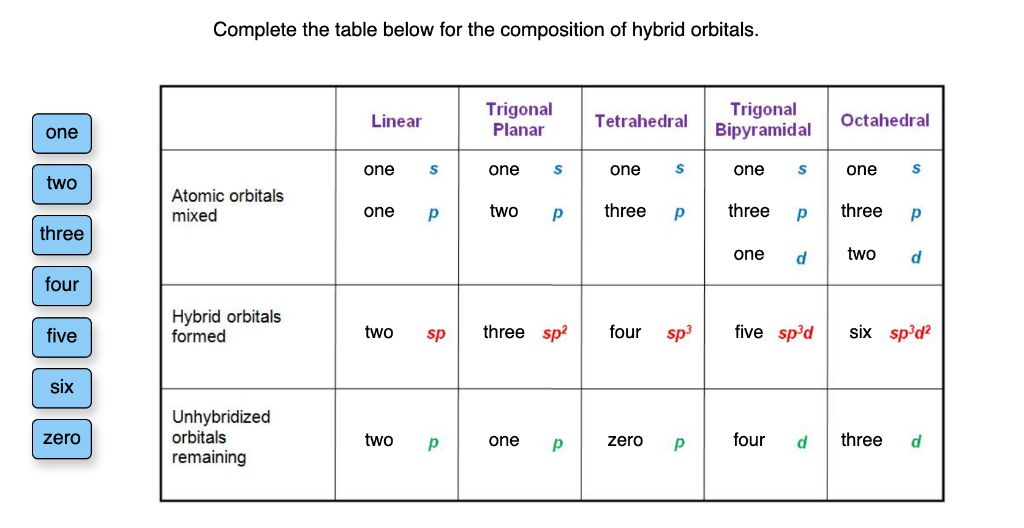
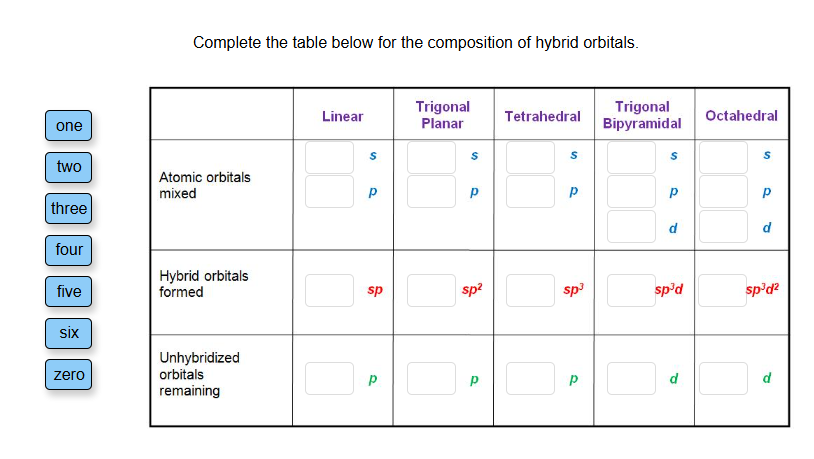
Complete The Table Below For The Composition Of Hybrid Orbitals.
Alright, so, let's talk about something that might sound a bit… scientific. But stick with me, because it’s actually like figuring out what goes into your absolute favorite, totally-hit-the-spot sandwich. You know how a killer sandwich isn't just bread and some random fillings? It’s a specific combination, a blend, that makes it sing? That’s kinda what hybrid orbitals are all about in the wacky world of chemistry.
Think of it like this: atoms are these tiny, tiny building blocks of everything. And these building blocks, they have these "rooms" where their electrons hang out. We call these rooms orbitals. Now, imagine you’ve got some plain old orbitals, like perfectly good, but maybe a little boring, single slices of cheese. Or maybe just the plain white bread. They’re fine, they do the job, but they’re not exactly setting the world on fire, are they?
