduct experiments involving burning fuels like wood or ethanol to observe the release of energy, the production of gases like carbon dioxide and water vapor, and the transformation of matter. This hands-on approach makes abstract concepts tangible and fosters critical thinking. Beyond the classroom, combustion is at play in our daily lives every single day. The gas stove you use to fry an egg, the fireplace that warms your living room on a cold night, and the gasoline powering your car all rely on controlled combustion. Even a simple candle flame is a miniature combustion reaction!
So, how can you explore this exciting topic without needing a full-blown chemistry lab? One simple and safe way is to observe a candle flame. Notice the wax melting and vaporizing, then reacting with the oxygen in the air to produce light and heat. You can also explore different materials that burn and compare how they combust – does a small piece of paper burn faster than a larger log? If you have access to a safe outdoor space, you might consider a small, controlled campfire (with adult supervision, of course!) and observe the different stages of burning wood. Another accessible idea is to research the combustion of liquid methanol. While you shouldn't experiment with it yourself without proper safety precautions, learning about its properties and how it burns can be a fascinating exercise. For instance, 25.0 ml of liquid methanol undergoing combustion would react with oxygen, releasing significant heat and producing carbon dioxide and water. Imagining this chemical dance can deepen your understanding of energy transformations.
Ultimately, understanding combustion is about appreciating the powerful chemical processes that have shaped our world and continue to be vital for our future. It’s a journey from the simple flick of a lighter to the complex engines that drive innovation, and it’s a topic that’s both profoundly practical and wonderfully engaging.
