Complete The Ka1 Expression For H2co3 In An Aqueous Solution

Hey there, lovely readers! Ever feel like your brain just needs a little… calm? Like, all those complex concepts you’re supposed to understand are just swirling around in a giant, confusing cloud? Yeah, me too. Especially when it comes to the nitty-gritty of science. But here’s a little secret I’ve learned to embrace: even the most intricate stuff can be broken down into bite-sized, totally manageable pieces. Think of it like making your favorite smoothie – you just add ingredients one by one, right? Today, we’re going to tackle something that might sound a tad intimidating: the Ka1 expression for H2CO3 in an aqueous solution. But trust me, by the end of this, you’ll be nodding along like you’ve got this chemistry thing down. Let’s dive in, shall we?
Unpacking the Mystery: H2CO3 and Its Aqua Adventures
So, what exactly are we dealing with here? H2CO3. That’s the chemical formula for carbonic acid. Now, don’t let the “acid” part scare you. Carbonic acid is actually pretty common and, in small doses, it’s even your friend. Ever enjoyed a fizzy soda? That pleasant tingle on your tongue? Yep, that’s carbonic acid at work. It’s formed when carbon dioxide (CO2) dissolves in water. It's like a dynamic duo, constantly interacting. Think of CO2 as a social butterfly at a party, and water as the welcoming host, always ready to chat.
The “aqueous solution” bit simply means it’s dissolved in water. Water, the universal solvent, the stuff of life! It’s where so much of the magic happens, from the chemistry in our own bodies to the reactions in a lab. So, when we talk about H2CO3 in an aqueous solution, we’re just talking about carbonic acid hanging out with its buddy, water.
The Ka1 Expression: What's the Deal?
Now, for the main event: the Ka1 expression. In chemistry, Ka is a constant that tells us how readily an acid will donate a proton (a tiny positively charged particle, basically a hydrogen atom that’s lost its electron) in water. Acids are all about giving away these protons. The higher the Ka value, the stronger the acid. Think of it like someone who’s super generous with their compliments – they just can’t help but spread the positivity!
The “1” in Ka1 specifically refers to the first proton that the acid donates. Carbonic acid, being a bit of a multifaceted molecule, can actually donate more than one proton. But for now, we’re focusing on that initial, crucial donation. It’s like the opening act of a great concert – setting the stage for everything that follows.
So, what does this expression actually look like? It’s a mathematical way of describing the balance of a reaction. When carbonic acid dissolves in water, it undergoes a reversible reaction. This means it can go forward (donating a proton) and backward (accepting a proton) at the same time. It's a constant dance, a never-ending back-and-forth.
Building the Expression, Piece by Piece
Let’s visualize this dance. We start with our carbonic acid (H2CO3) chilling in water (H2O). What happens? The carbonic acid decides to be generous and donates one of its hydrogen atoms. This hydrogen atom, now a proton (H+), hops over to a water molecule. When a water molecule gains a proton, it transforms into something called a hydronium ion (H3O+). And what’s left of the carbonic acid? It becomes a bicarbonate ion (HCO3-).
