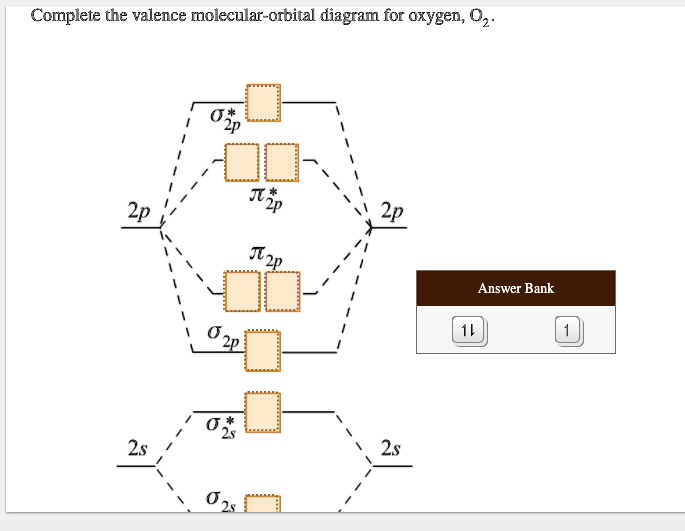
Complete The Valence Molecular-orbital Diagram For Oxygen O2
Get ready for a super fun dive into the world of tiny, invisible building blocks called atoms! We're about to crack the code on Oxygen (O2), the stuff we breathe to live, and see how its electrons are doing a cosmic dance. It's like peeking behind the scenes of a chemical rock concert!
Imagine atoms are like little party planners. They want their electrons, which are like enthusiastic party guests, to be super happy and organized. This is where our special tool, the Molecular Orbital Diagram, comes in. Think of it as the ultimate party seating chart for electrons!
We’re focusing on Oxygen (O2) today, that amazing molecule that keeps us all going. It’s made of two oxygen atoms holding hands, and we need to figure out where all their electrons are chilling. It’s way more exciting than it sounds, promise!
The Atomic Party Setup
Before we make a molecule, let's look at each individual oxygen atom. Each oxygen atom has a bunch of electrons, like little bouncing balls of energy. We need to know how these balls are arranged in their atomic "rooms" or atomic orbitals.
Think of atomic orbitals as different types of dance floors. We have the s orbitals, which are like a nice, round, open dance floor. Then we have the p orbitals, which are more like three interconnected trampolines, each going in a different direction. Super cool, right?
An oxygen atom has 8 electrons in total. These electrons fill up its atomic orbitals in a specific order, kind of like filling seats at a concert. The lower energy levels get filled first, because everyone wants the best view!
Making the Molecular Party Happen!
Now, when two oxygen atoms decide to become a molecule (O2), their atomic orbitals get together and mix and mingle. It's like their individual dance floors decide to merge and create a massive, super-duper molecular dance floor! This is where the magic of molecular orbitals happens.

These new molecular orbitals are not just simple copies of the atomic ones. They're completely new spaces where the electrons from both atoms can hang out together. It's like a collaborative dance party where everyone shares the space.
We get two main types of these new molecular dance floors: bonding orbitals and antibonding orbitals. Think of bonding orbitals as the super popular dance floor where everyone is having a blast and holding the molecule together. These are low-energy, happy places for electrons.
Then we have the antibonding orbitals. These are a bit like the "no-go" zones at a party, or maybe the emergency exit. Electrons in these orbitals are actually a bit disruptive and tend to pull the molecule apart if they hang out there too much. They have higher energy.
The Grand Electron Seating Chart
So, how do our 16 electrons (8 from each oxygen atom) get distributed in these new molecular orbitals? It's all about following the rules of electron happiness and energy levels, just like in the atomic orbitals.
We start filling the lowest energy molecular orbitals first. These are usually the bonding molecular orbitals. They’re the most stable and attractive places for electrons.
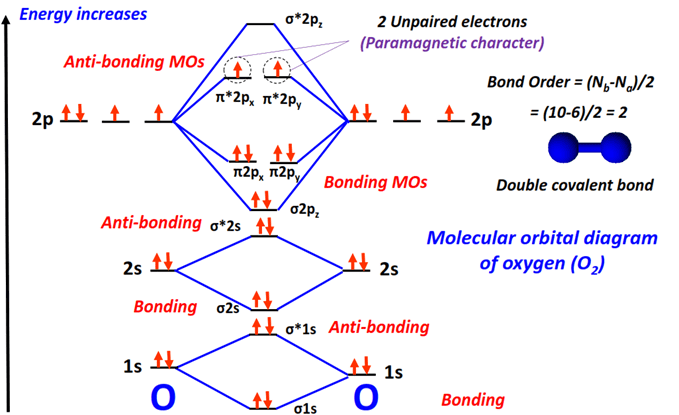
We have a couple of sigma bonding orbitals (let's call them σ2s and σ2p) that get filled first. These are like the VIP sections of the molecular dance floor. Electrons love being here because it’s super stable and makes the molecule strong.
Next, we fill the pi bonding orbitals (let's call them π2p). These are also fantastic places for electrons, contributing to the overall stability and creating that all-important chemical bond.
The Slightly Spooky Antibonding Zone
Now, things get a little more interesting. After filling up all the great bonding orbitals, we move on to the antibonding orbitals. These are the ones with a little star next to their names (like σ* and π), hinting that they're a bit more… dramatic.
The antibonding orbitals are higher in energy, meaning electrons are less thrilled to be there. They counteract the good vibes of the bonding orbitals.

We have sigma antibonding orbitals (σ2s and σ2p) and pi antibonding orbitals (π2p). These are like the energy sinks where electrons might end up if they have nowhere else to go.
The Final O2 Configuration!
Let’s count our electrons and place them carefully. We have 16 electrons in total for O2.
The σ2s bonding orbital takes 2 electrons.
The σ2s antibonding orbital also takes 2 electrons.
Then we have the three π2p bonding orbitals. Each can hold 2 electrons, so they take a total of 6 electrons.

Finally, we have the two π2p antibonding orbitals. These are the last ones to get electrons. And here's the super cool part: each of these gets 1 electron!
This leaves us with 16 total electrons accounted for! It's a beautiful, albeit slightly complex, arrangement.
Why Does This O2 Dance Matter?
This isn't just some abstract theory; it explains why Oxygen (O2) behaves the way it does! The fact that we have more electrons in bonding orbitals than in antibonding orbitals tells us that O2 is a stable molecule. Yay!
And the really mind-blowing part? The way the electrons fill the π*2p antibonding orbitals, each getting one electron individually, explains why O2 is paramagnetic. This means it's attracted to magnets, which is pretty wild for a gas we can't even see! It’s like our oxygen molecule has little magnetic personalities.
So, next time you take a deep breath, remember the incredible molecular dance happening inside that oxygen. It’s a symphony of electrons, all perfectly placed in their molecular orbitals, making life on Earth possible and even giving our air a little magnetic flair! Pretty awesome, right?
