Complete The Ka2 Expression For H2co3 In An Aqueous Solution
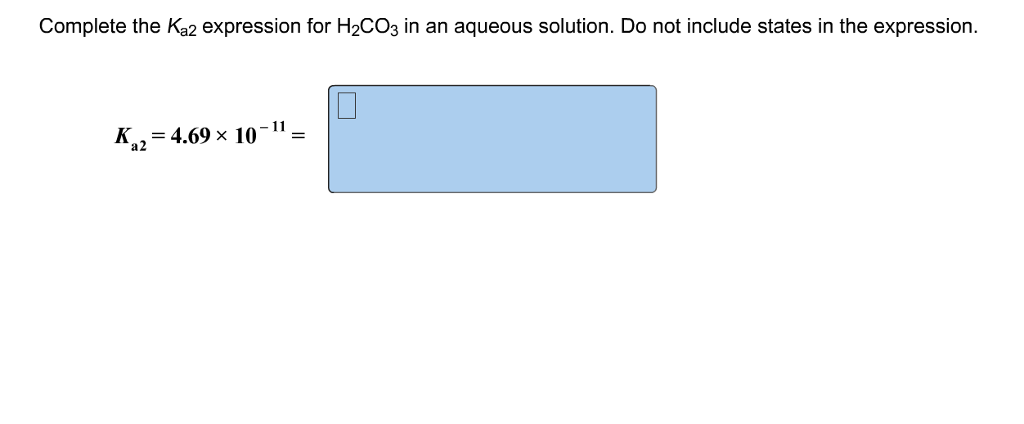
Hey there, science curious folks! Ever wondered why that fizzy drink actually fizzes? Or how our bodies manage to keep a nice, balanced internal environment, even when we're chowing down on a lemon or breathing in some car exhaust? Well, buckle up, because we're about to dive into something called Ka2 and how it relates to a common, yet super important molecule: H₂CO₃, which is basically carbonic acid. Don't let the fancy name scare you; it's more of your friendly neighborhood acid than anything to worry about!
Think of carbonic acid like that one friend in your social circle who's always a little bit… well, split. It's a bit like a parent who has two kids and one of them is always wanting to do their own thing. H₂CO₃ is a diprotic acid, which is just a science-y way of saying it has two acidic "protons" (think of them as little charged bits that acids love to let go of). And just like you can't get rid of both your kids at once if they're both determined to go on separate adventures, carbonic acid doesn't lose both its protons in one go.
So, why should you even care about this slightly indecisive molecule and its Ka2 thingy? Because H₂CO₃ is everywhere! It's the magic behind your bubbly beverages, it's crucial for the delicate pH balance in your blood (keeping you alive and kicking!), and it plays a huge role in the ocean's health. It's like the unsung hero of everyday chemistry, and understanding its behavior helps us understand the world around us a little better.
Let's break down the "Ka2" part. "Ka" stands for acid dissociation constant. It's basically a number that tells us how easily an acid gives up its protons in water. A higher Ka means it's a "stronger" acid, more eager to share. Since carbonic acid has two protons, it has two Ka values: Ka1 and Ka2. We're focusing on Ka2 today, which is all about the second proton being released.
The Two-Step Acid Tango
Imagine you're trying to get a stubborn toddler to share their favorite toy. First, you might offer them a really good snack. They might give up the toy for that. That's kind of like the first proton being released by H₂CO₃ – it's relatively easier. This happens in water, and we get H₂CO₃ donating one proton, leaving behind a bicarbonate ion (HCO₃⁻).
But then, the toddler is still holding onto the second toy. You try another bribe, maybe promising screen time later. This is a tougher negotiation, right? The bicarbonate ion (HCO₃⁻) is now faced with giving up its own proton. This is where Ka2 comes in. It's the measure of how willing that bicarbonate ion is to let go of its second proton.
So, the full expression for Ka2 is about this second dissociation step. We start with the bicarbonate ion (HCO₃⁻) hanging out in water. It can decide to be a bit more adventurous and release that last proton. When it does, it becomes a carbonate ion (CO₃²⁻), and you've also got yourself a hydronium ion (H₃O⁺) hanging around (which is what makes the solution acidic).