But then, bam! Chemistry happens. And sometimes, these plain orbitals get together, they mingle, they mix, and they create something new, something better, something more… versatile. This mixing process is what we call hybridization, and the result? You guessed it – hybrid orbitals! It’s like taking your plain cheese slices and bread and deciding, “You know what? Let’s mash these up a bit. Let’s toast the bread, maybe add some mayo, and get some really good cheddar in there too!” You’re not just getting bread and cheese anymore; you’re getting a grilled cheese masterpiece. That’s hybridization for atoms.
Why do they do this, you ask? Well, just like you wouldn’t want your sandwich falling apart after the first bite, atoms want their electron arrangements to be stable and strong. Hybrid orbitals are often more energy-efficient and allow atoms to bond with other atoms in a much more effective way. It’s all about getting that perfect, secure fit, like a lid that snaps perfectly onto your Tupperware, no leaks allowed!
Now, the "composition" part is where we get a little more specific, just like detailing the exact ingredients of that epic sandwich. We need to know what goes into making these hybrid orbitals. Think of it as listing out the bread, the cheese, the spread, and any extra goodies. We’re going to fill in a table, and it's going to be super straightforward, like following a recipe. This table is basically the ingredient list for different types of hybrid orbitals. We’re going to be looking at how many of the original, plain orbitals get mashed up to create the new, fancy hybrid ones. It’s a bit like counting how many scoops of ice cream you're adding to your cone, or how many marshmallows you're stuffing into your hot chocolate. You need to know the quantities!
The Grand Mash-Up: What Goes Into Our Orbital Smoothies?
Let's imagine we have our basic building blocks, our pre-mash-up orbitals. These are usually the s and p orbitals. The s orbital is like a perfectly round, unassuming ball. It's chill, it's simple. The p orbitals, on the other hand, are a bit more… shaped. They look like dumbbells, or sometimes like little balloons with two lobes. And they usually come in sets of three, oriented along different directions – imagine three little dumbbells pointing along the x, y, and z axes. They’re good, but they have their limitations when it comes to getting cozy with other atoms.
When an atom decides to get its hybrid on, it’s like it’s grabbing some of these plain s and p orbitals and throwing them into a cosmic blender. The number of original orbitals you toss in dictates the type of hybrid orbital you end up with. It’s a direct correlation, no surprises. It’s like if you add one scoop of something and one scoop of something else, you get a specific flavor. If you add two scoops of one and one of another, well, you get a different flavor entirely.
So, we’re going to look at a few common scenarios. Think of these as our go-to sandwich combos. We’ve got the basic, the classic, and the, let’s say, adventurous.
The "Just a Little Mix" - sp Hybridization
First up, we have the sp hybrid orbital. This is like deciding to make a peanut butter and jelly sandwich, but with a twist. You take one s orbital and one p orbital, and you blend them together. Imagine taking that round s ball and smooshing it with one of those dumbbell-shaped p orbitals. What do you get? Two new orbitals, called sp hybrids. These new hybrids are like a combination of the original two – they've got a bit of that roundness, but also that directional, dumbbell-like shape. They're not quite s, not quite p, but something entirely new and more useful for bonding. They're like the perfectly seasoned croutons on your salad – they add a certain something.
The resulting sp orbitals are linear, meaning they're oriented in opposite directions. This is great for atoms that like to form straight lines, like in a diatomic molecule, or when they're connecting to two other things in a straight shot. Think of carbon atoms bonded together in a simple chain – they often use sp hybridization to keep things nice and linear.
In our table, this would mean we started with 1 s orbital and 1 p orbital. And voilà! We end up with 2 sp hybrid orbitals. Simple math, really, but with atoms. It's like counting your fingers and toes, but way more impactful for the universe.
The "Classic Combo" - sp² Hybridization
Next, we move to sp² hybridization. This is where things get a little more interesting, like upgrading your basic hot dog to a gourmet sausage with all the fixings. For this blend, we take one s orbital and two p orbitals. So, you’re grabbing that round s ball and two of those dumbbell-shaped p orbitals. You toss them all into the cosmic blender. What pops out? Three new sp² hybrid orbitals! These orbitals are all equivalent in shape and energy, and they’re arranged in a flat, triangular shape, like a little propeller. This is perfect for forming bonds in a plane, like in molecules where atoms are arranged in a flat structure.
