Consider The Chemical Equation For The Combustion Of Sugar
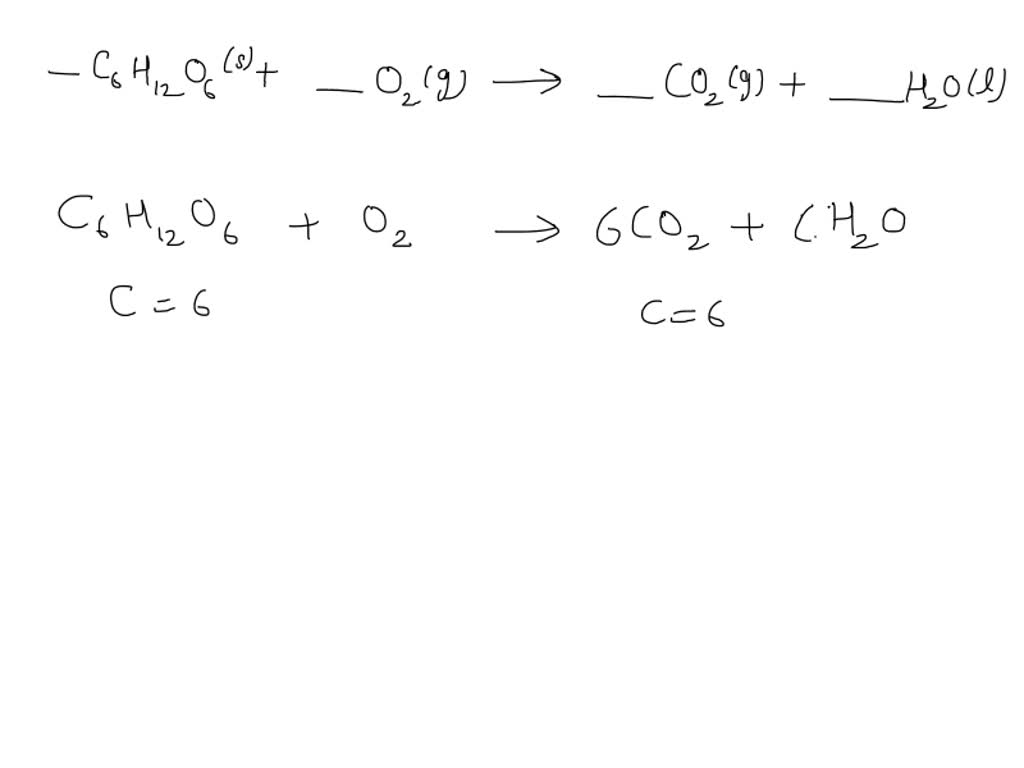
Ever looked at a bubbling pot of caramel or a perfectly toasted marshmallow and thought, "Wow, that's chemistry in action!"? Well, you'd be absolutely right! The humble chemical equation for the combustion of sugar, often written as C6H12O6 + 6O2 → 6CO2 + 6H2O + Energy, is more than just a bunch of letters and numbers. It's the secret sauce behind countless delicious transformations and a surprisingly inspiring concept for creators of all kinds.
For artists, hobbyists, and even the casually curious, understanding this fundamental reaction can unlock a world of creative possibilities. Think about it: the precise balance of ingredients, the controlled release of energy, the transformation from simple to complex. These are the very elements that fuel artistic endeavors! Whether you're a baker looking to perfect your créme brûlée, a sculptor inspired by the ephemeral nature of flame, or simply someone who enjoys the magic of a kitchen experiment, this equation offers a framework for understanding and innovating. It’s a reminder that even the most complex creations often stem from elegantly simple beginnings.
The applications are surprisingly diverse. Imagine a painter capturing the vibrant, fleeting colors of caramelized sugar, inspired by the release of energy as the molecules rearrange. Or a ceramicist creating glazes that mimic the glassy sheen of burnt sugar, a visual representation of the final products in the equation. Even writers can find inspiration, metaphorically speaking, in the way simple ingredients combine under specific conditions to produce something entirely new and often, quite beautiful. Think of it as a blueprint for transformation – from the raw potential of sugar and oxygen to the delicious aromas and textures that delight our senses.
Curious to explore this yourself? You don't need a lab coat to get started! The easiest way to experience the combustion of sugar is, of course, through cooking and baking. Try making simple caramel, watching as the sugar crystals melt and change color. Or experiment with making toffee or brittle. Pay attention to the stages: the initial melting, the bubbling, the browning, and the final solidification. Notice how heat is the catalyst, driving the reaction forward. Safety first, of course – always supervise children and be mindful of hot surfaces and liquids. A simple candy thermometer can also be a fun tool to understand the different stages of sugar transformation.
What makes delving into the combustion of sugar so enjoyable is its tangible nature. We can see, smell, and taste the results. It’s a direct link between abstract scientific principles and everyday sensory experiences. It’s a celebration of transformation, a testament to the fact that with a little heat and the right ingredients, something truly wonderful can emerge. So next time you're enjoying a sweet treat, take a moment to appreciate the silent, elegant dance of atoms that made it possible. It’s chemistry, yes, but it’s also pure, edible magic!