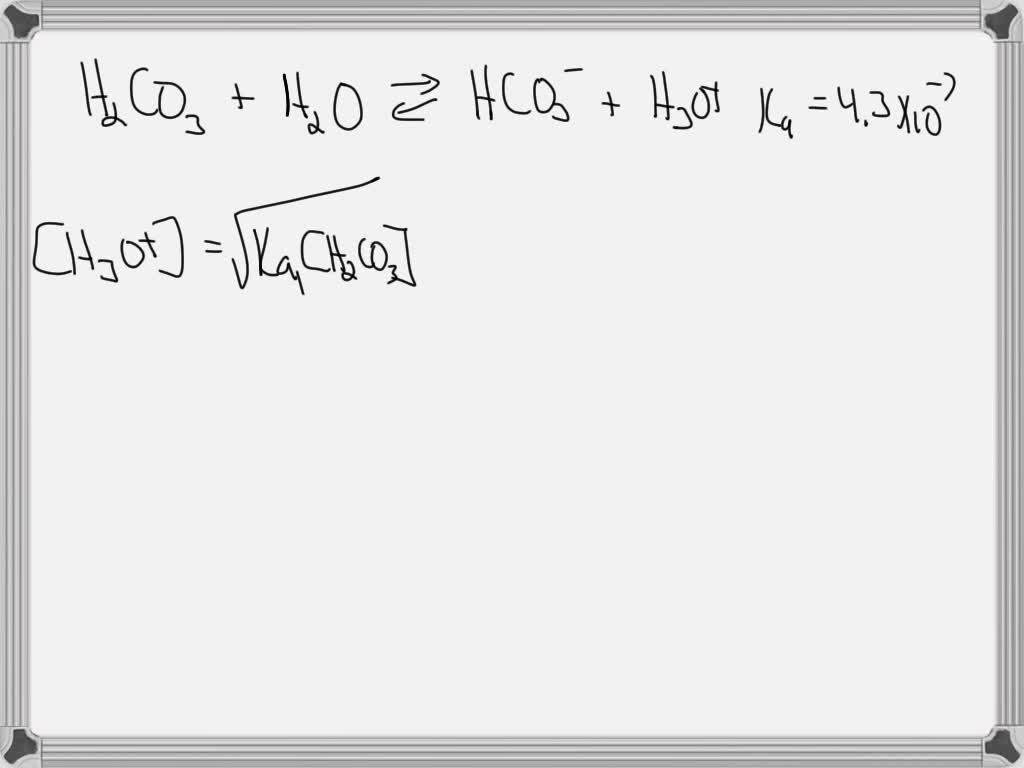
So, the reaction looks like this: H2CO3 (aq) + H2O (l) ⇌ H3O+ (aq) + HCO3- (aq)
Don't be intimidated by the symbols. (aq) just means "aqueous," dissolved in water. (l) means "liquid." The arrows (⇌) are super important – they signify that this is a reversible reaction, a constant equilibrium. It’s like a perfectly balanced seesaw, always finding its center.
The Magic Formula: Ka1
Now, how do we translate this reaction into our Ka1 expression? It's all about the concentrations of the products versus the reactants at equilibrium. The chemists who came up with this were pretty clever. They realized that if you know the concentrations of everything in the balanced equation when the reaction has settled down (reached equilibrium), you can calculate a value that tells you about the acid’s strength.
The Ka1 expression is essentially a ratio. It’s the product of the concentrations of the products divided by the product of the concentrations of the reactants. Let's break it down:
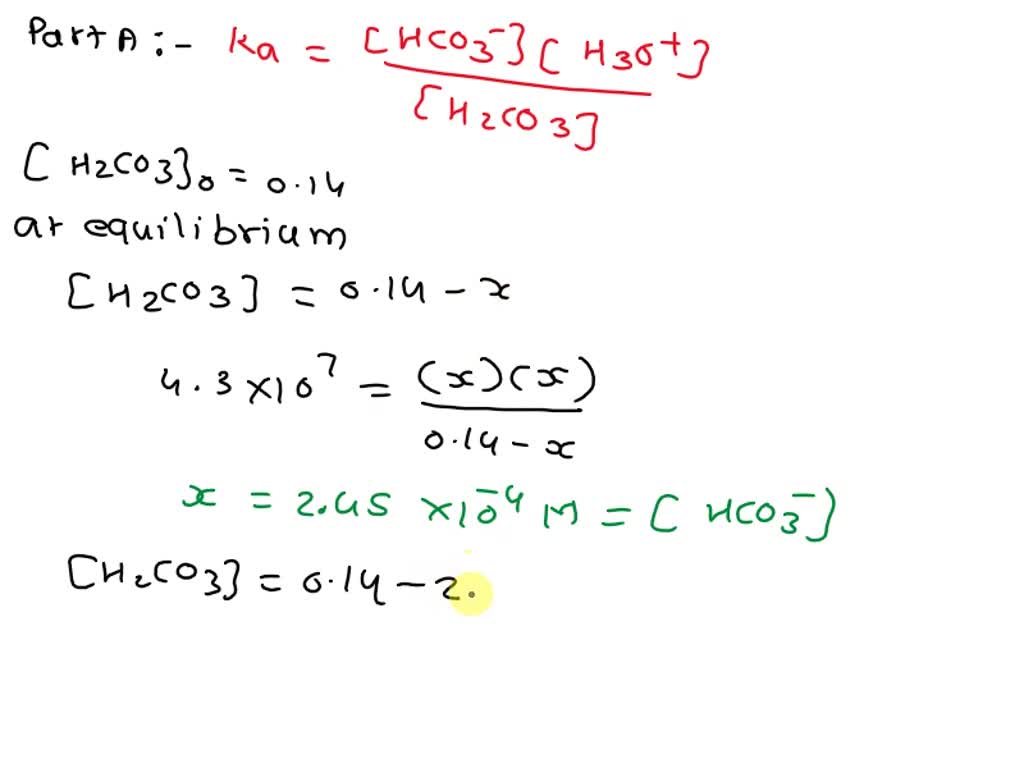
Numerator (Products):
- Concentration of hydronium ions: [H3O+]
- Concentration of bicarbonate ions: [HCO3-]
Denominator (Reactants):
- Concentration of carbonic acid: [H2CO3]
- Concentration of water: [H2O]
Wait a minute! What about the concentration of water? You might be wondering. Well, in most aqueous solutions, water is the solvent and its concentration is so incredibly high and changes so little during the reaction that it's considered a constant. So, for simplicity, we usually don't include it in the Ka expression. It’s like leaving out the air in a room when you’re talking about the furniture – it’s always there, but not the focus of the arrangement.
