Conductivity In A Metal Results From The Metal Having

Ever wonder why your spoon gets super hot when you leave it in your coffee? Or why those fancy copper pots are so darn good at cooking? It’s all thanks to something called conductivity! And when we’re talking about metals, it’s like they’re just built for this stuff. Think of metal as having a superpower. A really, really useful superpower. And this superpower is all about letting things flow.
So, what exactly is this flowy magic? In simple terms, metal’s conductivity means it’s really good at letting heat and electricity zip through it. Like a highway for tiny energy particles. It’s not like wood, which is a terrible conductor. If you put a wooden spoon in hot soup, it stays nice and cool for a while, right? That’s because wood is an insulator. It’s like a grumpy bouncer, saying "Nope, no energy allowed through here!"
Metals, on the other hand? They’re like the VIP lounge of energy transfer. They’re saying, "Come on in! All aboard the energy express!" And the reason they’re such rockstars is all about their tiny, zippy little particles. You know, atoms? Metals have a special arrangement. It's like they've got a bunch of loose electrons. Imagine a party where everyone’s holding hands, but then some people just let go and start dancing around. Those free-spirited dancers are the free electrons!
The Electron Party
This electron party is the secret sauce. In most materials, electrons are pretty tightly held by their atoms. They’re like kids who refuse to leave their parents’ side. But in metals, these electrons are liberated. They’re free to roam around the entire metal structure. Think of it as a giant, interconnected dance floor. When you add heat to one end of the metal, it’s like turning up the music. Those free electrons get excited and start bumping into each other, passing that energy along like a hot potato.
And electricity? It's pretty much the same story! An electric current is just a flow of electrons. Since metals have so many of these little guys just waiting to go, they can easily get nudged along by a voltage. It’s like giving a gentle push to a whole bunch of skaters on an ice rink. They start gliding!

So, when your electric kettle heats up so fast, it’s those speedy electrons doing the work. They’re grabbing onto that electrical energy and whizzing it through the metal wires, turning it into heat. It’s pure metal magic. And it happens ridiculously fast.
Why Are Some Metals Better Than Others?
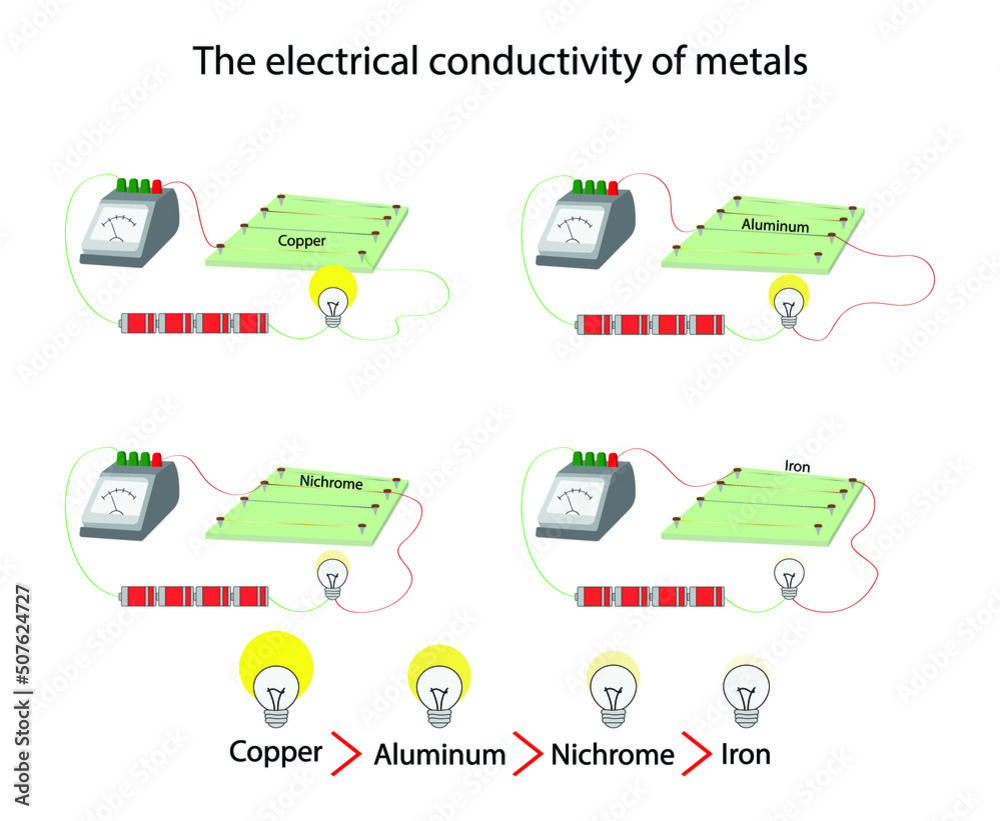
Now, not all metals are created equal in the conductivity game. Some are absolute champions, while others are more like… well, good, but not super good. Copper is a legendary conductor. It’s often used in electrical wiring for a reason. It’s like the Usain Bolt of electron movement. Then you have silver, which is even better than copper! But, you know, silver is a bit pricey for your toaster wires.
Gold is also an amazing conductor, which is why you see it used in high-end electronics and connectors. It’s shiny, it’s beautiful, and it lets the electrons flow with minimal fuss. It’s the diva of conductivity. It’s like, "Oh, you need me to carry electricity? Darling, it's my pleasure."