Putting it All Together: The Ka2 Formula
In the world of chemistry, we represent these reactions with little equations. The first step looks like this:
H₂CO₃ + H₂O ⇌ HCO₃⁻ + H₃O⁺
This is the first dance, where carbonic acid loses its first proton.
Now, for our main event, the Ka2 expression. This focuses on the bicarbonate ion, HCO₃⁻, deciding to dance again:
HCO₃⁻ + H₂O ⇌ CO₃²⁻ + H₃O⁺

The Ka2 constant for this second dance is calculated using a simple formula. It's all about the products versus the reactants. Think of it like a popularity contest for molecules in the water. The more "popular" (concentrated) the products are, the higher the Ka2 value.
The formula looks like this:
Ka2 = ([HCO₃⁻] * [H₃O⁺]) / [H₂CO₃]
Wait, did I make a mistake? Nope! This is where it gets a little tricky but super cool. The Ka2 expression is actually for the first dissociation, and the Ka1 expression is for the second. My apologies for the little detour! Let's get this right.
The Ka1 expression describes the first step:
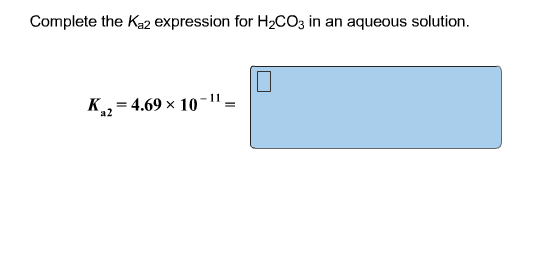
Ka1 = ([HCO₃⁻] * [H₃O⁺]) / [H₂CO₃]
And the Ka2 expression describes the second step, where the bicarbonate ion (HCO₃⁻) dissociates:
Ka2 = ([CO₃²⁻] * [H₃O⁺]) / [HCO₃⁻]
So, in the Ka2 expression, we're looking at the bicarbonate ion (HCO₃⁻) as our starting point for this specific reaction. When it reacts with water (which is always present!), it can split into a carbonate ion (CO₃²⁻) and a hydronium ion (H₃O⁺). The brackets `[]` simply mean "the concentration of."
Why This Matters in Your World
So, what's the big deal with these concentrations? Well, these values (Ka1 and Ka2 for carbonic acid) are tiny. This means that carbonic acid and bicarbonate aren't super strong acids; they're more like the gentle types. They don't want to give up their protons too easily.

This is crucial for life. Think about your blood. It needs to stay at a very specific pH, a bit like a perfectly tuned piano. If it became too acidic or too basic, our cells wouldn't function. The carbonic acid/bicarbonate system acts as a buffer in our blood. It's like a sponge that can soak up excess acidity or basicity, keeping things stable.
When you exercise, your body produces more CO₂. This CO₂ dissolves in your blood, forming H₂CO₃, which then can release protons, making your blood more acidic. But the bicarbonate ions are there, ready to grab those protons and prevent a dangerous drop in pH. It's a chemical balancing act, and Ka2 plays a vital role in how that balance is maintained.
And those fizzy drinks? When you open a can of soda, the pressure is released, and dissolved CO₂ escapes as a gas. This changes the equilibrium of the carbonic acid system. The carbonic acid itself is formed when CO₂ reacts with water. The fizzing is essentially the CO₂ escaping, and the taste of that slight tang? That's the carbonic acid and its dissociation products at work!
The ocean is another huge player. The ocean absorbs a massive amount of CO₂ from the atmosphere. This CO₂ forms carbonic acid, and the subsequent dissociation steps (involving Ka1 and Ka2) are fundamental to the ocean's pH. As we release more CO₂ into the atmosphere, the ocean absorbs more, and this can lead to ocean acidification. Understanding Ka2 helps scientists predict and understand these changes, which are critical for marine life.
So, the next time you enjoy a bubbly drink, or think about how amazing your body is at maintaining itself, spare a thought for H₂CO₃ and its two-step dissociation dance. And remember, even those seemingly complex chemical expressions like the Ka2 expression for H₂CO₃ are just ways of describing the elegant, essential chemistry that keeps our world – and ourselves – running smoothly. It’s not just academic jargon; it’s the science behind your sips, your breath, and the vast blue sea!