Think of it like building a sturdy little table. You’ve got three legs, all spread out evenly to support the tabletop. These sp² orbitals are brilliant for creating double bonds, where an atom shares two pairs of electrons with another atom. It’s all about that extra stability and connection. It’s like having a really good handshake that’s extra firm and reassuring. For example, in the common molecule ethylene (C₂H₄), each carbon atom uses sp² hybridization to connect to the other carbon atom and two hydrogen atoms. It’s a very common and stable arrangement.
So, for our table, this means we started with 1 s orbital and 2 p orbitals. And out come 3 sp² hybrid orbitals. It's like a baker meticulously measuring flour, sugar, and eggs – the proportions are key to getting that perfect cake!
The "All-In" - sp³ Hybridization
Finally, we have the sp³ hybrid orbital. This is the full monty, the everything-but-the-kitchen-sink approach. For this spectacular blend, we take one s orbital and all three p orbitals. Imagine taking that round s ball and all three dumbbell-shaped p orbitals, and really giving them a good whirl in the blender. The result? Four brand-new sp³ hybrid orbitals! These four orbitals are identical and they arrange themselves in a tetrahedral shape, pointing towards the corners of a tetrahedron. This is the ultimate for creating stable, single bonds in three dimensions.

Think of a perfectly balanced pyramid, or a sturdy tripod. These sp³ orbitals are incredibly important for forming the backbone of many organic molecules. For instance, in methane (CH₄), the carbon atom is bonded to four hydrogen atoms, and it uses sp³ hybridization to achieve this tetrahedral arrangement. It's like building with LEGOs; you want a solid foundation and interlocking pieces that fit perfectly. This hybridization is responsible for so much of the diversity of life as we know it!
In our trusty table, this means we started with 1 s orbital and 3 p orbitals. And, drumroll please… we get 4 sp³ hybrid orbitals. It's like making a triple-layer cake with extra frosting – you're using all your best ingredients to create something truly substantial and impressive.
Filling in the Blanks: Our Hybrid Orbital Recipe Card
Now, let's formalize this a bit with our table. Think of it as a very simple recipe card. We’re just noting the ingredients and the output. This is where we’ll summarize what we've just discussed. It’s the kind of thing you’d put on a sticky note on your fridge so you don’t forget how to make your favorite molecular snacks.
The table will likely have columns like: "Type of Hybrid Orbital," "Number of s Orbitals Used," "Number of p Orbitals Used," and "Total Number of Hybrid Orbitals Formed." It’s straightforward, no need to overthink it. Just like deciding if you want extra pickles on your burger – it's a simple choice with a clear outcome.

So, when you see the table, just remember the blending process. Each row is a different way atoms can get creative and make their electron arrangements more effective for bonding. It's not about the atoms being forced into these shapes, it's about them finding the most stable and energetically favorable way to interact. It's like choosing the comfiest pair of shoes – you go with what feels best!
The beauty of this is that it explains so much about why molecules have the shapes they do. The angles between bonds, the lengths of bonds, the overall structure – it all stems from the type of hybridization happening at the atomic level. It’s the fundamental reason why water molecules are bent, and why carbon can form such complex structures. It’s the silent architect behind the scenes, making sure everything fits together just right.
So, next time you look at a molecule, or think about chemical reactions, remember the humble act of hybridization. It’s the atomic equivalent of making a really good cup of coffee – you’re not just throwing beans and water together; you’re coaxing out the best flavors through a specific process. And the resulting hybrid orbitals are the unsung heroes that allow the universe to be as diverse and complex as it is. They’re the versatile tools in an atom's toolbox, ready to tackle any bonding challenge that comes their way. Pretty neat, huh?
And don't worry if it still feels a little abstract. Even the best chefs have to practice their recipes. The more you see it in action, the more it will click. It’s like learning to ride a bike; at first, it feels wobbly, but soon you’re cruising. Chemistry, at its core, is just a series of very, very small, very organized events. And hybrid orbitals are a prime example of that organization and efficiency. So, go forth and conquer that table with confidence. You’ve got this!