On the other hand, metals like iron are decent conductors, but not as brilliant as copper or silver. They’re more like the reliable, everyday folks. They get the job done, but they’re not setting any speed records. And then there are metals that are downright bad conductors, like lead. Lead is so unenthusiastic about letting things flow that it’s sometimes used for things where you don't want heat or electricity to go, like in some shielding.
The Quirky Details
Here’s a fun little fact: temperature affects conductivity. Generally, as you heat up a metal, its conductivity goes down. It’s like the electrons start getting a bit sluggish, like us on a hot summer day. They’re still dancing, but maybe at a slightly slower tempo. Conversely, when you cool metals down, their conductivity can actually increase. Some materials, when cooled to super, super low temperatures, become superconductors. They have zero resistance. Imagine a perfectly smooth, frictionless highway for electrons. That's superconductivity! It’s like the ultimate electron dream.
![Specific thermal conductivity of copper [12], aluminum and magnesium](https://www.researchgate.net/publication/314783498/figure/download/fig3/AS:471272007180290@1489371410312/Specific-thermal-conductivity-of-copper-12-aluminum-and-magnesium-compared-to-the.png)
This superconductivity thing is super cool. We’re talking about electricity flowing with absolutely no energy loss. Think of lightning zipping through a superconductor. No sparks, no wasted energy, just pure, unadulterated flow. Scientists are really excited about this because it could revolutionize things like power grids and high-speed trains. Imagine trains that float on magnetic fields powered by superconductors! It’s like a scene from a sci-fi movie, but it’s real science.
Another quirky detail is how impurities can mess with conductivity. Even a tiny bit of something else mixed into a pure metal can make it a worse conductor. It’s like someone crashing the electron party and tripping everyone up. The metal’s structure is disrupted, making it harder for those free electrons to do their thing. So, that’s why those super pure metals are often the best conductors.
Why Should We Care (Besides Hot Spoons)?
Okay, beyond the obvious of not burning your fingers, understanding conductivity is pretty neat. It’s why we have smartphones with their intricate circuits, why your fridge keeps your food cold (using heat transfer), and why power lines can carry electricity across vast distances. It’s the unsung hero of modern technology.
Think about it: the whole digital world runs on the back of this simple principle. Every time you send an email, watch a video, or play a game, you’re benefiting from the amazing conductivity of metals. It’s like the invisible glue that holds our tech-filled lives together.
And it’s not just about the practical stuff. There’s a certain elegance to it, isn’t there? This idea of matter having these inherent properties that allow for such amazing feats. Metals, with their generous offering of free electrons, are just naturally gifted at this energy passing game. They’re built for it. They’re the ultimate energy commuters.
So next time you’re using a metal object, whether it’s a pan, a wire, or even just a coin, give a little nod to those hardworking, free-roaming electrons. They’re the real MVPs, making sure our world keeps humming along. It’s a simple concept, really, but the implications are HUGE. And isn’t it fun to think about how even something as seemingly basic as a metal spoon has such a fascinating scientific story behind it? It’s a little bit of everyday magic, all thanks to conductivity!
