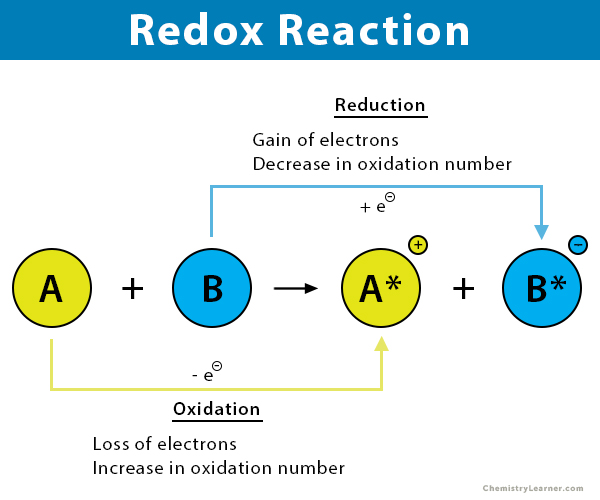
Complete The Statements About The Redox Reaction Below.
Ever wondered what makes a battery tick, why a piece of fruit turns brown, or how your body gets energy from food? The answer often lies in a fascinating and surprisingly everyday phenomenon called redox reactions! Think of it as a cosmic dance of electrons, where some atoms politely give them away and others happily snatch them up. It’s a fundamental process that powers so much of the world around us, and understanding it can make everyday observations feel a little more magical.
For those just starting out, grasping redox reactions can feel like unlocking a secret code to chemistry. It's less about memorizing complicated formulas and more about understanding the transfer of electrons. This knowledge is incredibly useful for anyone curious about how things work, from troubleshooting a dead phone battery to understanding how plants perform photosynthesis. Families can have a blast exploring simple experiments that demonstrate these concepts – imagine watching iron rust (a slow redox reaction!) or seeing how a lemon can power a tiny LED light. For hobbyists, whether you’re into DIY electronics, gardening, or even cooking, understanding redox can offer new insights and improve your results.
Let’s look at some examples. The most common one you probably encounter daily is rusting. When iron meets oxygen and water, it undergoes a redox reaction, forming iron oxide – that lovely reddish-brown stuff. Batteries are another prime example; the chemical reactions inside them involve the flow of electrons, which is the essence of a redox reaction, generating the electricity we use. Even the way your body digests food involves complex redox processes to extract energy. A variation you might see in action is combustion, like burning wood. This is a rapid redox reaction where fuel reacts with oxygen, releasing heat and light.
Getting started with redox reactions doesn't require a fancy lab coat. You can begin by simply observing the world around you. Pay attention to how metals tarnish or how food oxidizes. If you want to get hands-on, simple experiments are readily available. You can try making a "potato clock" using zinc and copper nails in a potato – the chemical reaction between them and the potato's electrolytes creates a small electrical current. Another easy step is to research common examples like oxidation of apples or the reaction in an Alka-Seltzer tablet. Look for educational videos online that simplify the concepts of oxidation and reduction.
Ultimately, exploring redox reactions is about gaining a deeper appreciation for the invisible forces that shape our world. It's a journey that can be both intellectually stimulating and surprisingly fun. So, next time you see a battery powering your device or a piece of metal starting to change color, you'll have a better understanding of the exciting electron exchange happening right before your eyes!