Therefore, the complete Ka1 expression for carbonic acid in an aqueous solution is:
Ka1 = ([H3O+][HCO3-]) / [H2CO3]
And there you have it! The elegant, yet simple, Ka1 expression. It's a snapshot of the acid's behavior in water, a little window into its personality.
Why Does This Even Matter? Real-World Coolness
Okay, so we’ve got this expression. But why should you care? Is this just for folks in lab coats? Absolutely not! Understanding Ka values, even for something as seemingly obscure as carbonic acid, has real-world implications. Think about it:
The Power of pH: Your Daily Dose of Balance
The Ka1 expression is directly related to the pH of a solution. pH is that scale we use to measure how acidic or basic something is. From the shampoo you use to the way your body maintains its internal balance, pH is everywhere. Carbonic acid plays a crucial role in regulating the pH of our blood, which is vital for our survival. If the pH of your blood shifts even slightly, it can have serious consequences. So, that seemingly dry chemical equation is actually a key player in keeping you alive and kicking!
Beyond Blood: Environmental Chemistry
Carbonic acid is also a big deal in our environment. It’s involved in the weathering of rocks, a slow but powerful process that shapes our landscapes. It's also a major factor in ocean acidification. As the oceans absorb more CO2 from the atmosphere (thanks, industrial revolution!), more carbonic acid forms, making the water more acidic. This has a significant impact on marine life, especially organisms with shells and skeletons made of calcium carbonate, like corals and shellfish.
Imagine a world where oysters struggle to build their homes. That's a direct consequence of changes in carbonic acid levels. It’s a stark reminder that even the smallest chemical reactions can have monumental global effects. It’s like a butterfly flapping its wings in Brazil and causing a hurricane in Texas, but on a molecular scale.
Fun Fact Alert!
Did you know that carbonic acid is also present in your saliva? It helps to buffer the acids produced by bacteria in your mouth, protecting your teeth. So, next time you feel a tingle from your morning coffee, remember that your saliva is working overtime to keep your smile intact, thanks in part to the magic of carbonic acid and its equilibrium!
A Moment of Reflection: The Beauty of Interconnectedness
As we wrap up our little exploration into the Ka1 expression for H2CO3, I hope you’ve realized that science isn't just a collection of dry facts and figures. It’s a beautiful, interconnected web of processes that influence our world in profound ways. From the fizzy comfort of a soda to the delicate balance of our planet's ecosystems, these chemical principles are at play.
Thinking about the Ka1 expression is like looking at a tiny, intricate clockwork mechanism. Each gear, each spring, has a specific role, and together they create something functional and beautiful. And just like a well-oiled clock can tell us the time with precision, this chemical expression helps us understand the behavior of acids and the intricate dance of molecules in water.
In our busy, often chaotic lives, it’s easy to feel disconnected. But by taking a moment to understand these fundamental principles, we can begin to see the underlying order and elegance that governs everything. It reminds us that we are all part of a larger system, a grand, ongoing experiment. So, the next time you’re sipping on that bubbly beverage or enjoying a crisp, clear day, take a moment to appreciate the silent, invisible chemistry that makes it all possible. It’s a reminder that even the most complex things can be understood, one simple expression at a time. And that, my friends, is a truly calming thought.
